Reimer-Tiemann Reaction
Description: The Reimer-Tiemann reaction is a method for installing a formyl group (CHO) onto an electron-rich aromatic such as phenol using CHCl3 and strong base. Yields are generally not great.
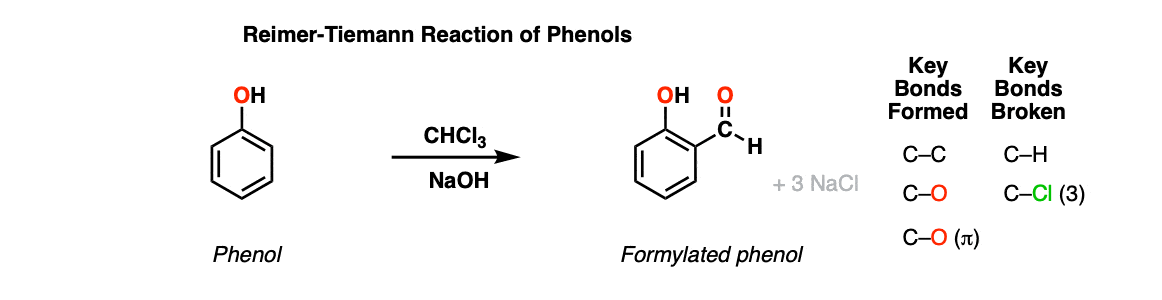
Notes: The reaction proceeds through formation of a dichlorocarbene from CHCl3 through deprotonation with a concentrated aqueous solution of NaOH followed by alpha-elimination.
In the reaction above, both ortho- and para– products are formed, although ortho– dominates.
See Also: Dichlorocyclopropanation
Examples:
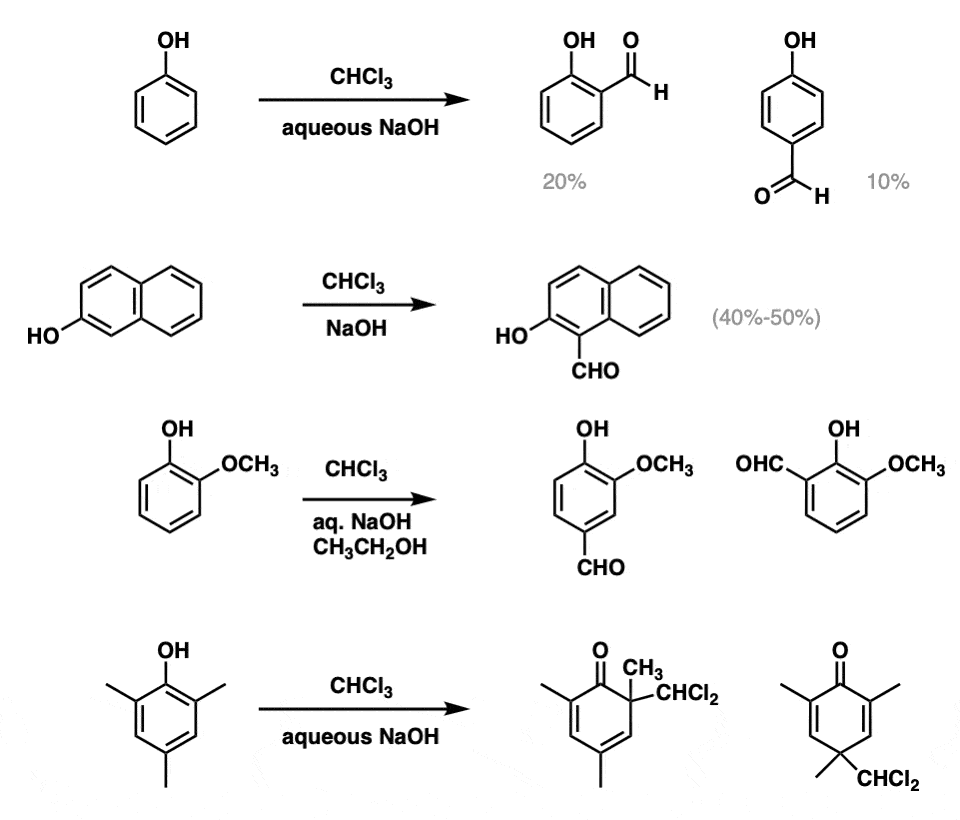
Notes: Note that this reaction only really works with very electron-rich aromatics (e.g. phenols) and yields are typically quite poor. A mixture of ortho– and para– products are obtained.
