Reduction of carboxylic acids to alcohols with borane (BH3)
Description: Carboxylic acids can be reduced to primary alcohols with borane, BH3 (or the equivalent reagents BH3•THF and BH3 •DMS). The reaction is very mild (unlike LiAlH4!) and can often be done selectively in the presence of other groups (ketones and alkyl halides) that would normally be reduced by LiAlH4.
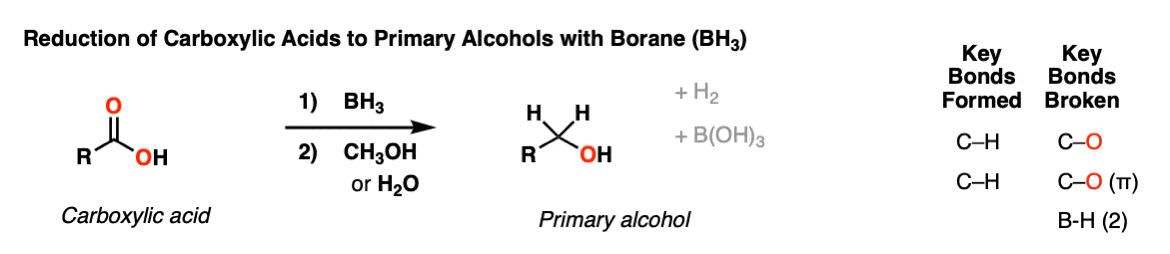
Notes: Although BH3 is shown, it is very common to use the reagents BH3•THF (borane – tetrahydrofuran complex) or BH3 • DMS (borane – dimethylsulfide complex) since these are often slightly more convenient to use.
One equivalent of hydrogen gas is formed. After the reaction a mild workup with methanol or water is generally performed.
See Also: Reduction of carboxylic acids with LiAlH4
Examples:

Notes:
Example 1 shows a typical reaction of a long chain carboxylic acid. Yields are good (>90%). The reaction is slightly slower with benzoic acid (Example 2) but also works well.
Note that BH3 does not reduce nitro groups (Example 3) and can even selectively reduce carboxylic acids in the presence of ketones (Example 4) as well as alkyl halides (Example 5). All of these functional groups would be reduced by LiAlH4.
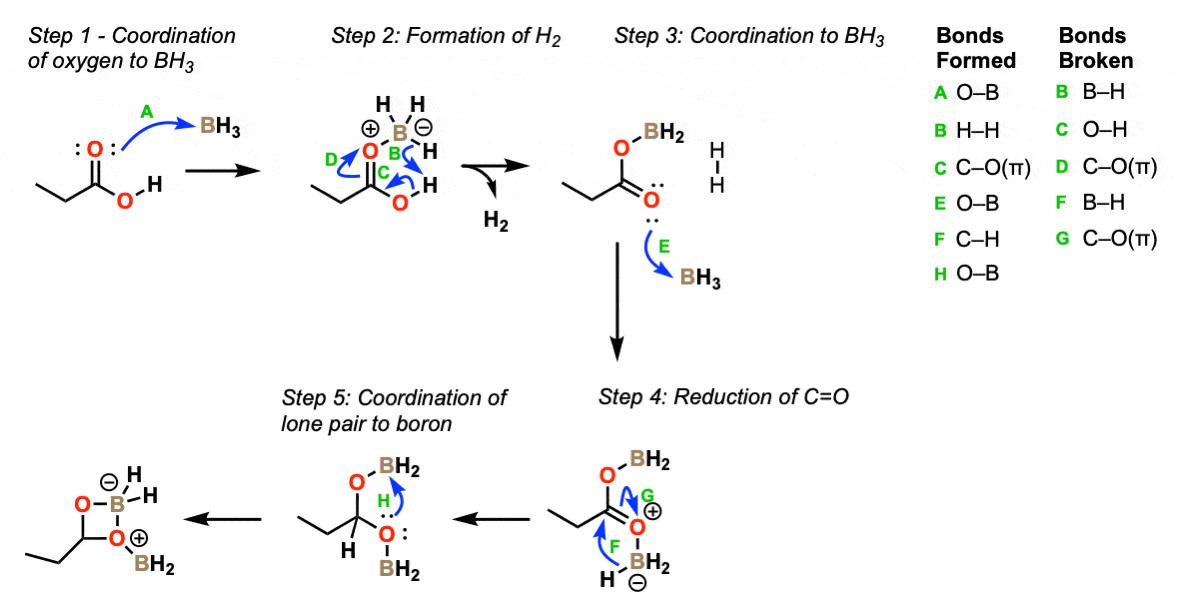
Mechanism:
The first step in this process is coordination of the Lewis basic carbonyl to the Lewis acidic borane (Step 1, arrow A). This enables what is essentially an acid-base reaction (reaction of hydride, H(-) with the acidic proton, H(+) ) to give hydrogen gas (Step 2, arrows B, C and D).
