Acid Base Reactions
Five Key Factors That Influence Acidity
Last updated: May 28th, 2026 |
Five Key Factors That Affect Acidity
Today we’ll talk about five key factors that determine acidity for different molecules and a mnemonic (ARIO, or CARDIO) to help you remember these factors.
These represent important trends in acidity where changing one important variable results in a change in acidity. If you can spot the relevant trend, then you will be able to determine which of various molecules is more acidic – without having to know pKa values.
Let’s quickly review the basics of acidity and basicity. Here’s the condensed version:
- Bronsted acids are proton donors, Lewis acids are electron pair acceptors. Converse: Brønsted base = proton acceptor, Lewis base = electron pair donor.
- A conjugate base is what remains after a molecule loses H+. For instance, HO– is the conjugate base of water. O2- is the conjugate base of HO–.
- Conversely, conjugate acids are what is formed when you add a proton (H+) to a molecule. The conjugate acid of water is H3O+.
- Acidity is an equilibrium where an acid loses H+ to give its conjugate base.
- According to this, anything which stabilizes the conjugate base will increase the acidity. Strong acids have weak conjugate bases, and weak acids have strong conjugate bases.
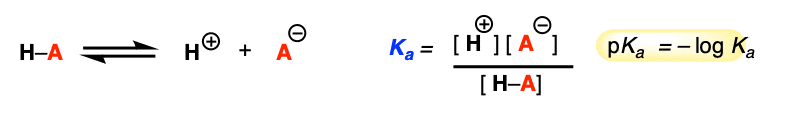
With that out of the way, let’s get started.
Table of Contents
- The Effect of Charge on Acidity
- Electronegativity and Polarizability of the Atom and Acidity
- The Effect of Resonance on Acidity
- Inductive Effects and Acidity
- Orbital Hybridization and Acidity
- The ARIO Mnemonic
- Quiz Yourself!
- Notes
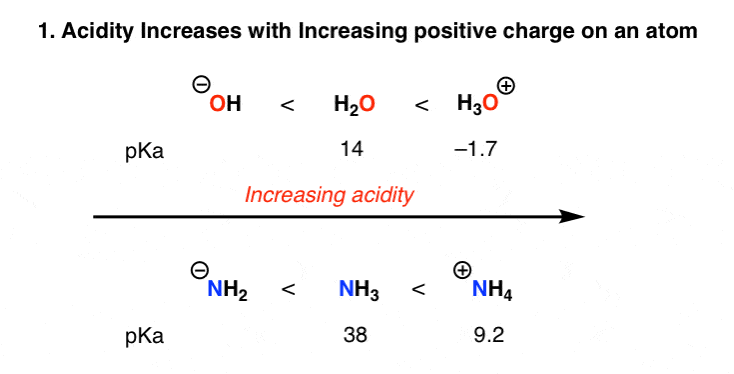
1. The Effect of Charge On Acidity
Removal of a proton, H+ , decreases the formal charge on an atom or molecule by one unit. This is, of course, easiest to do when an atom bears a charge of +1 in the first place, and becomes progressively more difficult as the overall charge becomes negative.
The acidity trends reflect this. H3O+ is a stronger acid than H2O, which in turn is a stronger acid than HO(-).
Note that once a conjugate base (B-) is negative, a second deprotonation will make the dianion (B 2-). While far from impossible, forming the dianion can be difficult due to the buildup of negative charge and the corresponding electronic repulsions that result.
2. The Effect of Electronegativity and Polarizability on Acidity
This point causes a lot of confusion due to the presence of two seemingly conflicting trends.
Here’s the first point: acidity increases as we go from left to right across a row in the periodic table, which corresponds with increasing electronegativity.
This makes sense, right? It makes sense that HF is more electronegative than H2O, NH3, and CH4 due to the greater electronegativity of fluorine versus oxygen, nitrogen, and carbon. A fluorine bearing a negative charge is a happy fluorine.
But here’s the seemingly strange thing. HF itself is not a “strong” acid, at least not in the sense that it ionizes completely in water.
HF is a weaker acid than HCl, HBr, and HI. What’s going on here?
You could make two arguments for why this is. The first reason has to do with the shorter (and stronger) H-F bond as compared to the larger hydrogen halides.
The second has to do with the stability of the conjugate base. The fluoride anion, F(–) is a tiny and vicious little beast, with the smallest ionic radius of any other ion bearing a single negative charge.
Its charge is therefore spread over a smaller volume than those of the larger halides, which is energetically unfavorable: for one thing, F(–) begs for solvation, which will lead to a lower entropy term in the ΔG.
Note that this trend also holds for methanol (CH3OH) and methanethiol (CH3SH) with methanethiol being about a million times more acidic.
