Reformatsky Reaction
Description: The Reformatsky reaction involves formation of an enolate through reduction of an alpha-halo ester (usually) with zinc, followed by addition to an aldehyde or ketone followed by mild acidic workup. The final product is an alpha-hydroxy ester, similar to the aldol addition reaction.
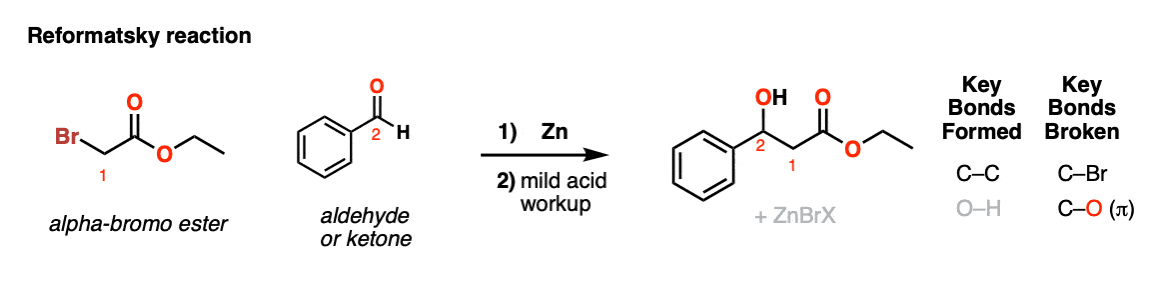
Notes: Typically esters are used (ketones tend to react with themselves unless highly hindered). The electrophile is typically an aldehyde or ketone. Zinc is the most common metal although others can be used. Typically zinc is activated with acid or treated with I2 to remove the oxidized surface and expose the unreacted metal.
After the addition reaction the reaction is quenched with mild acid to give the beta-hydroxy ester product.
See Also: Aldol addition, Grignard reaction , Claisen condensation
Examples:
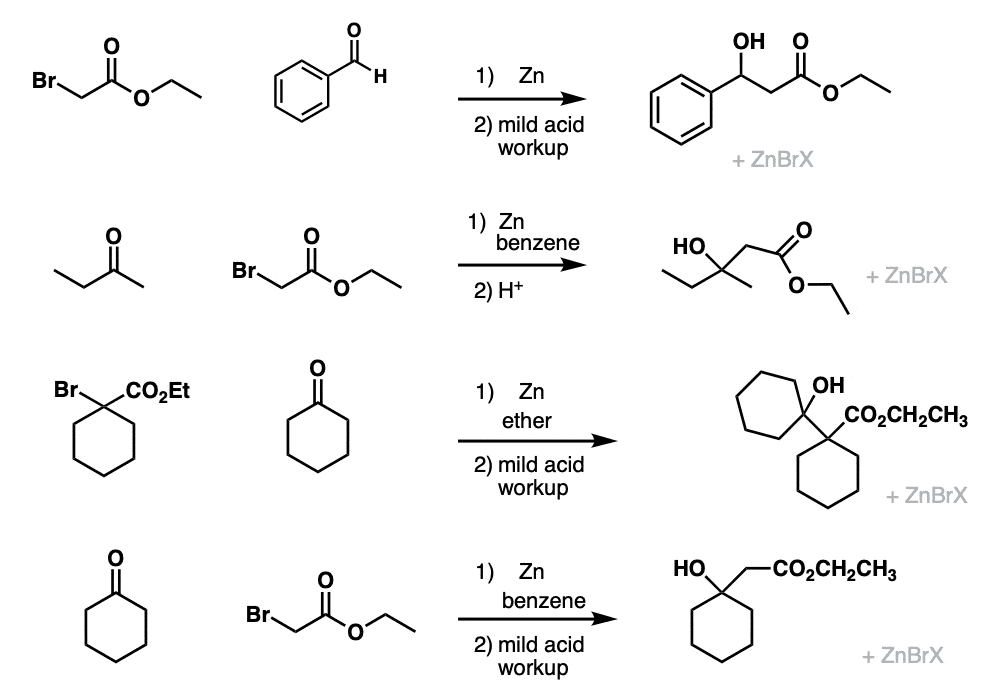
Notes:
Example 1 is a typical Reformatsky reaction between an alpha-bromo ester and an aldehyde.
Examples 2, 3 and 4 show the Reformatsky reaction with various ketones.