When the ortho and/or para positions are blocked (example 4) then dichloromethyl products are obtained.
Mechanism:
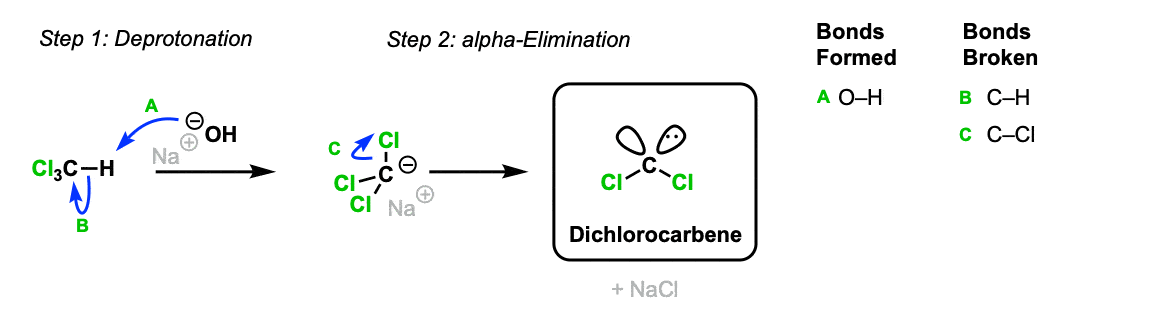
The first step is formation of a dichlorocarbene from chloroform. In the first step, hydroxide ion deprotonates chloroform to form its conjugate base, (Step 1, arrows A and B) which can then lose chloride to form the neutral dichlorocarbene.
Part 1: Formation of Dichlorocarbene
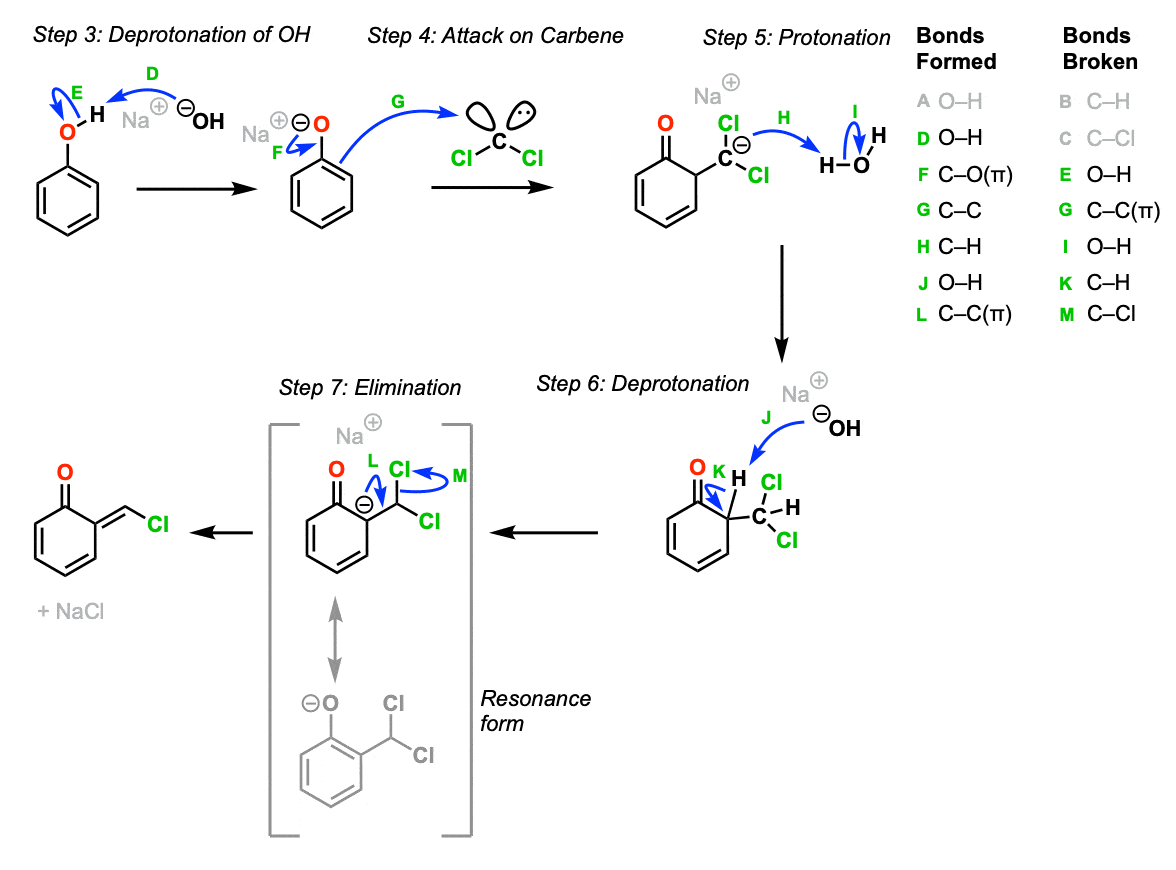
At the same time, phenol is deprotonated by strong base to give the phenoxide ion (Step 3, arrows D and E) which is a more reactive nucleophile than phenol itself.
Attack of the dichlorocarbene by the phenoxide ion (Step 4, arrows F and G) forms a new C-C bond and gives a carbanion. Protonation of the carbanion (Step 5, arrows H and I) followed by deprotonation adjacent to the ketone (Step 6, arrows J and K) gives another phenoxide. (These two steps are often just combined and described as “proton transfer”. ) Note that the phenoxide formed after step 6 has two prominent resonance forms. For simplicity, we’ve chosen to draw the final elimination step (Step 7, arrows L and M) as elimination from the carbanion resonance form, although you could certainly draw it coming from the phenoxide resonance form as well.
Part 2: Addition of Dichlorocarbene To The Aromatic Ring
The final sequence involves a conjugate addition/elimination reaction to give a product that tautomerizes to the final product.
In the first step, hydroxide ion performs a conjugate addition to the alpha, beta unsaturated ketone (Step 1, arrows A, B, and C). This results in an anion; we can draw resonance forms for both the phenoxide ion (O– ) and carbanion. In the next step, elimination of chloride ion occurs (Step 2, arrows D, E, F, and G) to give what is essentially a substitution product.
Deprotonation of the hydroxyl group (Step 3, arrows H and I) gives its conjugate base, which can then tautomerize to restore aromaticity to the phenol (Step 4, arrows J, K, and L). Protonation of the phenoxide (Step 5, arrows M and N) gives the final product.
