Dienes and MO Theory
Exo vs Endo Products In The Diels Alder: How To Tell Them Apart
Last updated: May 28th, 2026 |
Today we’ll cover how to tell endo– and exo- products apart in the Diels-Alder reaction. Shortcut below. For the long-winded walkthrough with lots of examples, read more below.
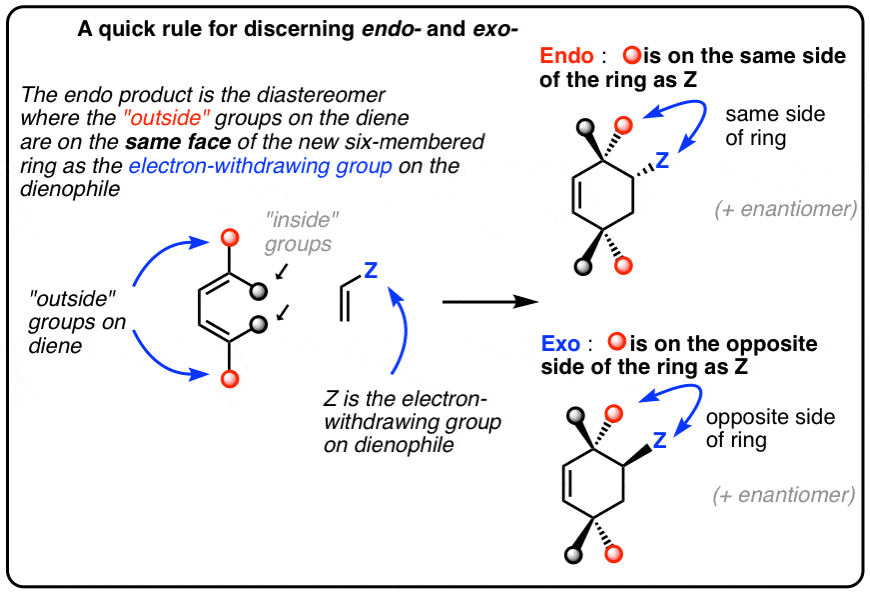
Table of Contents
- Exo and Endo Products In The Diels-Alder
- Exo And Endo Diels-Alder Products Are Diastereomers Of Each Other
- 3-D Models Of The Endo And Exo Products In The Diels-Alder Between Cyclopentadiene And Maleic Anhydride
- Distinguishing ”Endo” vs “Exo”: Pay Attention To The Relationship Between The “Outside” Groups On The Diene And The EWG On The Dienophile
- Another Worked Example: Cyclopentadiene And Quinone
- How Do You Know If A Diels-Alder Will Give You Endo And Exo Products?
- Four More Examples (Including A Trick Question)
- Transition States In A Diels-Alder That Does NOT Lead To Endo And Exo Products
- Transition States In A Diels-Alder That Leads To Exo and Endo Products
- So Why Is The Endo Product Favored?
- Notes
- Quiz Yourself!
1. Exo and Endo Products in the Diels-Alder
In this series of articles on the Diels-Alder reaction, we’ve seen that:
- the Diels-Alder reaction always involves the breakage of 3 pi bonds and the formation of 3 new bonds (2 sigma, one pi), resulting in the formation of a new six-membered ring. [Intro]
- The stereochemistry of the dienophile is preserved. A cis arrangement of groups in a double bond will result in a cis arrangement in the new six-membered ring. And likewise, trans → trans
- For dienes, the two “outside” groups on the diene each end up on the same face of the new six-membered ring. Likewise, the two “inside” groups each also end up on the same face of the new ring.
(These last two parts are from the last post, where we went through the stereochemistry of the Diels-Alder reaction.)
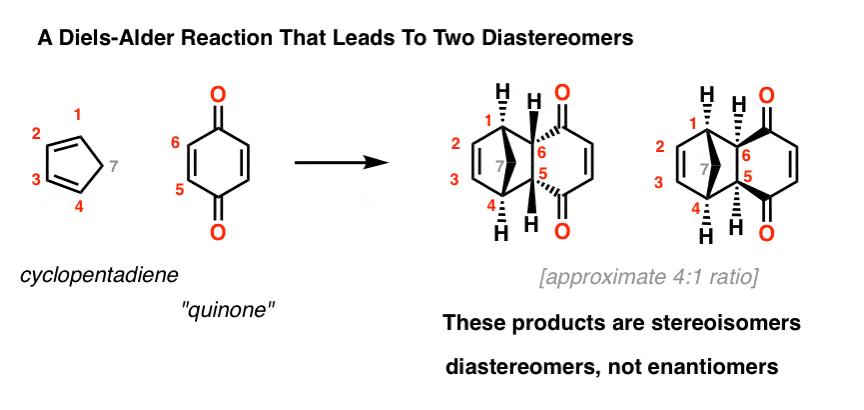
Now let’s add the next layer of detail. For certain diene/dienophile combinations, we can end up with two products that fulfill all of those criteria. For instance:
2. Exo And Endo Diels-Alder Products Are Diastereomers Of Each Other
These two products are stereoisomers. But since they are not non-superimposable mirror images (i.e. not enantiomers), they are diastereomers. hover here to view a pop-up image [In fact, each of these molecules are achiral, due to the presence of a mirror plane – if you can’t see it, click here ]
These two molecules can be separated due to the fact that they have different physical properties.
An as it turns out, the major product is the one above left. The minor is the one above right.