3. The Impact of Resonance on Acidity
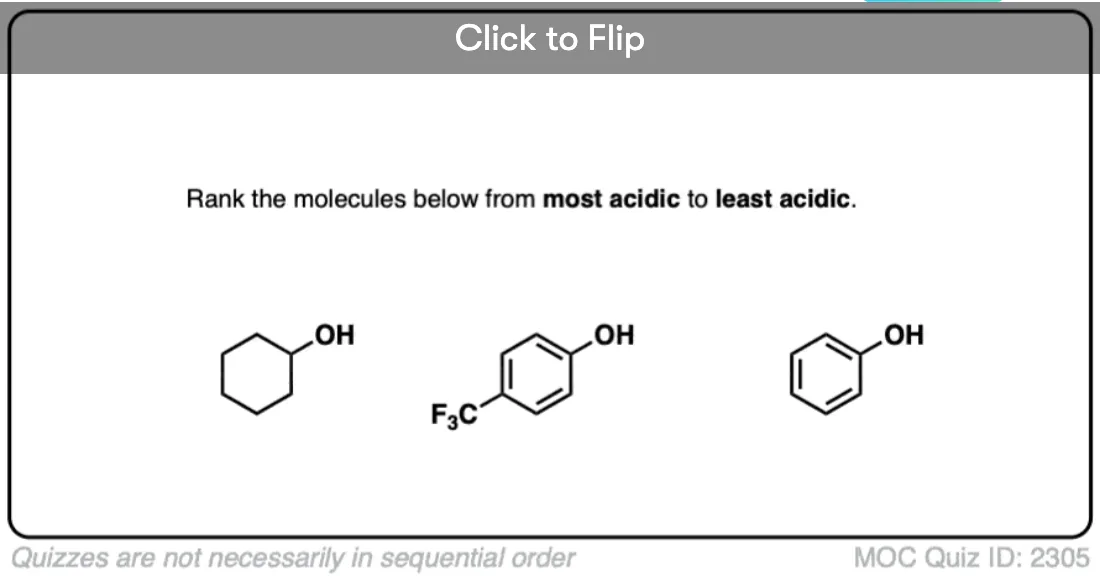
A huge stabilizing factor for a conjugate base is if the negative charge can be delocalized through resonance. The classic examples is phenol (C6H5OH) which is about a million times more acidic than cyclohexanol.
This is because the negative charge in the conjugate base of phenol can be distributed to other atoms through resonance, which helps to reduce the charge density on the molecule and thereby stabilize it.
Resonance also helps to explain the much greater acidity of acetic acid (pKa of ~4) compared to ethanol (pKa about 15-16).
Watch out though – it isn’t enough for a π system to simply be adjacent to a proton – the electrons of the conjugate base have to be in an orbital which allows for effective overlap.
4. Inductive Effects and Acidity
Electronegative atoms can draw negative charge toward themselves, which can lead to considerable stabilization of conjugate bases.
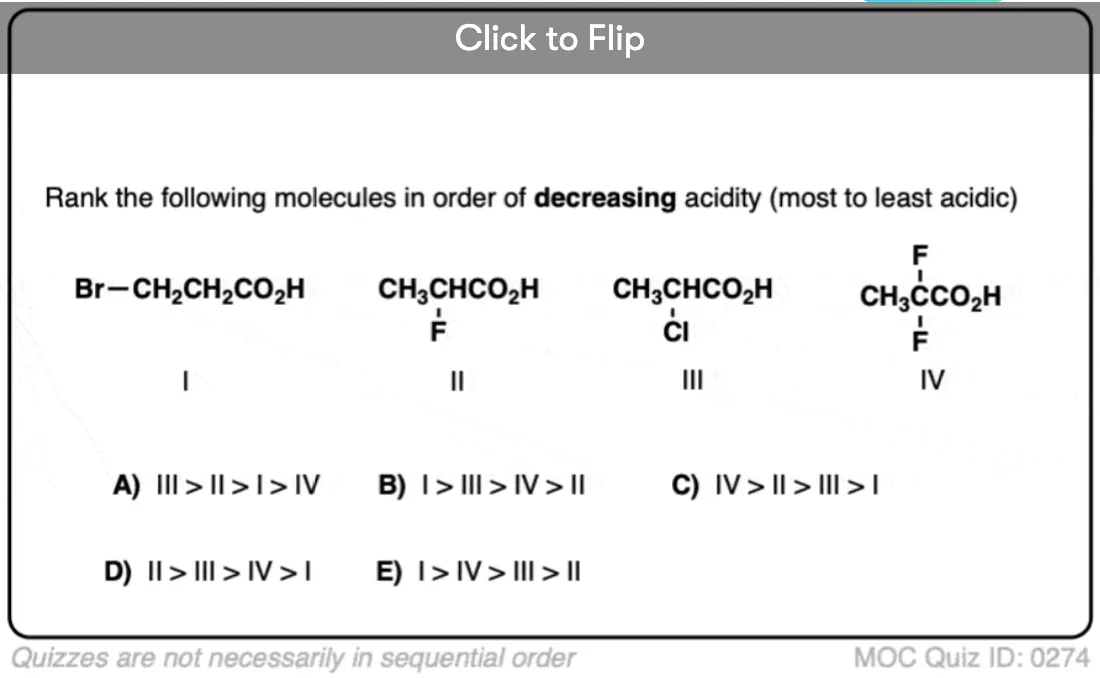
For example, trifluoroethanol (CF3CH2OH) is more acidic than ethanol due to the fact that the electron-withdrawing CF3 group can draw away some of the electron density from the negatively charged oxygen in the conjugate base, which helps to stabilize it.