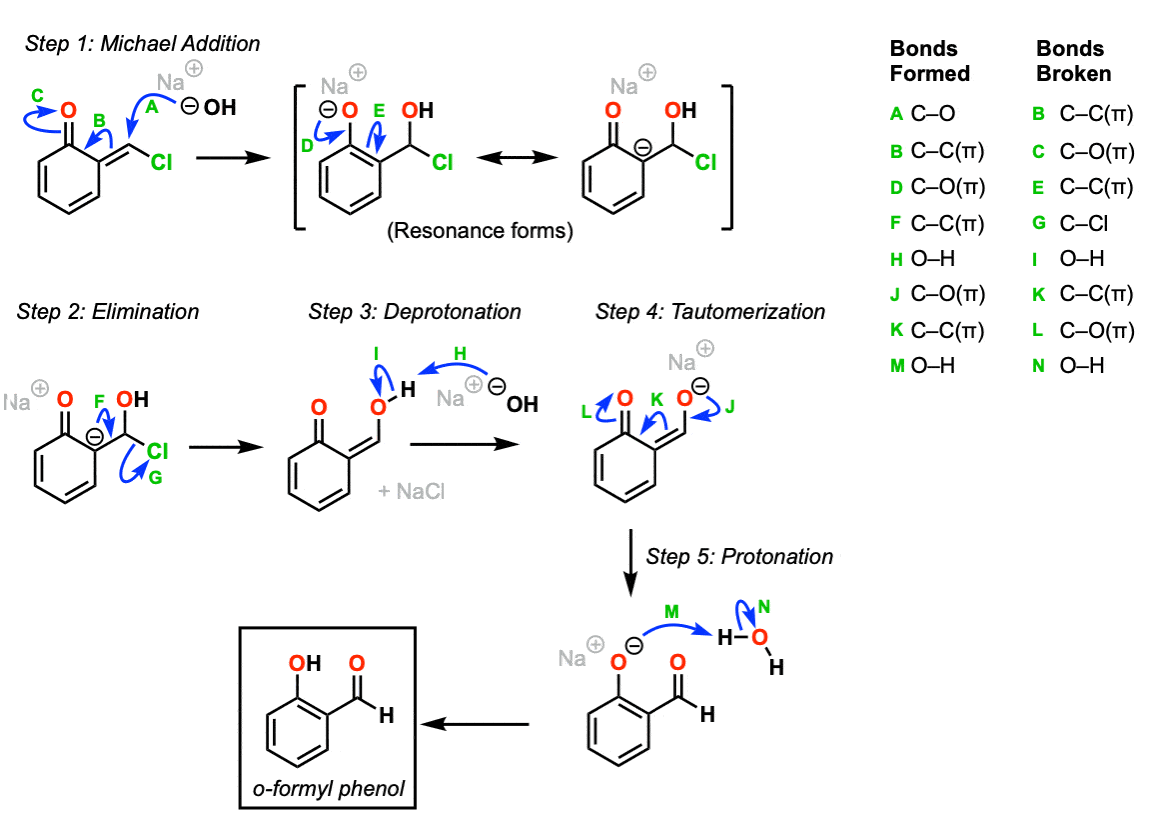
That’s a lot of work for a 20% yield.
Notes: As noted, you can draw the elimination step as arising from several different resonance forms; the carbanion was chosen here because it’s the simplest.
- No selectivity for double bond geometry (E/Z) is implied.
- The driving force for tautomerization is formation of the aromatic ring.
- Although the ortho product is shown here, the para product will be formed as well (not shown).
(Advanced) References and Further Reading
- The Reimer-Tiemann Reaction.
Hans Wynberg
Chemical Reviews 1960 60 (2) 169-184
DOI: 10.1021/cr60204a003
Early review on the RT, with discussion and mechanism. - The Reimer-Tiemann Reaction.
Dauben, W. G.
Organic Reactions, Volume 28. Hoboken, NJ: Wiley-Interscience. pp. 1–36
DOI: 10.1002/0471264180
Comprehensive treatment of this reaction, with every example up to this point. - The Mechanism of the Reimer-Tiemann Reaction
Jack Hine and James M. Van Der Veen
Journal of the American Chemical Society 1959 81 (24) 6446-6449
DOI: 10.1021/ja01533a028
First major study on the mechanism of the Reimer-Tiemann, and our view of the mechanism is essentially unchanged today. - Ueber die Einwirkung von Chloroform auf Phenole und besonders aromatische Oxysäuren in alkalischer Lösung
K. Reimer and Ferd. Tiemann
Berichte der deutschen chemischen Gesellschaft 1876 9 (2) 1268-1278
DOI: 10.1002/cber.18760090270
Original report on the Reimer-Tiemann.