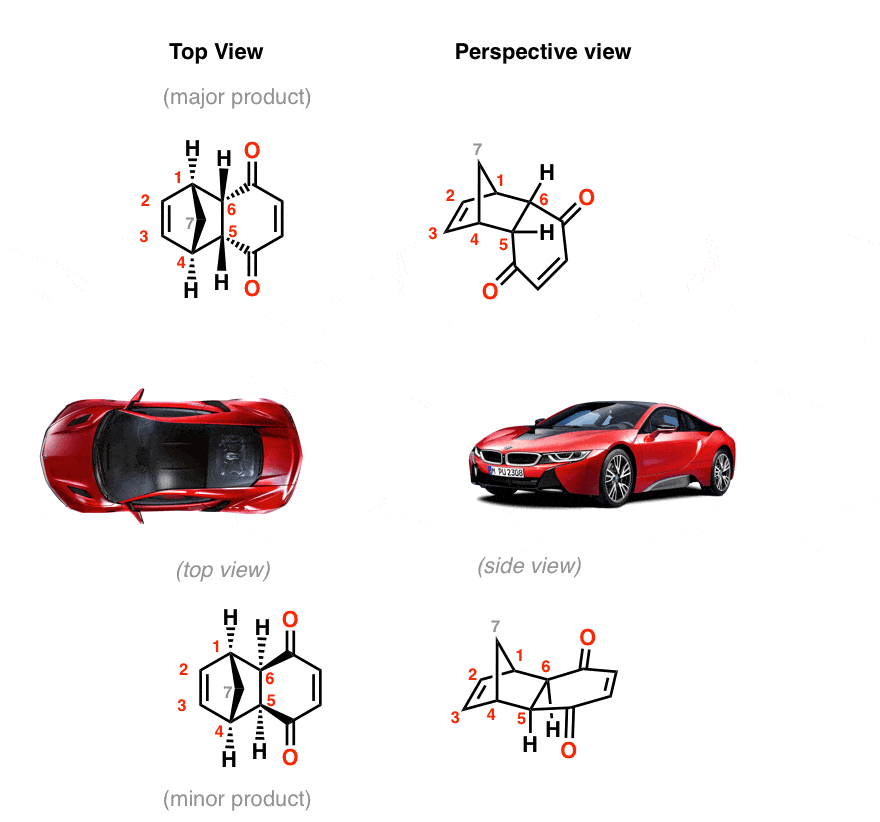
Sometimes it can help to look at these things in perspective:
3. 3-D Models Of The Endo And Exo Products In The Diels-Alder Between Cyclopentadiene And Maleic Anhydride
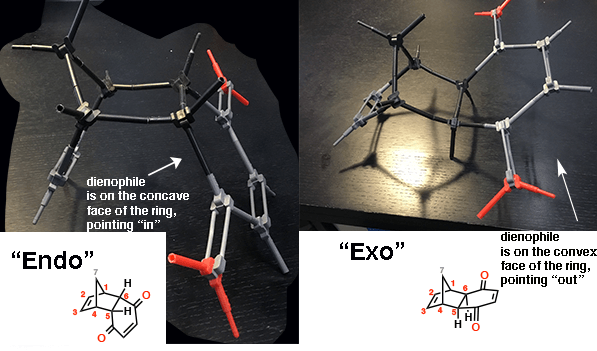
Here are the two products again (major and minor) built as models.
- on one product (bottom left), the dienophile is on the underside (“concave”) face of the ring, pointing “in” toward the alkene
- on the other product, the dienophile is on the convex face of the ring, pointing “out”, away from the alkene.
When Diels and Alder originally discovered this phenomenon, they assigned the name “endo” to the major product (where the dienophile is pointing “in”, towards the alkene) , and the term “exo” (outside, such as in “exoskeleton”) to refer to the minor product (where the dienophile is pointing “out”, away from the alkene).
In this particular example, identifying “exo” and “endo” seems straightforward based more on an appeal to intuition than anything else. But trust me when I say that there are plenty of examples where it isn’t so easy to assign.
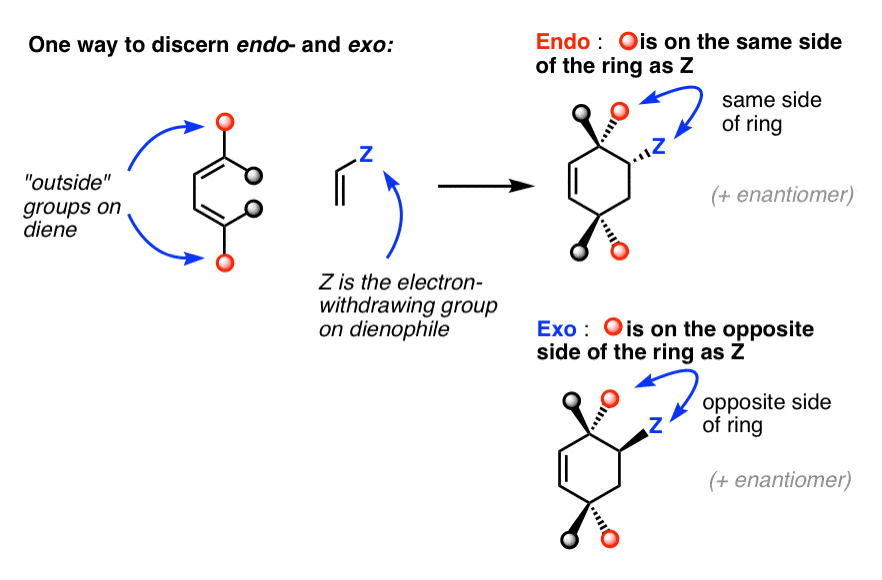
4. How To Recognize The “Endo” And “Exo” Products In A Diels-Alder Reaction: Pay Attention To The Relationship Between The “Outside” Groups On The Diene And The EWG On The Dienophile
In that vein, it’s helpful to have a standard “rule” that we can apply to each situation.
Here’s one way to do it:
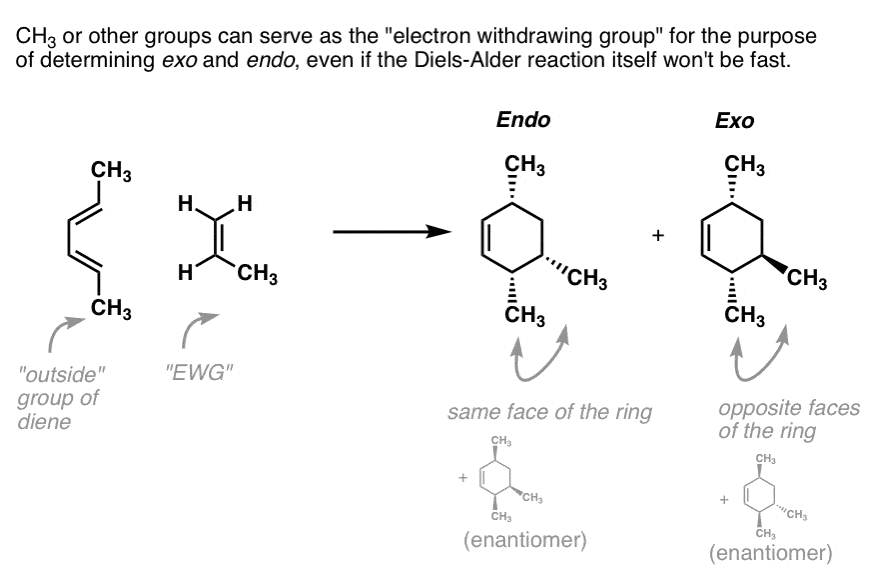
Think of the endo as being the diastereomer where the “outside” group on the diene is on the same side of the new six-membered ring as the “electron-withdrawing group” on the dienophile.