Inductive effects increase with increasing electronegativity of the attached atom (I < Br < Cl < F).
These inductive effects decrease in importance as the distance increases between the electron withdrawing group and the acidic functional group:
5. Orbital Hybridization and Acidity
Again, the acidity relates nicely to the stability of the conjugate base. And the stability of the conjugate base depends on how well it can accomodate its newfound pair of electrons.
In an effect akin to electronegativity, the more s character in the orbital, the closer the electrons will be to the nucleus, and the lower in energy (= stable! ) they will be.
The difference in acidity between a typical alkane such as propane (pKa = 50) and acetylene (pKa = 25) is 25 pKa units, which is to say 1025 . That’s 100 times bigger than Avogadro’s number, just for the record.
Just to give you an idea of scale. That’s the amazing thing about chemistry – the sheer range in the power of different phenomena is awe-inspiring. (See article: pKa Values Span 60 Orders Of Magnitude)
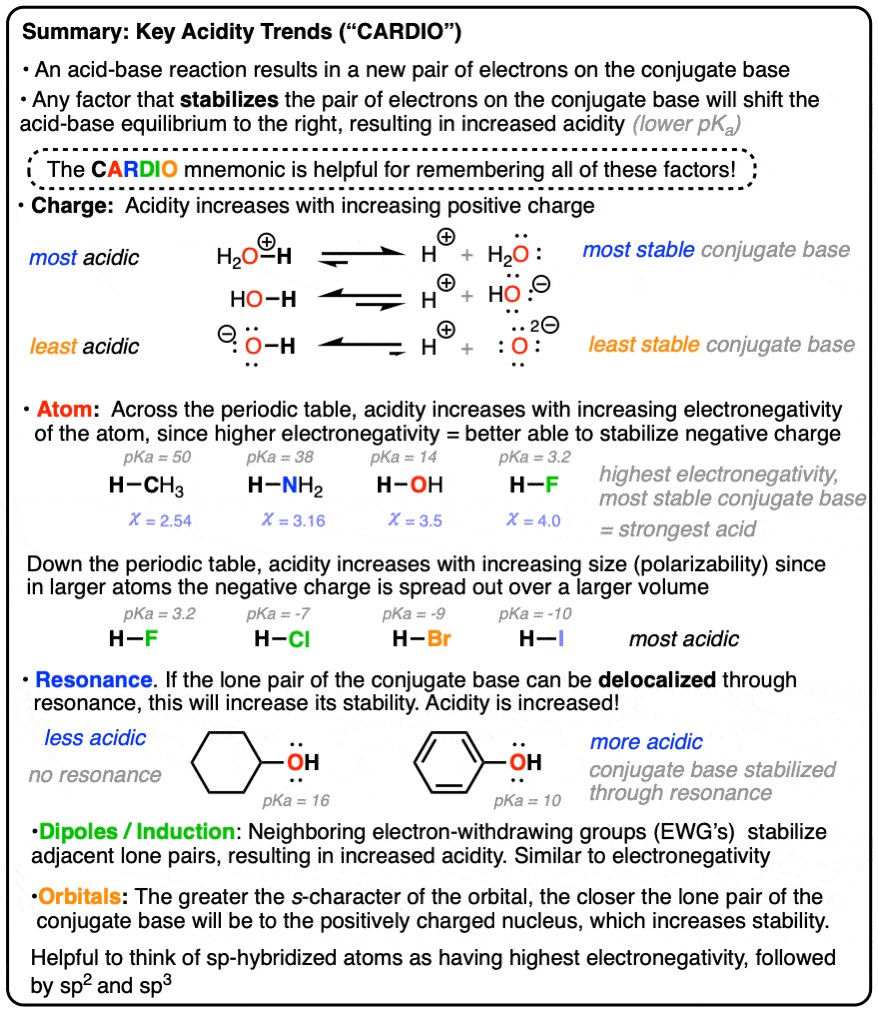
6. The ARIO Mnemonic
To help simplify things, these trends are often combined into the mnemonic ARIO, where
- A is for the identify of the acidic atom (relevant trends – electronegativity across a row of the periodic table, polarizability going down a column of the periodic table)
- R is for resonance
- I is for inductive effects
- O is for orbitals (i.e. sp vs. sp2 vs. sp3.
