Knoevenagel Condensation Reaction
Description: The Knoevenagel Condensation is a reaction between dicarbonyl compounds with an acidic CH2 carbon (e.g. malonates or acetoacetic esters) and aldehydes or ketones in the presence of an amine base to give an alpha-beta unsaturated condensation product.
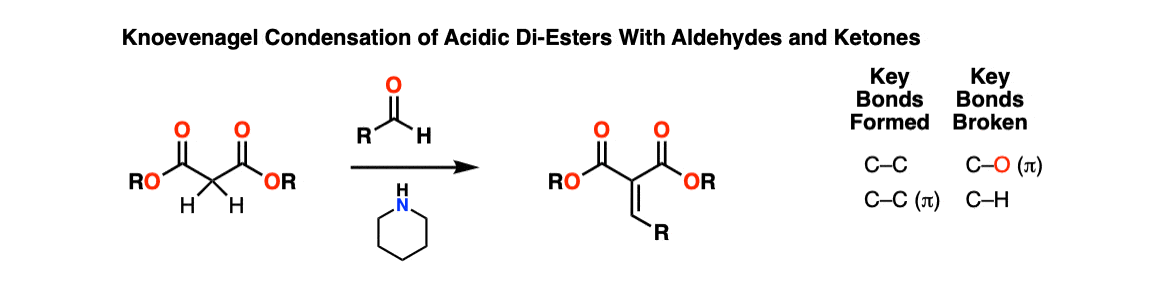
Notes: Typically the base is piperidine although other amine bases can be used. Aldehydes or ketones can undergo this reaction. Strictly speaking, the reaction is not a Knoevenagel if a strong inorganic base like NaOH or KOH is used.
The alpha-carbon of dicarbonyl compounds are typically quite acidic (pKa 8-12) so a strong base is not required.
See Also: Aldol condensation, malonic ester condensation, acetoacetic ester synthesis, Mannich reaction.
Examples:
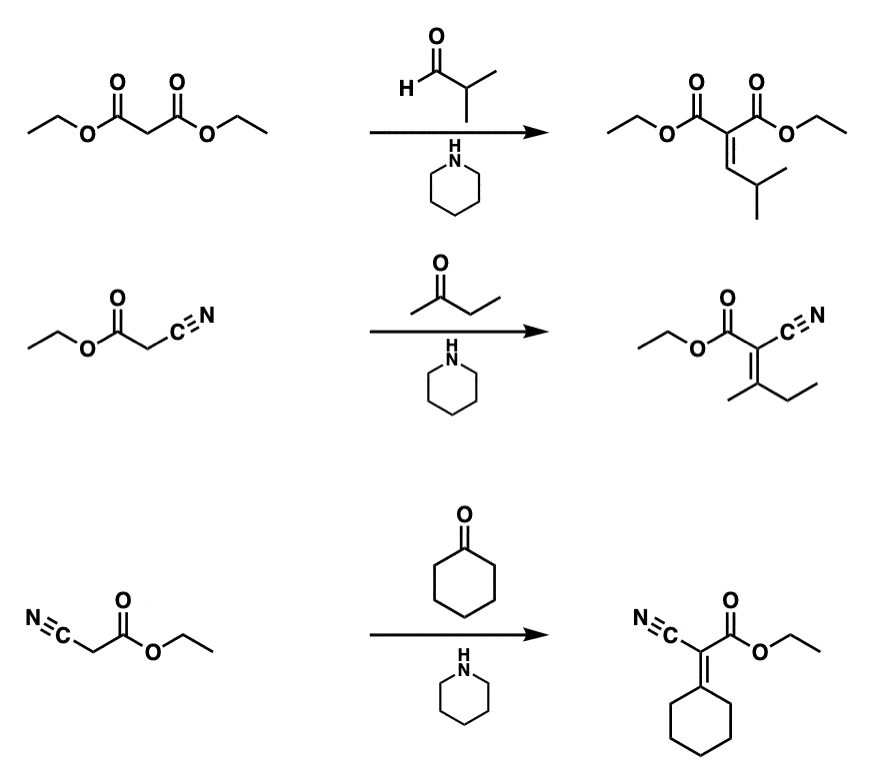
Notes:
Example 1 is a Knoevenagel reaction between diethyl malonate and an aldehyde to give the condensation product.