The exo is the diastereomer where the outside group on the diene is on the opposite side of the new six-membered ring as the electron-withdrawing group on the dienophile.
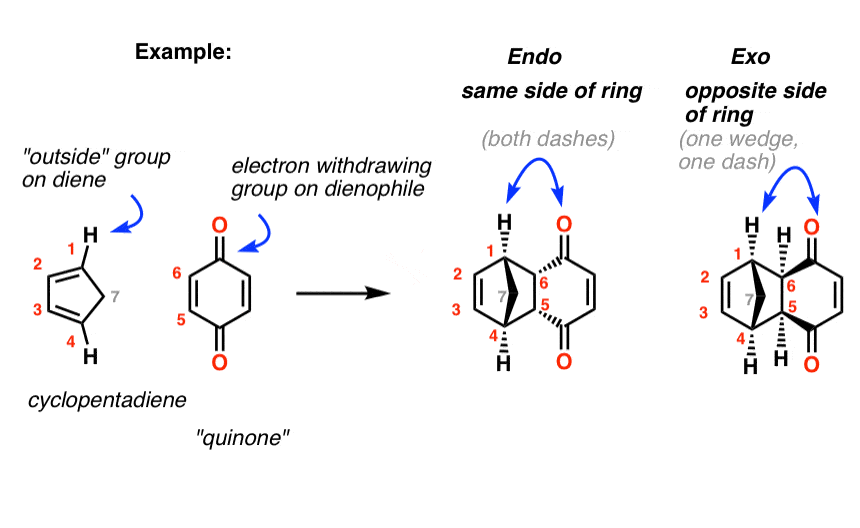
5. Another Worked Example: Cyclopentadiene And Quinone
Let’s try applying those rules to the products of the first reaction we looked at. In the endo drawn below, the “outside” group on the diene (the hydrogens) points down, and the bond to the carbonyls on the dienophile point down as well. Same side of the new ring.
In the exo the outside group (hydrogen) is down and the bond to the electron-withdrawing group points up. Opposite sides of the new ring.

6. How Do You Know If A Diels-Alder Will Give You Endo And Exo Products?
So how do you know if a given Diels-Alder is going to have endo and exo products?
Here’s a short answer. (A longer, more comprehensive answer follows at the bottom of the post).
- If all of the “outside” groups on the diene are the same as the “inside” groups, then there can’t be exo or endo. Butadiene itself, for example; the “inside” and “outside” groups are both hydrogens.
- If the dienophile lacks a substituent that can be distinguished as the “electron withdrawing group”, then there won’t be exo or endo. Ethylene is a clear example, as is tetracyanoethylene. Or acetylene and derivatives like DMAD. [Note 1] There are also a few interesting dienophiles [example 4] where exo– and endo- products are not possible for a more subtle reason.
7. Four More Examples (Including A Trick Question)
Let’s look at a few more examples.
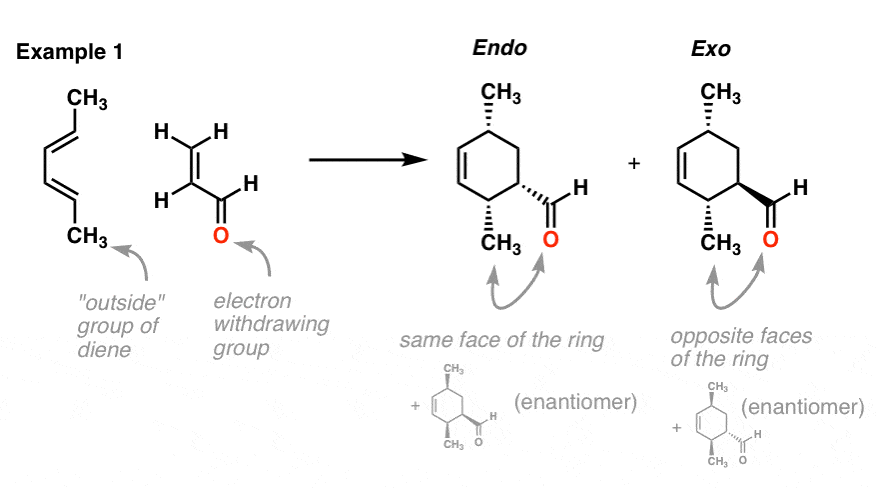
Example 1: 2,4-hexadiene (diene) and acrolein (dienophile)
Note that the endo product has the methyl groups on the same face of the ring as the aldehyde (both dashes, as pictured, or both wedges, as in the enantiomer). The exo product has the methyl groups on the opposite face of the ring as the aldehyde.
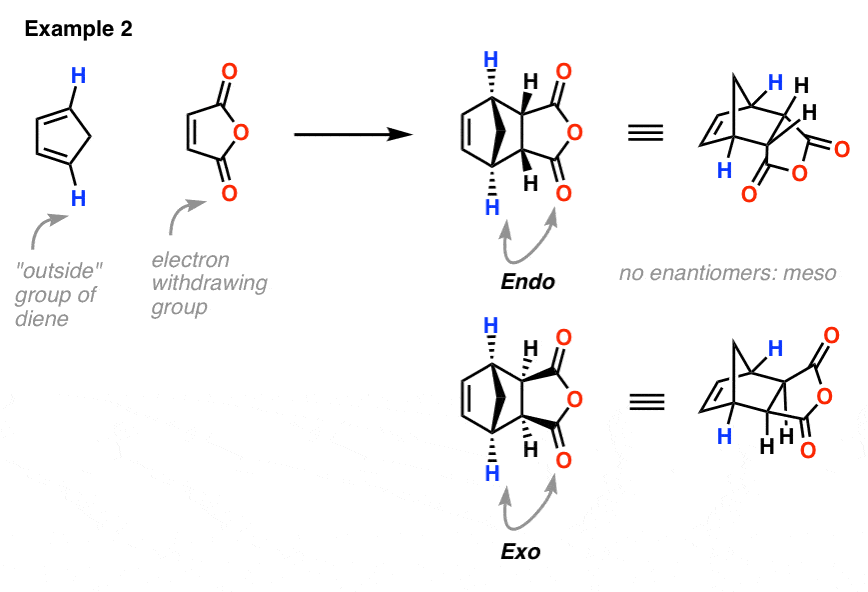
Example 2: Cyclopentadiene and maleic anhydride
This is very similar to the original example, where the endo has the anhydride ring on the concave face, and the exo has the anhydride on the convex face.