For completeness, I suppose you can put in the C for charge, which would make it CARIO, or CEPRIO if you separate the “A” into the electronegativity and polarizability effects.
A word of caution with ARIO and related mnemonics: it works well when you are comparing the acidity of similar molecules where only one variable is being changed.
For example, a perfect time to use ARIO is when you’re comparing the acidity of different alcohols (same atom, O) or different carboxylic acids with various nearby electron-withdrawing groups.
It doesn’t work well when two or more variables are being changed, such as in comparing the acidity of CH3CH2OH and H-Cl. In cases like that you really have to rely on knowing values from a pKa table.
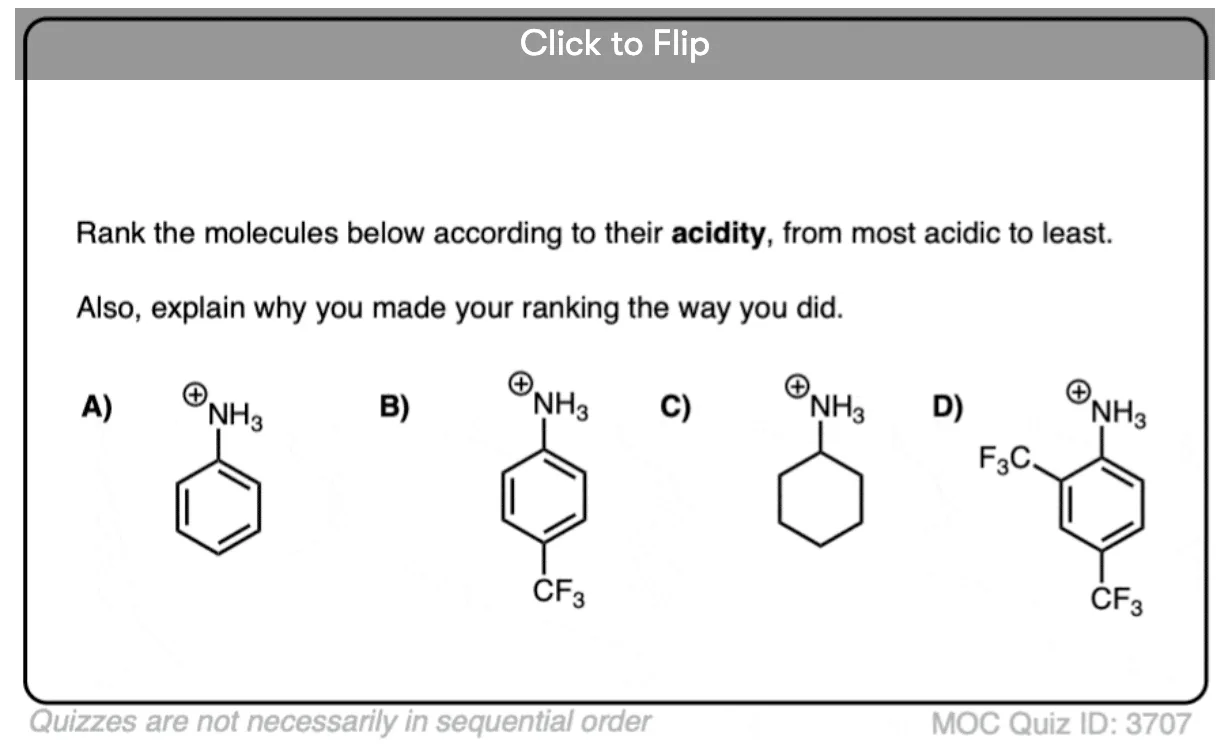
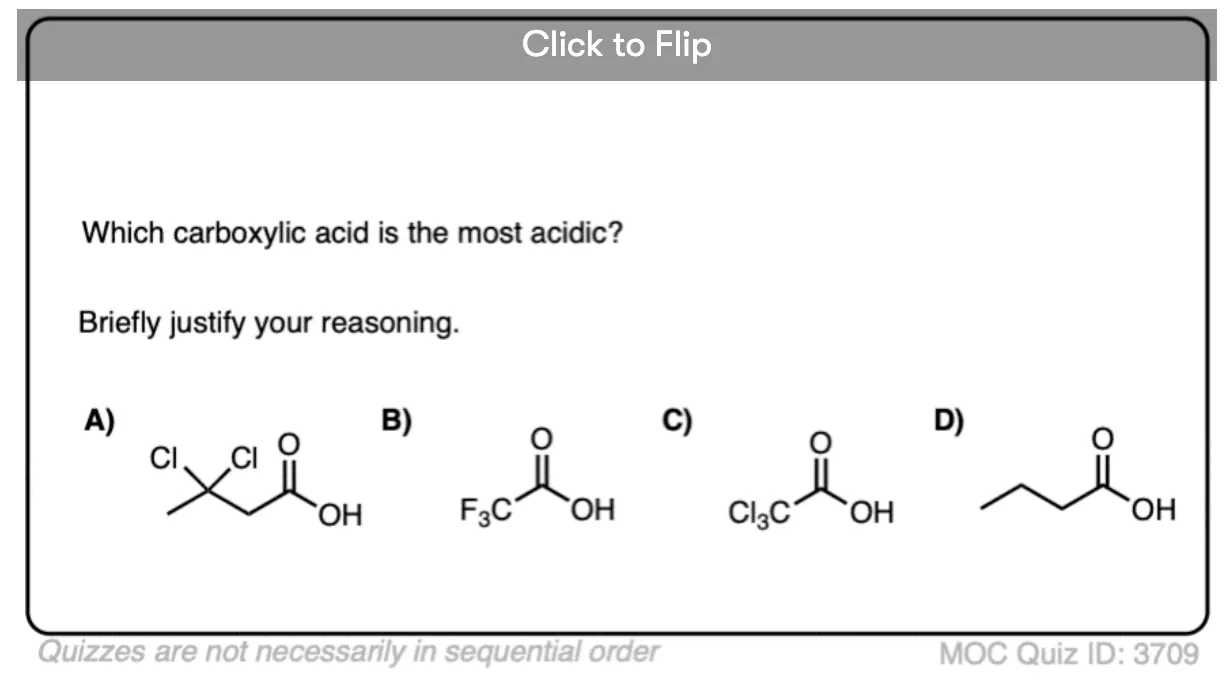
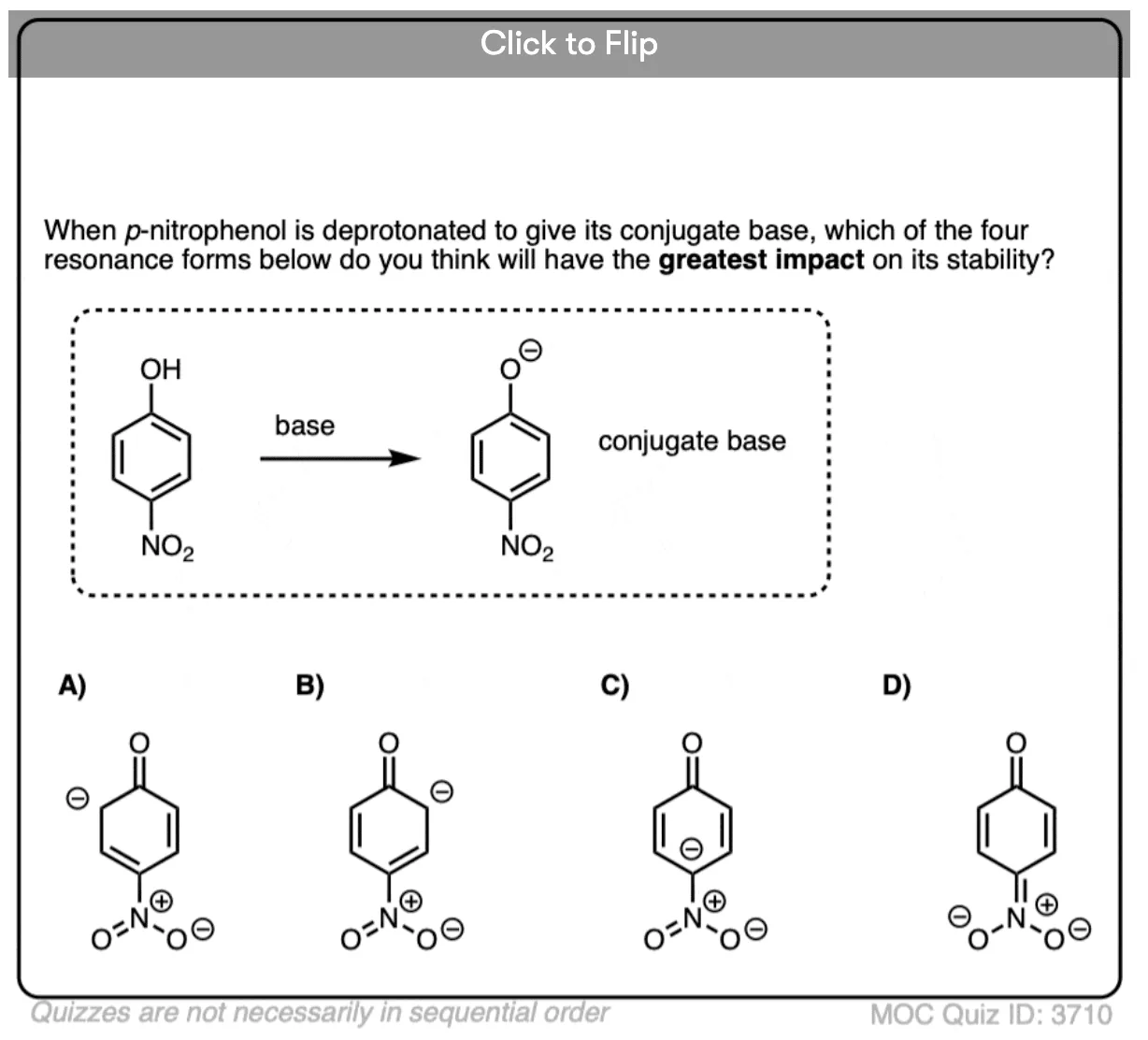
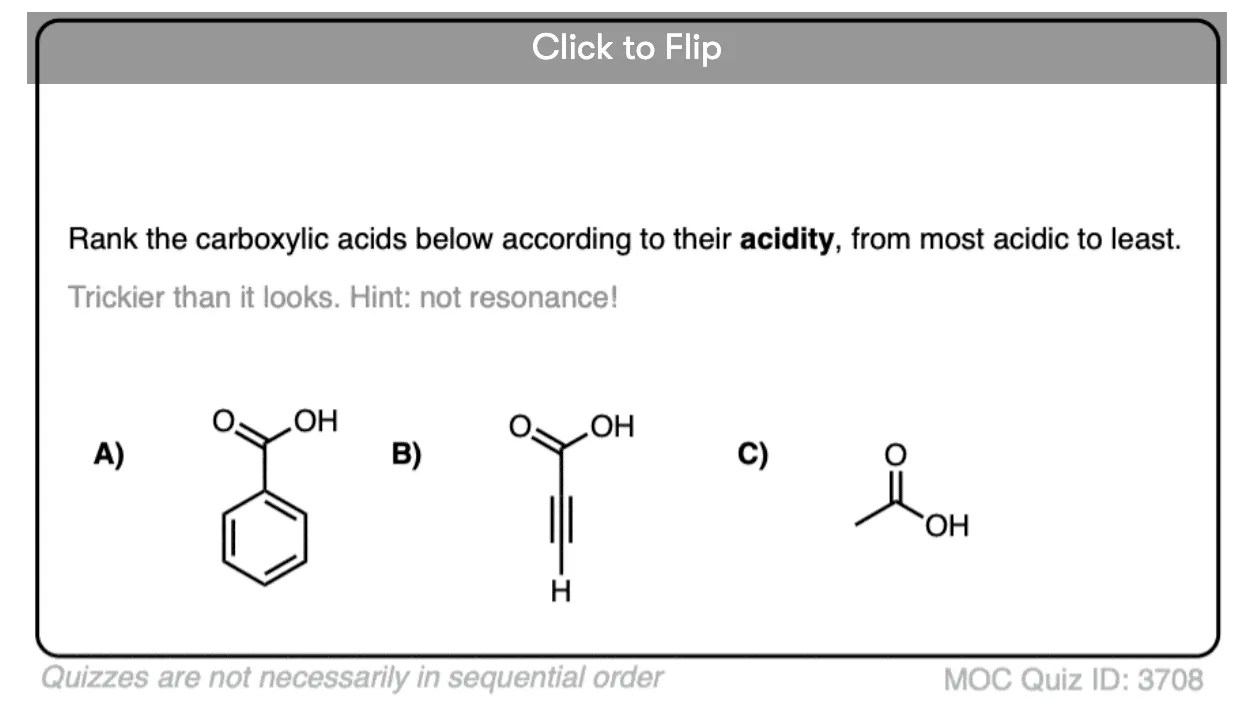
Quiz Yourself!
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.











What about cycles? Do they increase the acidity of a molecule as the number of carbon atoms in their structure decreases?
Thank you for your work! From Brazil.
Hello,
I believe I have told you this before, but it is well worth repeating: this is an amazing website! Thank you for all the work you have done preparing it.
I am the guy scouring the internet trying to clen up the confusion about the pKa of water and the pKa of H3O+ and the pKb of OH-. I see that you have the correct value of 14 for the pKa of water, but I have noticed on this page in section 1 and in the table on https://www.masterorganicchemistry.com/2012/05/09/acid-base-reactions-ka-and-pka/#three
that you have the pKa of H3O+ as -1.74. It is actually 0.00 (Because the pKb (and pKa) of water is 14.00, then the pKa of water’s conjugate acid must be 0.00.) The pKb of OH- is also 0.00.
We just published an open access paper in Helvetica Chimica Acta (https://onlinelibrary.wiley.com/doi/10.1002/hlca.202400103) in which we show that the operation of a combination pH electrode is based on the fact that the pKa of H3O+ is 0.00. This paper also gives a thorough description of many other aspects of the pKa of water discussion. It is worth a read!