In the second example, a cyano group is present instead of an ester but the CH2 is still acidic. No E/Z selectivity is implied.
Example 3 shows another Knoevenagel condensation with a ketone.
Mechanism:
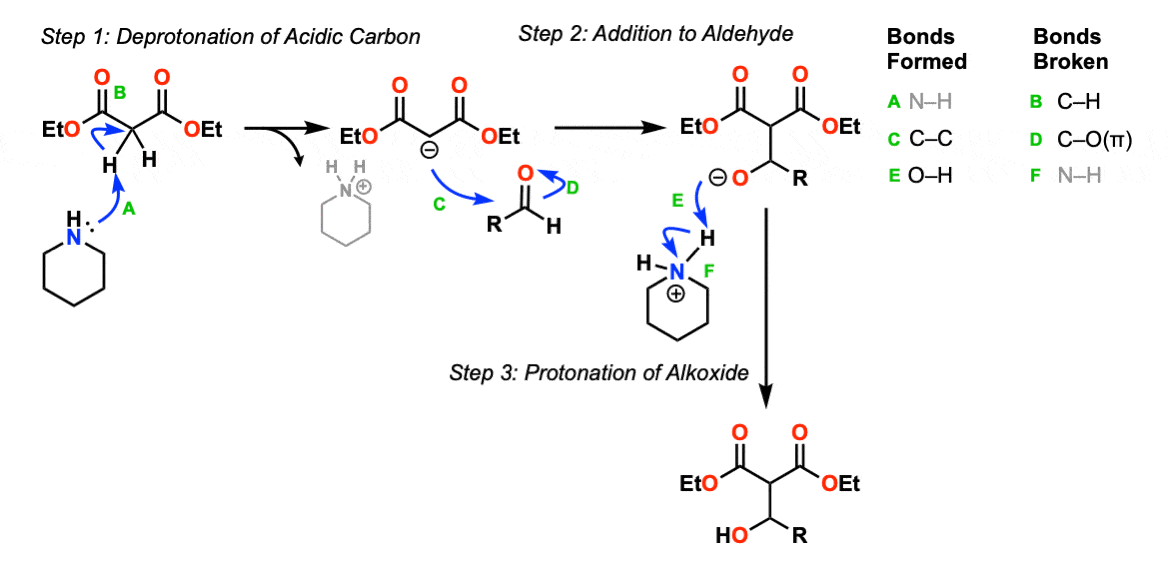
The first step of this reaction is deprotonation of the acidic CH2 group with the moderately strong base piperidine (pKaH about 10) to give the enolate (Step 1, arrows A and B). The resulting enolate can then act as a nucleophile to attack the carbonyl carbon of an aldehyde or ketone (Step 2, arrows C and D) to give an addition product.
In the third step the alkoxide can be protonated (Step 3, arrow E) to form the beta-hydroxy addition product. (Several different acids could work for this step, but just the piperidinium ion is shown).
To form the condensation product, the alpha-carbon is again deprotonated with base (Step 4, arrows G and H) to give the enolate, which can then perform an elimination reaction to form a new C-C pi bond with elimination of hydroxide ion (Step 5, arrows I and J). This gives the final condensation product.
Note that this step is goes through an E1cB mechanism (elimination, unimolecular rate-determining step) since it involves formation of the enolate before elimination occurs.