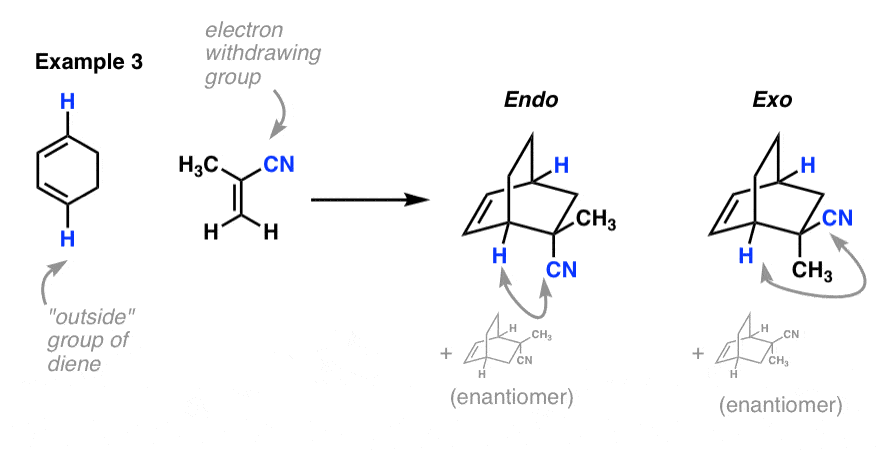
Example 3: Cyclohexadiene and methacrylonitrile
Here, the nitrile is clearly the electron withdrawing group, and so it’s the functional group we judge endo and exo by, rather than the methyl group. I should note that when you look at a model of the product, the hydrogen is not really pointing “down” (it points out, more than anything else) but it’s certainly “down”, relative to the inside group (the CH2CH2 bridge).
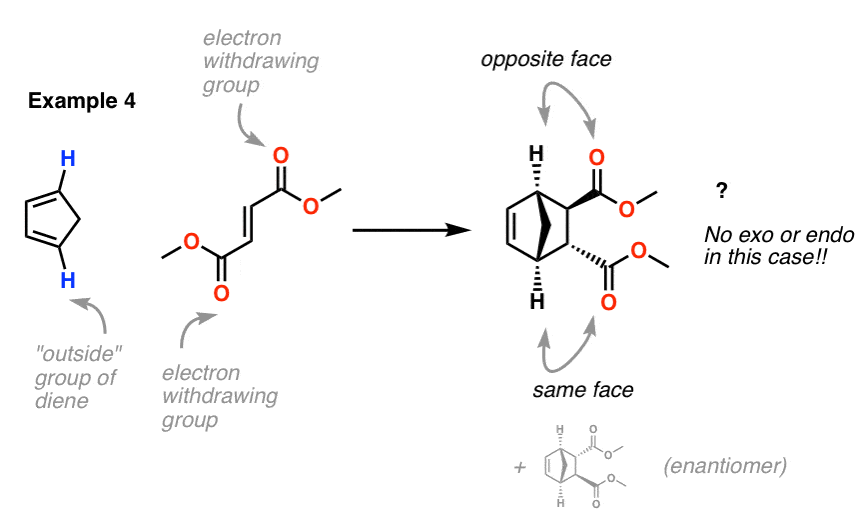
Example 4. Here’s a fun one: Cyclopentadiene and dimethyl fumarate
Our dienophile has two electron withdrawing groups placed on opposite sides of the pi bond. So which electron withdrawing group do we point to when determining exo or endo ?
We can’t distinguish them! Therefore there is no exo or endo in this case. The product will, however, exist as a mixture of enantiomers. [Note 2] . And yes, these two molecules are enantiomers, not diastereomers. Hover here for a pop-up view or click this link.
8. Transition States In A Diels-Alder That Does NOT Lead To Endo And Exo Products
This post is just intended to give some examples of discerning exo and endo, and we can leave it at that.
It’s worth one last look, however, at how exo and endo might arise.
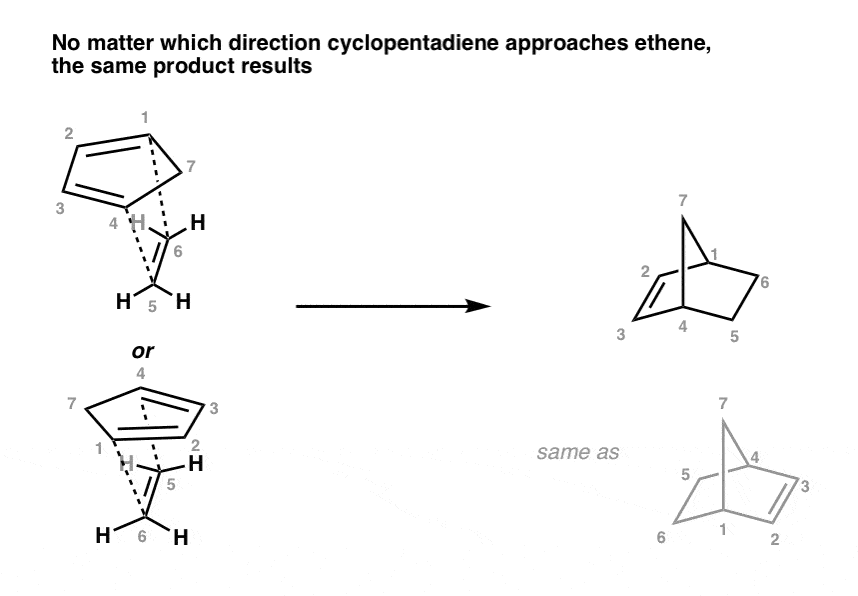
First, let’s imagine how cyclopentadiene might come together with ethene in a Diels-Alder.