Mechanism:
The first step of this reaction is technically called an “oxidative addition” reaction, whereby the C-Br bond is broken and the zinc metal is “inserted” into the C-Br bond. This can occur through numerous pathways (including through radical mechanisms) and the arrows shown here are just one possibility. (Step 1, arrows A and B).
The resulting product is an enolate with ZnBr(+) as a counterion. Like any enolate we can draw resonance structures with the negative charge either on carbon or on oxygen.
Since this reaction is done in the presence of the electrophile (aldehyde or ketone) the enolate, once formed, performs an addition reaction on to the aldehyde (Step 2, arrows C and D) to give the addition product. Addition of mild acid (Step 3, arrows E and F) protonate the alkoxide ion and result in the beta-hydroxy ester product.
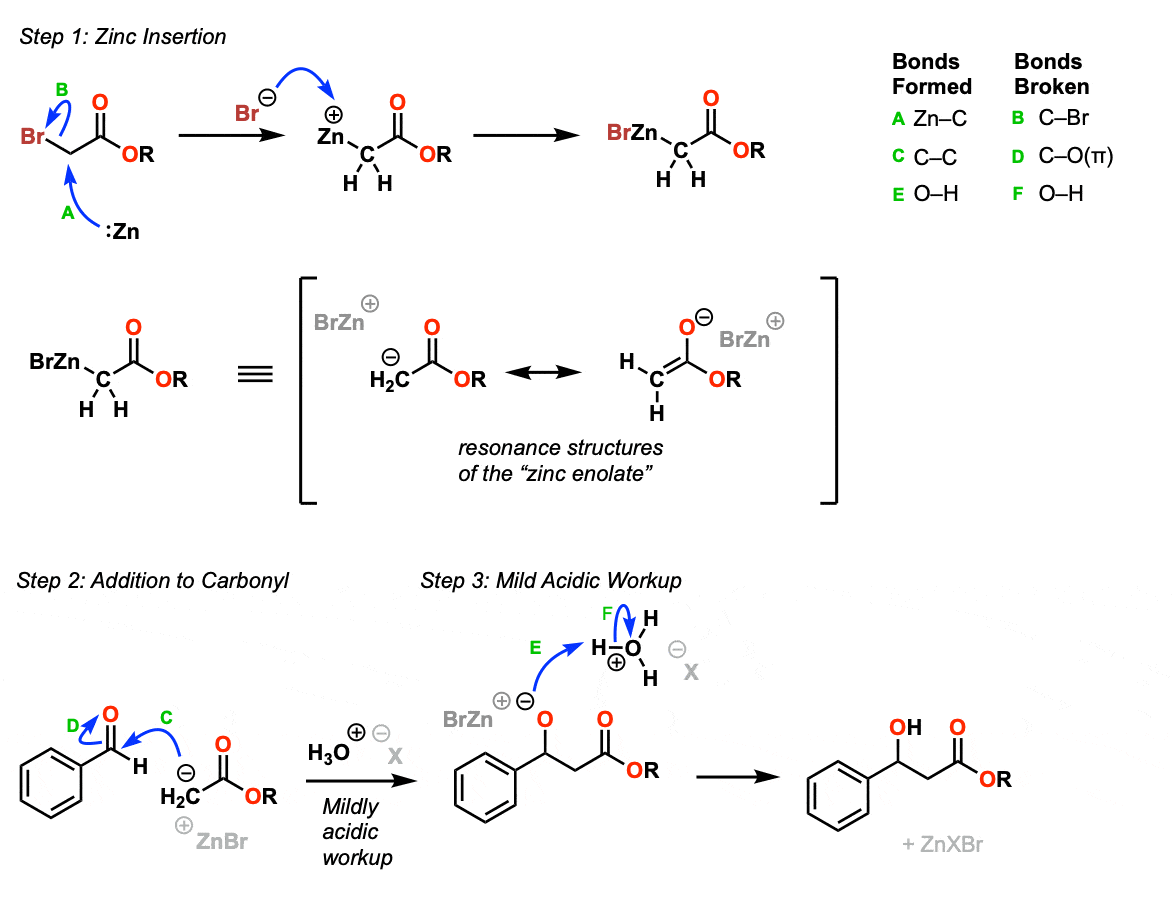
Notes: With extended heating the addition product can be dehydrated to form a C-C pi bond (condensation).
(Advanced) References and Further Reading