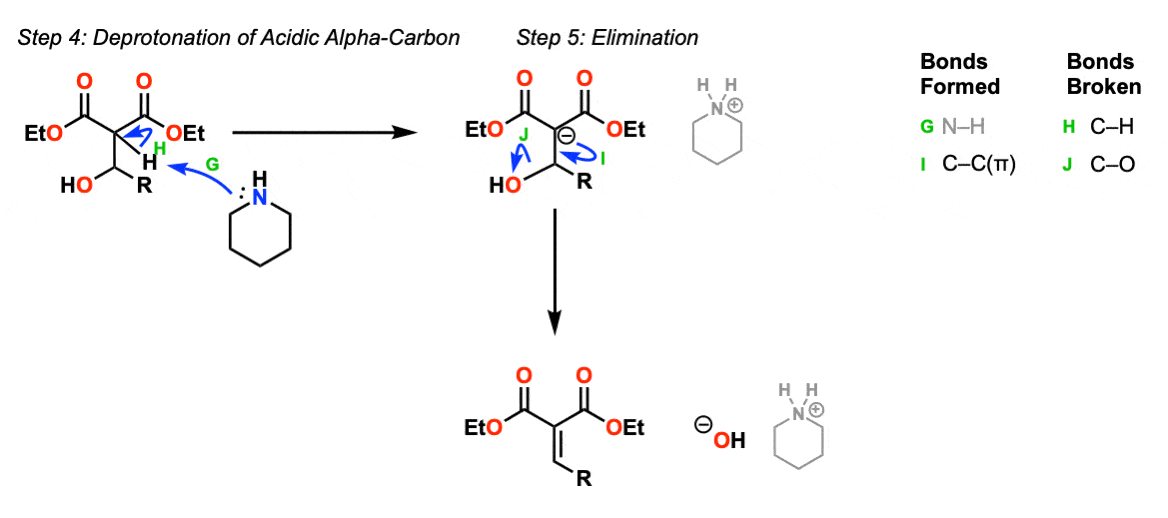
Notes: The final products here are shown as hydroxide ion and piperidiniuum ion, which would then likely react in an acid base reaction to regenerate hydroxide ion and piperidine. Therefore piperidine is catalytic in this process.
Advanced note: The above is just one of the two major proposed mechanistic pathways for the Knoevenagel (the “Hann – Lapworth” mechanism) which is sufficient for the purposes of introductory organic chemistry. If you go on in organic chemistry, you will discover that the Knoevenagel reaction can go through an entirely different mechanism entirely – one where the first step is formation of an imine from the condensation of piperidine and the aldehyde/ketone, followed by protonation (to give an iminium ion) and subsequent attack by the enol (or enolate) of the dicarbonyl compound to give the product of a Mannich reaction. The Mannich product is then protonated at the beta position and undergoes beta-elimination to give the final condensation product.
Thus the purpose of the piperidine is not just to act as a base but also to form a reactive iminium ion from the aldehyde/ketone.
(Advanced) References and Further Reading
- Condensationen zwischen Malonester und Aldehyden unter dem Einfluss von Ammoniak und organischen Aminen
E. Knoevenagel
Ber. 31(3) 1898 2585
DOI: 10.1002/cber.18980310307 - The Knoevenagel Condensation
G. Jones
Org. React. 15: 204 (1967)
DOI: 10.1002/0471264180.or015.02
Any time you see an Organic Reactions reference for a specific reaction, expect a definitive treatment. This is no exception. - Kinetic Evidence for a Schiff Base Intermediate in the Knoevenagel Condensation
Thomas I. Crowell and David W. Peck
Journal of the American Chemical Society 1953 75 (5), 1075-1077
DOI: 10.1021/ja01101a018 - Condensation Reactions. II. Alkylidene Cyanoacetic and Malonic Esters
Arthur C. Cope, Corris M. Hofmann, Cornelia Wyckoff, and Esther Hardenbergh
Journal of the American Chemical Society 1941 63 (12), 3452-3456
DOI: 10.1021/ja01857a057