Cyclopentadiene can approach “head-first” (where C-7 floats over ethylene first) or “butt-first” (where C2-C3– floats over ethylene first). But because both sides of ethene are exactly the same, each of these approaches lead to the same product.
There is no possibility of exo and endo here.
9. Transition States In A Diels-Alder That Leads To Exo and Endo Products
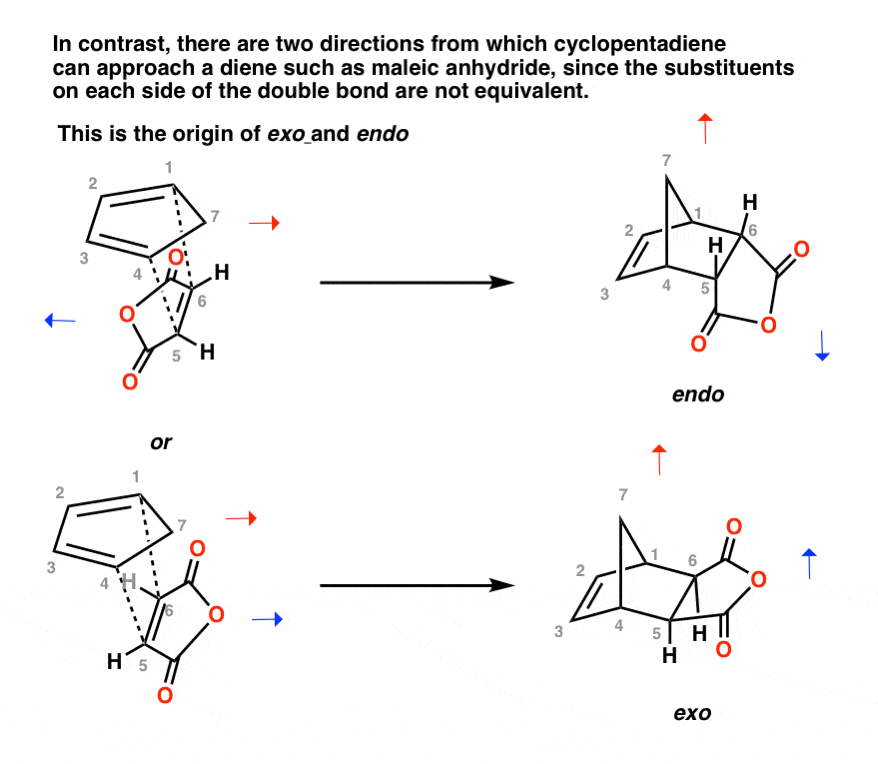
Now, let’s look at how cyclopentadiene can approach a substituted dienophile like maleic anhydride.
Maleic anhydride does have a “head” and a “tail”, unlike ethylene. The two approaches of cyclopentadiene to maleic anhydride are therefore not equivalent, which results in two different transition states (diastereomeric transition states) which leads to two different products. [Note 3]
This is the origin of endo and exo!
10. So Why Is The Endo Product Favored?
But, you might ask, why is it that the endo product is favored over the exo by so much – especially when it seems like the endo product has much more steric hindrance than the exo?
