Henry Reaction
Description: The Henry Reaction is the reaction between the enolate of a nitroalkane (such as nitromethane or nitroethane) and an aldehyde or ketone. Similar to the Aldol reaction, the first product is an addition reaction, which can undergo elimination if heated.
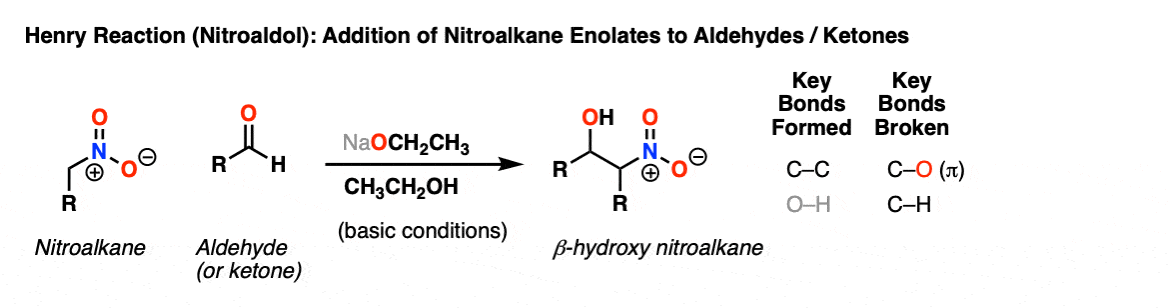
Notes: The product here is a beta-hydroxy nitroalkane, similar to the product of an Aldol addition reaction except with NO2 in place of an aldehyde or ketone.
Mild bases are sufficiently strong to deprotonate the nitroalkane, which has a pKa of roughly 8.5 (nitroethane) to 10 (nitromethane), similar to the pKa of phenyl alcohol (phenol).
Although not shown here, heating will result in elimination to give an alkene product.
See Also: Aldol condensation, aldol addition
Examples:
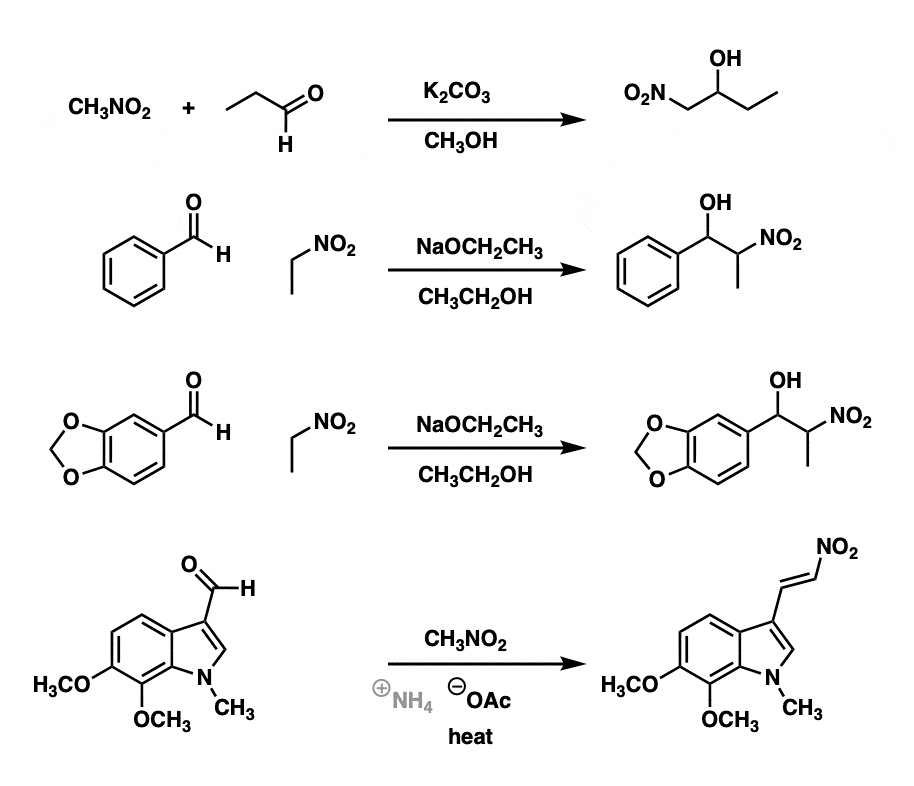
Notes:
Note that bases as poor as carbonate and even acetate are sufficient to perform the nitroaldol reaction (examples 1 and 4).