Thanks for listening!
can you able to explain everything based on nature that around us?
can you able to explain everything based on nature that around us
Please let me know the reason for increase in acidity of carboxylic acid when a vinyl group directly attaches to it which is contrary to resonance effect.
It isn’t resonance. It’s an inductive effect, essentially.
Alkene carbons are sp2 hybridized and the electrons are held slightly closer to the nucleus. This has the effect of making sp2 hybridized carbons effectively more electronegative than sp3 hybridized carbons.
The effect is even stronger for alkynes (sp hybridized carbons) which have even higher effective electronegativity. For example the pka of propiolic acid is about 1.8.
One more question…How am i supposed to determine that which reason is correct for reasoning the acidity?
Like I got it wrong in above mentioned case!
Thank you
H-I is more acidic than H-F because of longer bond length but in inductive effect OHCOCH2F is more acidic why???
In comparing H-I and H-F, when we lose a proton the charge will be directly on the atom (I- or F-). In that case it’s more stable on I(-) because as we go down a column of the periodic table the atoms get larger and can spread out their charge over a larger volume (polarizability).
In the case of the alcohol, the negative charge is actually on oxygen so polarizability is not a key difference here. The fluorine is electronegative and helps to reduce the amount of negative charge on oxygen through induction.
If acidity increases with increasing positive charge on an atom, is it true that acidity decreases with increasing negative charge on an atom? Carbocations I take it would not increase in acidity because they lack a full octet even with a positive charge?
Quick question.What is the difference in this article from the one previously which describes the seven factors that affect stability of a negative charge?Both seem to be saying the same thing ,but this article does not have the aromaticity part included nor does it say anything on lower charge density stabilizing negative charge.
you just helped me save a whole day in studying acid/basic trends into 30 minutes of the best content review I’ve ever come across. You are an absolute GODSEND.
Glad you found it useful Jane!!!
So, the PKa of H3O+ is 14-14=0?
Is the pka of water 14? Not 15.8?
Yes, the pKa of water is 14.
Your clear and concise explanations are awesome. Hats off to your efforts.
Happy you find it helpful, Raghav.
I have a doubt:
when a polar protic solvent is used as a solvent for its conjugate base, will the conjugate base be the strongest base/nucleophile in that solvent?
Thanks!
I think in the second paragraph of factor #2, you meant, “It makes sense that HF is more acidic (not electronegative) than H2O, NH3, and CH4 due to the greater electronegativity of fluorine versus oxygen, nitrogen, and carbon.”
This is the best explanation that i ever got. Thank you very much. At last i found someone who could clear my confusions thanks again.
Glad it was helpful Raj!
Hi is this mnemonic in order of decreasing effect? Meaning if there were two molecules that I’m trying to decide which one is more acidic, can I say that since molecule A wins out on the dipole induction despite that molecule B wins out on having more “s” character, molecule A is more acidic?
It’s difficult to estimate the magnitude of different effects directly. That’s why pKas are measured. What you should look for is to identify trends within one of these key factors. However if you want to share an example in the comments, that’s cool.
Thank you so much, this was so clear and concise and exactly what I needed to review for my exam!
Excellent, glad it was useful Christine!
Thank you! This helps a lot.
You have done a great job. I was so confused with this topic and after reading reading your explanation my concepts were very clear. Thanks a lot ! ?
Sir you are a fantastic teacher and I am sure that you are also a gentleman. And of course your knowledge of organic chemistry is commendable Really out of the world. Your post are very good and really a big fan of your THANKU ?
Hey I want to know that why ketones are better acids than alkanes??
Draw out the conjugate base (when you remove H+ from the carbon adjacent to C=O). You should have a lone pair that has a negative charge. Now draw the resonance form. You should be able to show that you can form a new C=C bond and the negative charge moves to the oxygen making it O- . So the negative charge has been transferred from a less electronegative atom (carbon) to a more electronegative atom (oxygen) and is therefore more stable. The more stable the negative charge, the weaker the base it is. That’s it in a nutshell. It’s a stronger acid because the negative charge of the conjugate base is weaker.
Taking my DAT in 2 days, and I kept getting these problems wrong on both the Inorganic and Organic chemistry sections… This page cleared it all up!
Glad to hear it, and wish you luck on your DAT! – James
YOU’RE DOING GODS WORK SON
I was totally confused all this time. This teqnique is really helpful thanks a lot
HCOOH, CH3COOH, CH3CH2COOH. Sir, Would you please tell me the order of increasing acidic strength in these acids? It appeared in my exam and i have a doubt in it. Pleaseee help
What is the effect of electron donors on the stability of the conjugate base? Would they increase the negative charge or decrease the negative charge? Would this make the negative charge more stable or less stable?
I just wanted to say thank you for making this information easy to understand! Your website has helped me SO MUCH when I study for my exams. You are the best.