That is a great question. Let’s look at it next time.
[Next post: Why Are Endo Products Favored In The Diels-Alder? ]
Notes
Note 1. In a pinch, CH3 or other groups that are not technically “electron withdrawing groups” can serve as placeholders for our exo-endo shortcut.
This frequently comes up in inverse electron-demand Diels-Alder reactions, where the “dienophile” is electron rich and the diene electron-poor.
Note 2. Dimethyl fumarate has a property called “C2-symmetry” , which is the property that turning it 180° in one direction leads to an indistinguishable molecule. (The letter “S” has that property).
Note 3. Things get even more complicated when either the diene or dienophile is non-symmetrical; this results in 4 possible products; two enantiomeric endo transition states, and two enantiomeric exo transition states.
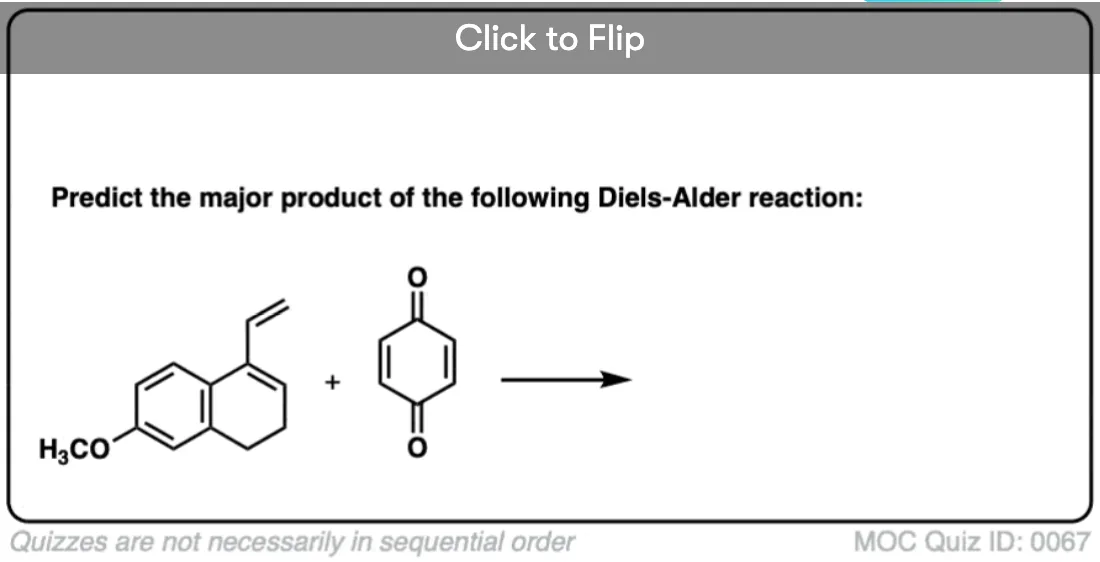
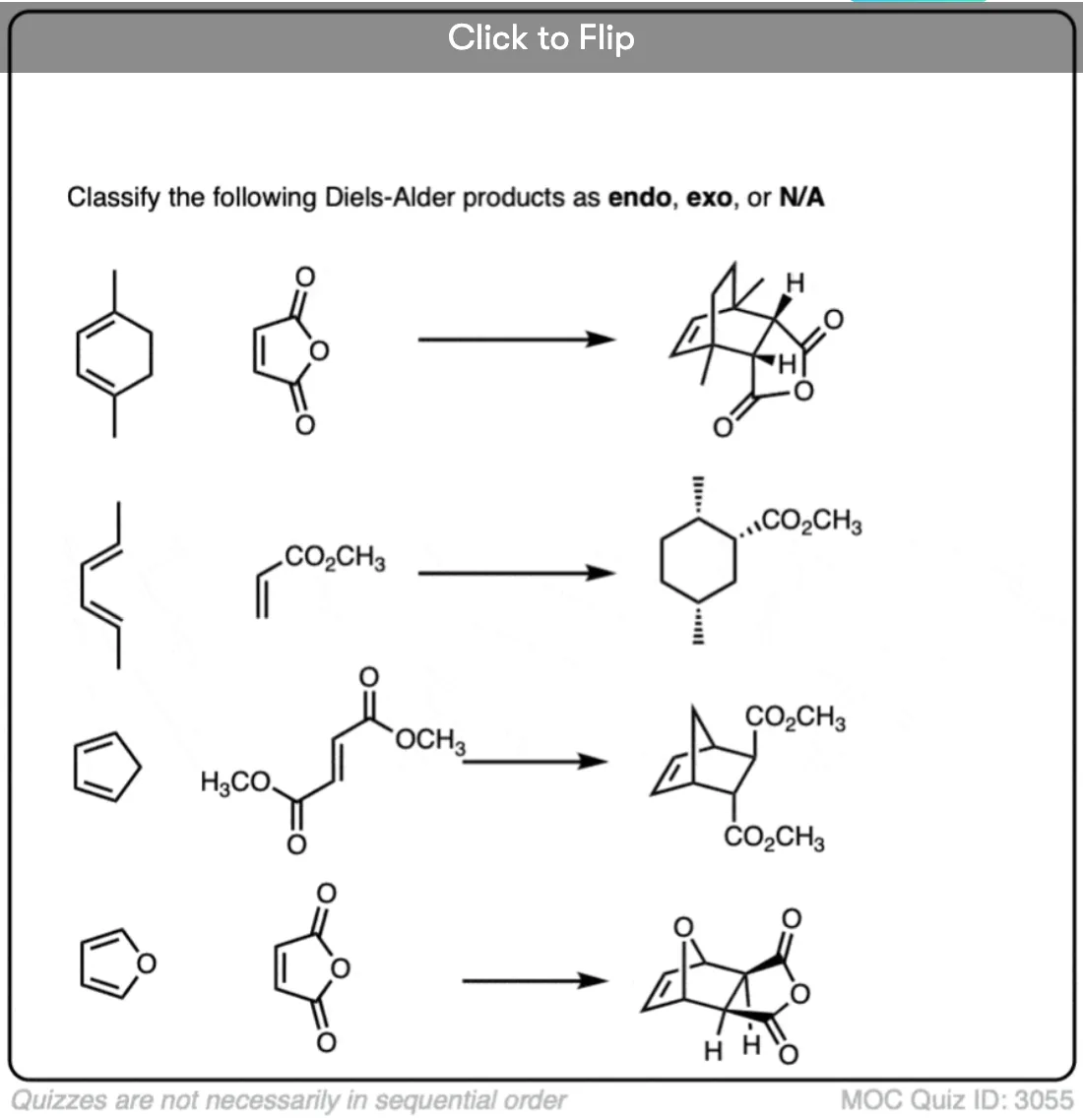
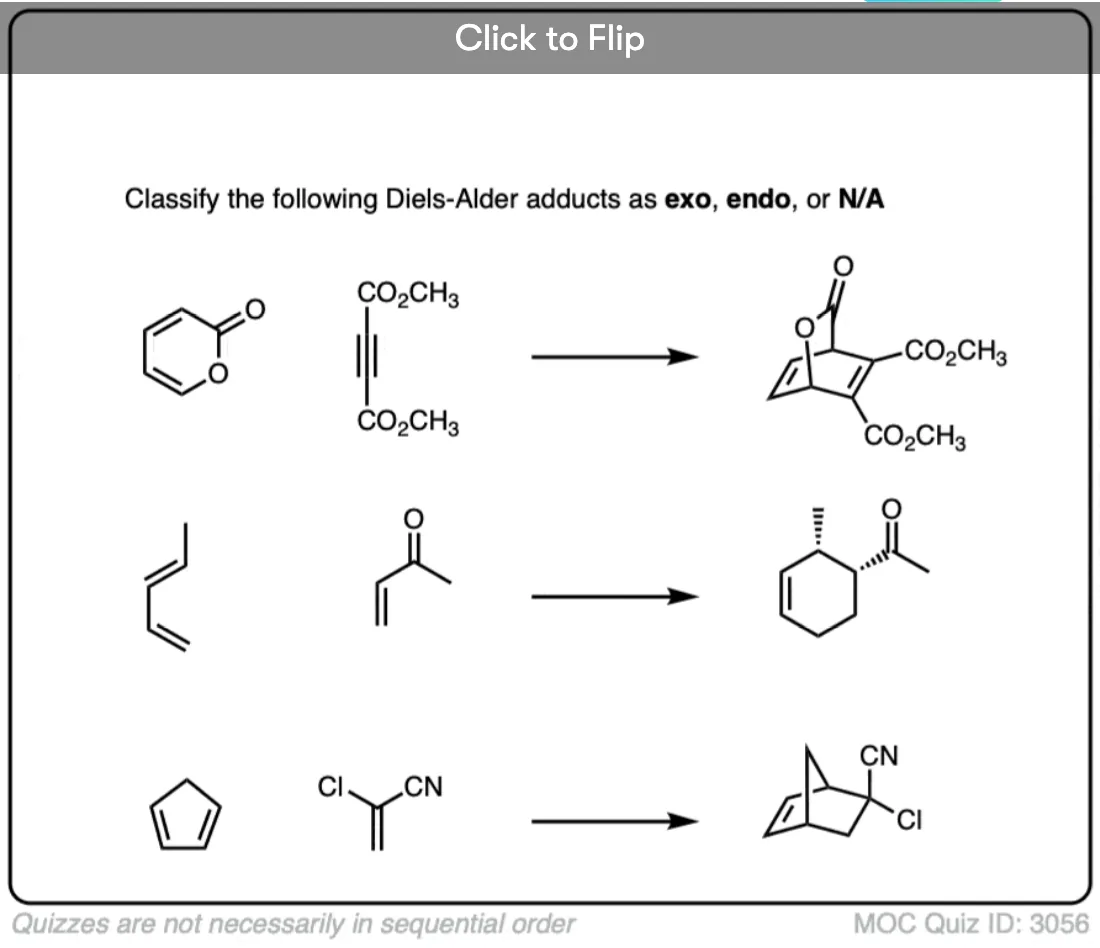
Quiz Yourself!
