Benzoin Condensation
Description: The Benzoin Condensation (or Benzoin reaction) is a combination of two aldehydes to form a new C-C bond, often catalyzed by cyanide ion. The product formed in the reaction of benzaldehyde has the trivial name “benzoin”, hence the name.
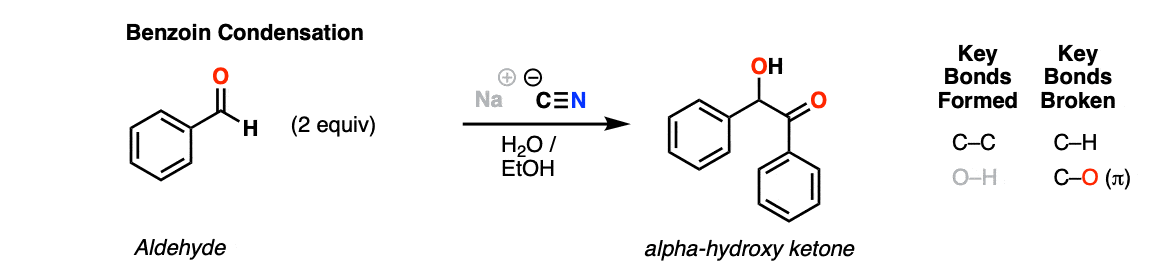
Notes: Cyanide ion is a catalyst. Typically sodium or potassium cyanide is used; for our purposes, there is no essential difference.
The solvent is often an aqueous alcohol such as ethanol; the cyanide salt is dissolved in water and then added to a solution of the aldehyde in ethanol and then heated.
Like the aldol reaction, the reaction can occur with two identical aldehydes or two different aldehydes (a “crossed” benzoin condensation).
Note that one aldehyde is oxidized and one is reduced.
See Also: Aldol addition, Cannizarro reaction.
Examples:
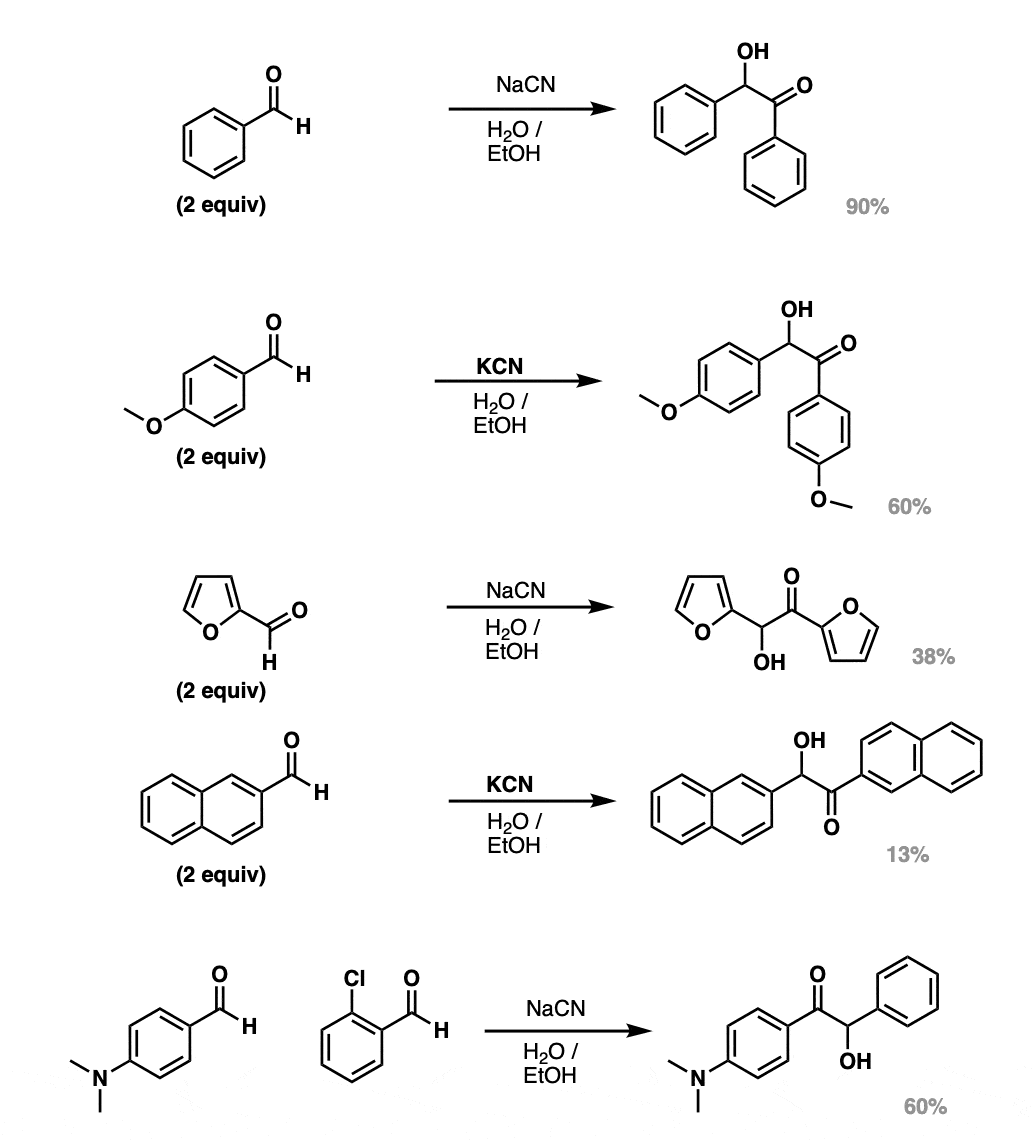
Notes: The first four examples are all self- Benzoin condensations. With cyanide ion as catalyst, aromatic aldehydes are typically used.