Example 2 shows the reaction of piperonal (3,4-methylenedioxybenzaldehyde) with nitroethane to form an addition product. This is used in the Shulgin synthesis of MDMA (Ecstasy). [Ref]
Example 4 is an example of a Henry condensation reaction; note that heating resulted in elimination to form the alkene.
Mechanism:
Deprotonation of the carbon alpha to the nitro group with strong base (Step 1, arrows A and B) gives a resonance-stabilized enolate.
Attack of the enolate carbon upon an aldehyde (Step 2, arrows C and D) form a new C-C bond and break the C-O (pi) bond. Protonation of the oxygen (Step 3, arrows E and F) results in formation of the beta-nitro alcohol product.
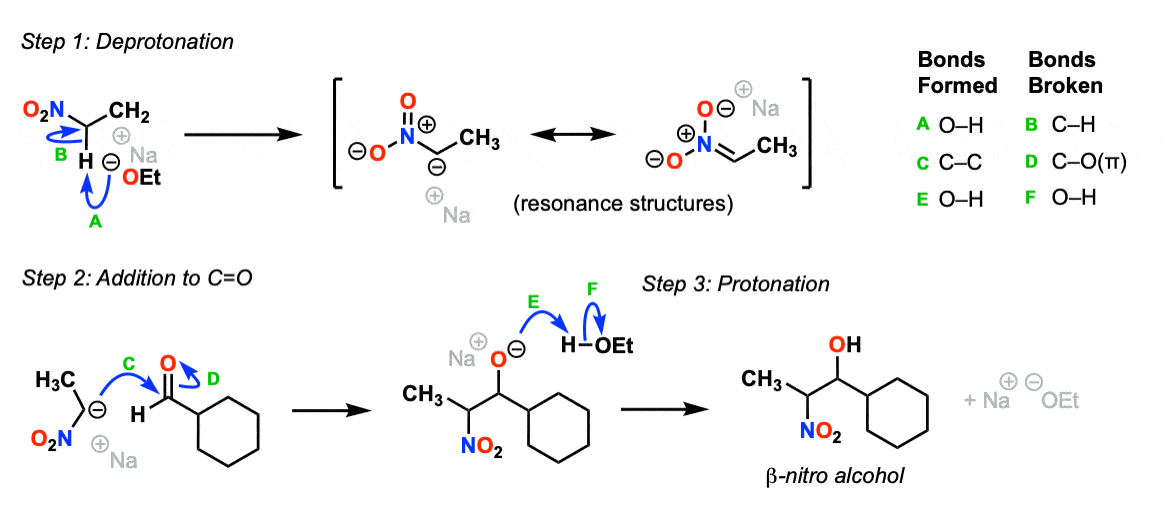
Further heating with strong base would deprotonate the carbon adjacent to the nitro group and result in elimination of HO(-) to give the condensation product.
Notes: For simplicity the nucleophile in step 2 is shown as the carbanion resonance structure, though of course with different arrows one could show it from the “nitronate” resonance structure.
(Advanced) References and Further Reading
For more modern methods on the Henry reaction, see this article on organic-chemistry.org .
-
EJOC 2006 1722 1725
JACS 1999 121 6771
https://gallica.bnf.fr/ark:/12148/bpt6k3076c/f1265.item.r=.zoom
https://chemistry-europe.onlinelibrary.wiley.com/doi/10.1002/ejoc.200700021
https://pubs.acs.org/doi/10.1021/jo9818733
- Isosafrole . https://www.erowid.org/archive/rhodium/chemistry/mdp2np.html