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.


For the answer on flashcard 6, question 2, can you help me understand why the diene was redrawn the way it was?
It looks like the diene was redrawn incorrectly on the second problem of the last flashcard. It lost a carbon
Ah yes. Fixed quiz 3061. Thank you.
I would like to know, in a case similar to Example 1, where the major product is an endo product, which enantiomer is the major product?
Since there is no chiral influence on the reaction, an equal proportion of enantiomers is formed (racemic mixture).
why there is no enantiomer in example 2
It’s a meso compound. There is an internal plane of symmetry that renders the molecule achiral overall even though it possesses chiral centers.
Really its a nice post & have great topic discussion .
its a concept booster…..
thankyou so much!
actually my query regarding this topic is that:
when cis,cis-3,4-dimethylcylobutene is subject to reaction photochemically dis-rotation ring opening, what will be the product?
either cis or trans ?
Try it for yourself. What do you think?
This is the IUPAC definition of endo/exo/syn/anti in bicyclo alkane systems: https://goldbook.iupac.org/terms/view/E02094
The names under the two structures seem to contradict the definition above. Is this an error?
Mike
There’s been some previous discussion on this thread regarding IUPAC and their use of exo/endo versus how the terms are actually used in practice. My general opinion on this one is that IUPAC can get stuffed because nobody follows their recommendations on this topic.
If there is a specific image that is causing confusion to you, could you specify which one (image filenames are numbered, so that would help).
this is incredibly well done.
i finally understand the endo rule
Thank you Eric. Glad you found it helpful
How do we depict formation of endo and exo products in cycloaddition reactions involving 1,3-dipolarophile such as a nitrone and a dipolariphile such as an activated olefin. Thanks
No worries! I always teach that the enantiomer has the opposite R,S designation so this exception is a very useful thing to keep in mind!!!
you are right. it was my fault for relying on ChemDraw to name these. should have relied on the old school do-it-by-hand.
these are the iupac names for the two “enantiomers” in example 4:
dimethyl (1R,2R,3R,4S)-bicyclo[2.2.1]hept-5-ene-2,3-dicarboxylate
dimethyl (1R,2R,3S,4S)-bicyclo[2.2.1]hept-5-ene-2,3-dicarboxylate
they are not enantiomers otherwise the 2nd molecule would be dimethyl (1S,2S,3S,4R)-bicyclo[2.2.1]hept-5-ene-2,3-dicarboxylate
If we started with dimethyl(1R, 2R, 3R, 4S)-bicyclo[2.2.1]hept-5-ene-2,3-dicarboxylate and did a single swap (i.e. flipped) all four stereocenters, you would get the enantiomer. Agree?
If so, then would you agree that the name of that enantiomer would be (1S, 2S,3S,4R)bicyclo[2.2.1]hept-5-ene-2,3-dicarboxylate?
As it turns out this is not correct (!!) due to a quirk of IUPAC nomenclature (priority of numbering). A picture says more than a lot of words would. Here is a link to an explanation I made. https://www.masterorganicchemistry.com/wp-content/uploads/2020/02/Supp-More-Iupac-exo-endo.gif
Here is a screenshot of the IUPAC rule from the Blue book: https://www.masterorganicchemistry.com/wp-content/uploads/2020/02/Supp-priority-of-R-over-S-1.png
example 4: the greyed out structure you have noted parenthetically as the enantiomer is actually a diasteromer
No, they’re enantiomers. Try rotating the structure in the plane of the page, you’ll see it’s a non superimposable mirror image. Image here: https://www.masterorganicchemistry.com/wp-content/uploads/2020/02/Supp-Case-4-enantiomers.gif
How can we explain endo and exo using secondary orbital overlap
See this post. https://www.masterorganicchemistry.com/2018/05/11/why-are-endo-products-favored-in-the-diels-alder-reaction/
NOTE. Updated April 15, 2019. The top image was modified so that it is consistent with the other images in the post (i.e. “outside” groups turning into dashes). The previous version had them turning into wedges which was confusing for some students. Thank you to Daniel A. for the correction.
“endo product has much more steric hindrance than the exo”
Is there any paper in rather high quality journal with this statement? I have no doubt on ut but my chief asking for pubs to cite such statenent in our manuscript. And I can’t just wrute that it is obvious.
This should not be too hard for you to find.
See one of many examples, Baldwin et. al. in J. Org. Chem. 1989 54, 5264.
This is really really really good. Thank you.
Thanks. If you find it helpful, that’s awesome.
Would have been nice to know all this before I wasted my limited time learning something that won’t be on my crucial entrance exam since it never includes anything controversial or in contradiction of IUPAC. Same thing all over the internet, so I wouldn’t have known if I hadn’t read this comments section.
I am thinking probably that the “out group” trick would predict the correct product for your reaction, but the proper application of IUPAC nomenclature would designate it an exo product? Secondary orbital overlap I think suggests the correct product.
My point still stands: if a formal definition, (like you’ve described from the IUPAC gold book) arbitrarily detracts from our understanding of the chemistry, then maybe it is time to reconsider the definition, or at least loosen it in certain contexts. I think the IUPAC books are updated very infrequently.
But don’t take my word for it, I think many chemists have already come to that conclusion. For example, a recent chemical review product where a monocyclic product is referred to as an exo product: https://pubs.acs.org/doi/abs/10.1021/cr078346g
Regardless, it’s interesting to learn about this quirky nomenclature fact!
According to the strict IUPAC rules, it will be anti, exo/endo are not applicable in this case as the group is on the highest numbered bridge. It’s a mess, I know. Yet, by being the devil’s advocate, I got everyone to scratch their heads more than once and made James write a lengthy and excellently elaborated comment on the topic :D
Would have been nice to know all this before I wasted my limited time learning something that won’t be on my crucial entrance exam since it never includes anything controversial or in contradiction of IUPAC. Same thing all over the internet, so I wouldn’t have known if I hadn’t read this comments section.