The bottom example shows a crossed Benzoin condensation. In a case like this one, the more electron-rich aromatic aldehyde is oxidized (to a ketone) and the more electron-poor aromatic aldehyde is reduced.
These examples are all taken from the Organic Reactions review (below).
Mechanism:
The first step of this reaction is addition of cyanide ion to the aldehyde (Step 1, arrows A and B) which is protonated by solvent to give a cyanohydrin (Step 2, arrows C and D).
The cyanohydrin can be deprotonated with moderately strong base (Step 3, arrows E and F) to give its conjugate base, which can then act as a nucleophile on a second equivalent of aldehyde (Step 4, arrows G and H). The resulting alkoxide is then protonated with solvent to give the product (Step 5, arrows I and J) and then deprotonated to give the conjugate base of the cyanohydrin (Step 6, arrows K and L). These two steps together constitute “proton transfer”.
The elimination of the cyanide ion (Step 7, arrows M and N) re-forms the C-O pi bond and regenerates the cyanide catalyst, which can then go on to do another reaction.
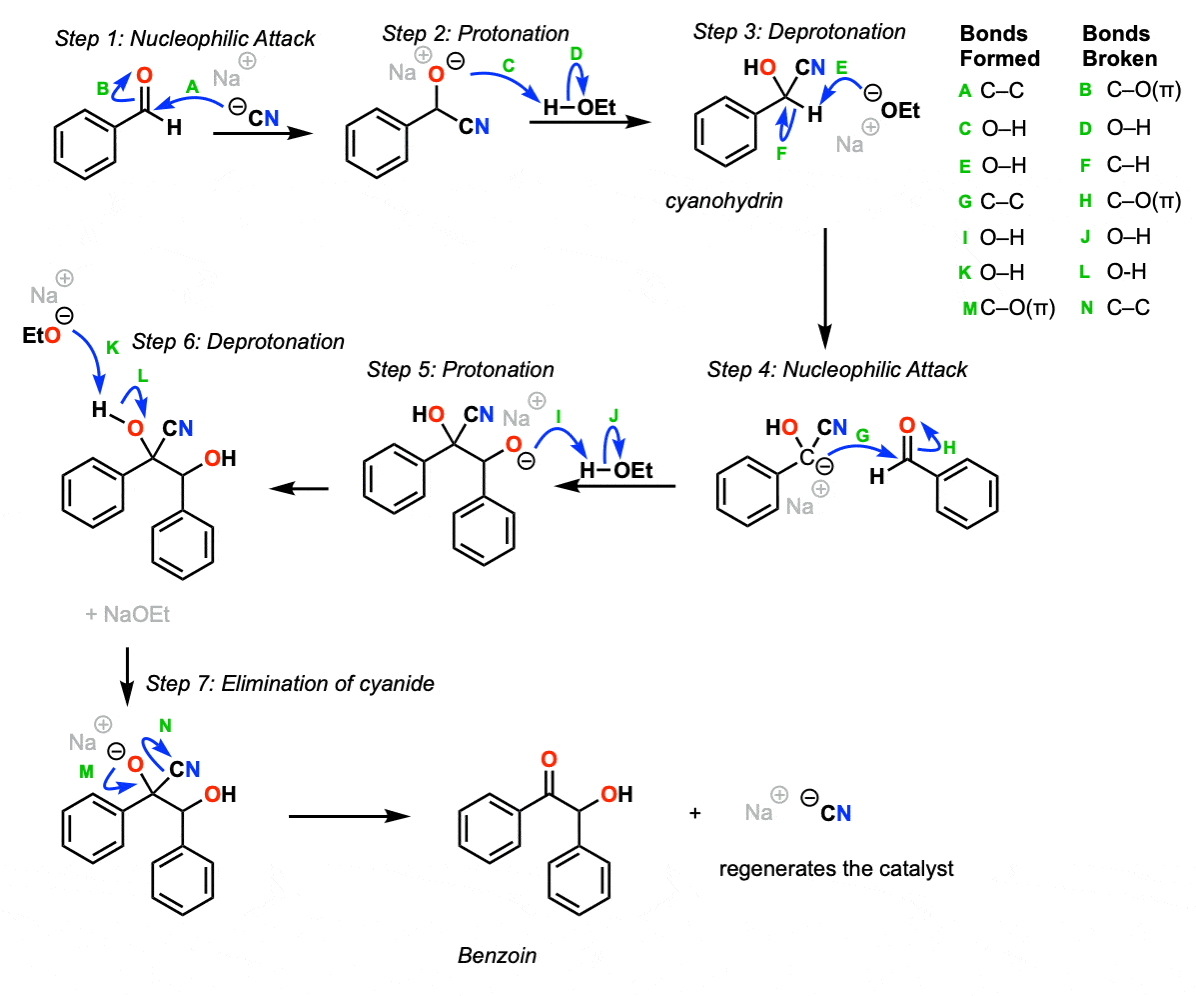
Notes: Although ethanol is shown as the acid in steps 2 and 5, it could very easily be water or another acidic molecule in solution. Similarly sodium cyanide is shown but for our purposes potassium cyanide would work just as well.
(Advanced) References and Further Reading