Next, a second equivalent of borane coordinates to the carbonyl oxygen (Step 3, arrow E) which is followed by addition of B-H to the carbonyl carbon (reduction) (Step 4, arrows F and G).
At this step the oxygen lone pair can coordinate with boron to give a 4-membered acetal intermediate (Step 5, arrow H).
Note that this is essentially a protected aldehyde at this point. The next step will be reduction of this to the primary alcohol!
Part 1 – Reduction of carboxylic acid to an acetal
Next, let’s do the full reduction to the primary alcohol.
The cyclic acetal we formed in the previous step can open (Step 6, arrows I and J) to give an aldehyde, which can then be reduced by with the nearby B-H to get to the primary alcohol stage (Step 7, arrows K and L).
The final step is protonation of the alcohol during workup (with either water, methanol, or mild acid) to give the primary alcohol (Step 8, arrows M and N).
Part 2 – Reduction of acetal to primary alcohol
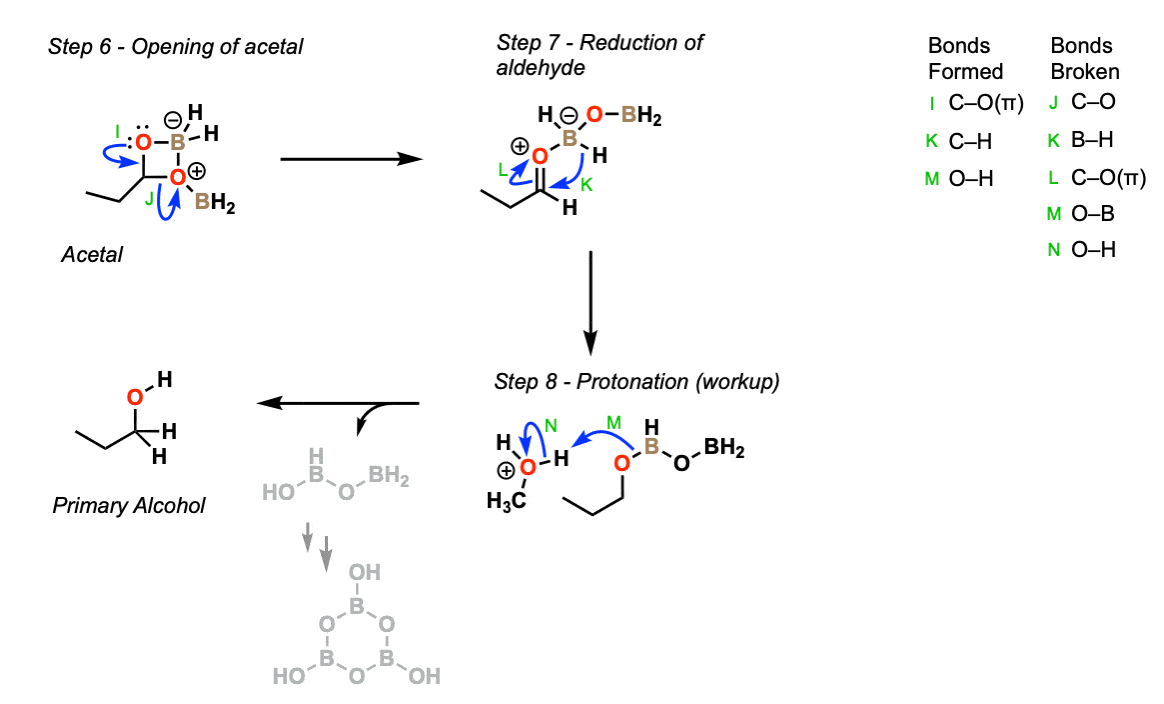
Notes: The specific identity of the borane attached to alcohol is not well defined here, so don’t take that part too literally. Methanol, water, and mild acid can all be used for the final protonation step.
(Advanced) References and Further Reading
For modern applications definitely see the page on Organic-Chemistry.org