Org 2 Summary Sheets
Org 2 Summary Sheets – A 20-Page PDF Summary of a Typical Org 2 Course
- These sheets provide a condensed summary of an entire typical Org 2 course, so you can access the key concepts and reactions from your textbook in just a few pages.
- Perfect for getting up to speed on your course if your midterm or final is fast approaching.
- Full-color, printable PDF file. No physical item will be mailed to you.
- Designed for introductory organic chemistry courses at all levels in the USA and Canada (including ACS exams). Also useful as a refresher on the basics.
These sheets are available for download with the MOC Membership. (Alternatively they can be purchased separately from the online store).
MOC members will see a download link below (must be logged in to see it).
MOC Org 2 Summary Sheets
17-page PDF · Full color
[Download link hidden - Join here]
What’s Included in the Org 2 Summary Sheets
Alcohols and Ethers • Molecular Orbitals and Conjugation • Reactions of Dienes • Diels-Alder Reaction • Pericyclic Reactions • Aromaticity • Reactions of Aromatic Compounds • Nucleophilic Aromatic Substitution • Aromatic Synthesis • Organometallics • Aldehydes and Ketones • Carboxylic Acids • Oxidation and Reduction • Enolates • Amines • Carbohydrates • Amino Acids
- Introduction to Alcohols and Ethers (free sample download) – Alcohols and alkyl halides – Williamson Ether synthesis – 3 more ways to make ethers – Epoxides from alkenes – Ring opening of epoxides under acidic and basic conditions – Oxidation of alcohols – Replacement of alcohols with halides using SOCl2 or PBr3 – Conversion to tosylates – Reduction of esters, carboxylic acids, aldehydes, and ketones

- Reactions of Dienes – Thermodynamic and kinetic control – Stability of alkenes and substitution – Isolated, conjugated and cumulated dienes – Resonance energy of dienes – 1,2 vs 1,4-addition to dienes – Kinetic and thermodynamic control in addition to dienes

- Dienes: Molecular Orbitals & Conjugation – Conjugation and pi-systems – pi bonding – Conjugation of carbo\cations, radicals and lone pairs – Counting p-orbitals and pi electrons – Constructing pi molecular orbitals for the “allyl” system – Identifying the HOMO and LUMO for molecular orbitals

- Diels-Alder Reaction – Diels-Alder Reaction – Dienes and dienophiles – s-cis versus s-trans – Cyclic dienes – Stereochemistry of the dienophile – Stereochemistry of the diene – The endo rule – Identifying exo and endo products
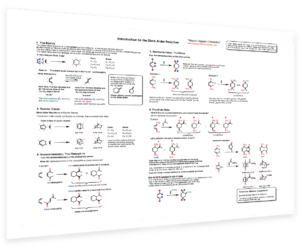
- Aromaticity – 4 rules for how to tell if a molecule is aromatic, antiaromatic, or non-aromatic – Resonance energy of benzene – Examples of aromatic compounds – Reactivity of aromatic compounds vs. non-aromatic compounds – Addition vs substitution reactions – Anti-aromaticity – Frost circles

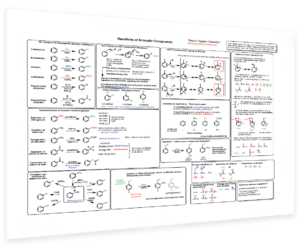
- Reactions of Aromatic Compounds – 6 key electrophilic aromatic substitution reactions – Mechanism of aromatic substitution – ortho, meta, and para directing groups – Why are groups ortho,para- or meta directing? – Activating vs deactivating – Steric effects – Drawing key resonance forms – Reactions of side chains – What to do when there are 2 or more groups on the ring
- Aldehydes and Ketones – A simple pattern for 7 key reactions of aldehydes – Electrophilic addition – Protonation – Grignard addition – 3 key factors affecting reactivity – Electronic and steric effects – Acid catalysis, synthesis by oxidation of alcohols – Synthesis of alkynes and alkenes – Protecting group formation – Imine formation

- Key Reactions of Carboxylic Acid Derivatives – Nucleophilic acyl substitution – The importance of the leaving group base strength – The addition-elimination mechanism – Carboxylic acids – Why do Grignards add twice to esters – Double addition – Synthesis of carboxylic acids from alcohols – Deprotonation – Conversion to acyl chlorides – Fischer esterification – Acidic hydrolysis of esters amides and nitriles

- Enols and Enolates – Acidity of the alpha-carbon – Structural features of the carbonyl group – Keto-enol tautomerism – Five factors that affect the keto-enol ratio – Factors that affect acidity of the alpha carbon – Acid catalyzes keto-enol interconversion – Conjugate addition – Enolates as nucleophiles

- Amines – Nomenclature of amines – How to understand amine basicity – Synthesis of amines from alkylation – Gabriel synthesis – Reduction of nitro groups – Reductive amination – Imine and enamine formation – Hofmann and Curtius rearrangements

- Carbohydrates – What’s a carbohydrate? – The Fischer projection – D and L – Aldoses and Ketoses – Chain and ring forms of sugars – Reducing sugars – Haworth projections – Converting Fischer projections to cyclic forms – Reactions of carbohydrates

Plus more summary sheets on amino acids, oxidation and reduction, molecular orbitals and conjugation, pericyclic reactions, aromatic synthesis, and more.
Join Master Organic Chemistry Membership Today