If endo/exo is technically incorrect for referring to monocyclic products, then how else should the two diastereomeric products be described?
Regardless of whether the product is a bicycle, the chemical reason for the observed product is the same: there is some attraction (its true sourced debated I think) between the group on the dienophile and the underside of the pi system in the diene. Given that fact, it seems this is a case where “official” nomenclature does a poor job of doing chemistry justice.
I looked through 3 textbooks (Wade, Bruice, Loudon) and 2/3 of the textbooks limit their discussion of endo/exo rule to bicycles. Loudon describes it similarly to what is in this blog post.
I also know that at most of the better schools I’ve tutored students at, the professor will describe the endo product in the same way as described in this blog post: in the endo product, the “out” groups on the diene are cis with the withdrawing group on the dienophile. This is also how I recall learning it in graduate school.
Using endo/exo for monocyclic compounds is as correct as using cis/trans for the two diastereomers of 3-methyl-3-heptene.
Nobody argues about the nature of the product, btw ;) Only how we call it and describe the process. Thus, there are better interpretations of the rule, still using the bridges.
Yes, Loudon p 740 does describe exo/endo same way as this post. Still doesn’t make it correct as the IUPAC is the final authority for the nomenclature.
There’s a lot of poor explanations and incoherent statements across the chemical education. So, as educators, we must hold ourselves to the high standard and acknowledge when we are presenting a trick or a shortcut as opposed to the official rule. Explaining exo/edo via the bridge is the interpretation of the actual rule, explaining it via the relative position of the out-groups and EWG is a shortcut or heuristics if you like. We also must acknowledged when we’re using the official language of chemistry and the “common-talk” when we explain things.
PS: so, what’s the descriptor for the reaction I proposed earlier?
khm…
IUPAC rule P-93.5.2.2.1 The stereodescriptors ‘endo’, ‘exo’, ‘syn’, and ‘anti’
I quote: “if the substituent is oriented TOWARDS THE HIGHEST NUMBERED BRIDGE it is given the description ‘exo’; if it is oriented away from the highest numbered bridge it is given the description ‘endo’. If the substituent is attached to the highest numbered bridge and is oriented towards the lowest numbered bridge it is given description ‘syn’; if the substituent is oriented away from the lowest numbered bridge it is given the description ‘anti’.
So, while your explanation of ‘exo’ and ‘endo’ work here, they are, strictly speaking, incorrect.
Hi Victor – thank you for noting the IUPAC rule, which was an important omission. Given that, can you come up with a reasonable example in the context of an introductory course where the rule-of-thumb rule I proposed doesn’t work? For many of the students I’ve tutored, applying the IUPAC rule as written is not something they are likely to do or to remember….
2H-pyran + maleic anhydride ;)
And I’m all for quick rules that make student’s life easier. However, I like to make sure that it is noted that “this is a trick” rather than the actual rule. Students might (and do) misread. Same thing like I occasionally see the lowest sum of numbers “rule” for numbering a chain which works almost always. With the big emphasis on ‘almost’.
The IUPAC rules are sometimes written in an awkward way, so I agree that they might not be very useful in the “raw” form. Yet, many textbooks also refer to the bridge when explaining the endo/exo, so that’s the big discrepancy here with how books/instructors approach and test it, and how you explain it. Randomly grab Solomons 11th ed from my shelf: p 613.
Finally, using endo/exo for monocyclic products is inappropriate. IUPAC only talks about the endo/exo-like approach, but the actual terms ‘endo’ and ‘exo’ are reserved for the bicyclic structures with combined length of 7 or less atoms between the bridges.
Here’s my view on this.


Short view is: Yes, it’s against IUPAC. Yes, it deserves a note that it does so. But only in a few select cases (below) does it create conflict. Given the tremendous utility of the Diels-Alder, the exo- and endo- descriptors above are omnipresent in non-bicyclic cases in prominent, high-impact journal articles used by all in the synthetic organic community. Journal editors + authors aren’t enforcing the IUPAC standard. Why should I? The article (and site) is meant to be utilitarian. I would rather have someone understand the terms as they are used in the field, rather than puzzle through the IUPAC definition and not obtain an answer. Like the Oxford English Dictionary, IUPAC is a lagging indicator of usage, and is mutable.
[Longer argument]
1. In the context of, say, epoxidation of norbornene or other systems, the IUPAC rules are useful and clear on identifying an exo or an endo face. For example, with the epoxidation products below, it’s clear and unambiguous which products are exo and endo.
2. Due to the popularity of the Diels-Alder reaction, the usage of the terms exo and endo has expanded far beyond this use-case. March (to take one example) and other canonical textbooks are careful to use (italicized) exo and endo to refer strictly to examples where the exo and endo products conform to the IUPAC designation. But when talking about the diastereomeric transition states for the self-reaction of butadiene, even March can’t avoid mentioning exo and endo transition states (even if it’s not italicized) for an example leading to a substituted cyclohexene ring. (I’m using 5th ed).
3. In most situations, endo products tend to dominate. Secondary orbital effects are often cited as the cause. An exo- selective Diels-Alder reaction is a rare thing, generally due to additional steric effects (e.g. 2-chloroacrylonitrile with cyclopentadiene).
4. However, according to IUPAC, the Diels-Alder of cycloheptadiene with (say) acrolein would lead to a product that in every respect follows the same stereoelectronic effects mentioned previously (“endo” selectivity) but because of what I would consider to be a quirk of IUPAC nomenclature, is officially called “Exo”:
5. So clearly there’s a conflict. Which to use? I would argue to screw IUPAC, which is not holy writ, and to still employ the endo and exo in the sense for smaller ring sizes. This avoids any disambiguation in describing the reaction of maleic anhydride with cycloheptadiene with maleic anhyrdide as “exo-selective” when it follows the exact same stereoelectronic course as, say, its reaction with cyclopentadiene.
IUPAC can retire the previous definition.
Thank you for the detailed comment, James. Now a careful reader will find the exhaustive discussion of this topic on this page :)
Great post.
Two small corrections suggested:
– in Example 1, that is not butadiene, but 2,4-hexadiene;
– in Example 3, that is not acrylonitrile, but methacrylonitrile (2-methylacrylonitrile).
Thank you very much.