Thank you Tori, I’m very pleased you find the site useful!
this is amazing. So much better than my textbook. Thank you so much!
why R-CH2-CN is less acidic than R-CH2-NH2?
?? Assuming R is alkyl, pKa of R-CH2-CN should be about 30 (DMSO), pKa of R-CH2-NH2 should be 35 or higher. Don’t see any evidence for your claim.
My question is “variations of stability of -I effect in hybridization
which one of the following is correct
(a). Sp>Sp2>Sp3
(b). Sp3>Sp2>Sp
I would consider an sp-hybridized C to be more electronegative than an sp2 hybridized carbon which would be more electronegative than an sp3 hybridized carbon. I hope that answers your question.
This site is just awesome !! Than you sir !!! It helped me a lot !! I recommend every one this site !! Hope this site gets world recognition !!
Thanks Raman, glad you like the site!
Finally, I’ve found a website which helps to break down these concepts into comprehensible pieces. I’m finishing up Gen Chem II this semester, and we’ve already started on Orgo topics since the end of Gen Chem I, so I’m taking Orgo next. This website will surely help me to get ahead, when I study during the summer time. Thank you! :)
Hi Sinigdha, I’m glad you are finding the website useful. Thanks for stopping by!
Hi!
I just want to say thank you, I’m a medicine student from israel and your explanation helped me a lot..
Glad to hear it noazio. Thanks for letting me know!
James, that acronym CARDO is genius. It really helps me understand the factors that affect acidity of molecules. Thank you so much!!
I can’t claim credit for it, but thanks!
All was awesome but I just confused in inductive effect when u compares acidities of halogen acid? As electronegative difference between F and O is less as compared to OH ? So how H lose? May be my question is low of standard as I m new kindly help
for sure this site helped me to improve my understanding on this concept, thank you much. I got 100% of marks on my exam
Your article has been very helpful and I now have actually a chance to pass my exams in general chemistry! I just want to ask, we have to apply these factors in sequence, as you have given them or not??? I mean first the charge, after the atom etc??
thanks alot but what of mesomeric effect
Covered here: https://www.masterorganicchemistry.com/2017/04/28/the-mesomeric-effect-and-aromatic-amines/
Covered here. https://www.masterorganicchemistry.com/2017/04/28/the-mesomeric-effect-and-aromatic-amines/
I am having difficulty determining the relative acidity of amines and ammonium ions. For instance:
a) H3C-NH3+
b) H2C=NH2+
c) HCNH+
d) (CH3)3NH+
How does the difference in bonding between carbon and nitrogen effect the acidity?
In this compounds, carbon atoms may exert two effects, related to the s character of nitrogen orbitals and the electron donating properties of alkyl groups.
The donating effect (by hyperconjugation) of methil groups rises the energy of nitrogen’s electron pair by electron-electron repulsion, making it more basic. Combine this with the fact that sp3 hybridized nitrogen are more acidic (less s character), and we obtain the order d>a>b>c (As would Darwin says: I Think)
Hi,
I found your posts extremely helpful !
In comparing the acidity/basicity of organic compounds, which of the following has the stronger effects?
electronegativity>resonance>s-Orbital>intramolecular force ??
And how to determine the acidity of substituted aromatic compounds, such as benzoic acid?
Thanks in advance.
hello sir,
First of all thanks and now the question- what is +R and -R and how to recognise them. Sir i am stucked please answer urgently.
Dude, it’s just awesome , people like you helps student to learn concepts rather then mugging formulas . I am an Indian , preparing for IIT JEE exam and recommend this blog for every jee aspirants……
May God bless you sir .
My sister had her exams and needed help.
I, although being a student of commerce stream , decided that I had to help her in every way I could. But I knew I sucked at organic chemistry in school,and so did my little sister .
I found your site , and the next day I am understanding things better than I ever did , .. …I would look up about anything I didn’t know on your website .
getting to the main thing , my sister scored 97 in chemistry , full marks in the organic portion.
Thank you sir :’-).
Can you please tell the answer for order of acidity of hydrogen in orbital section…
hey james,
isnt there any explanation fr basicity?
Check out this post on amines. https://www.masterorganicchemistry.com/2017/04/26/5-factors-that-affect-basicity-of-amines/
I love your site and it has helped me a lot :) just one doubt, is electronegativity of an atom related to it’s tendency to act as a LEWIS base. For example NH3 , and PH3,
we know that NH3 is stronger but why doesn’t the electronegativity of the central atom come under consideration here? N has a high electronegativity so it’s tendency to donate lone pair of electrons should be low right? Please please please help.
You mention two factors, electronegativity and size. From first principles it’s difficult to anticipate exactly which would be most important. However, from running experiments on reaction rates (and measuring the results) PH3 tends to be a better Lewis base (“Nucleophile”) than NH3 . The explanation is, the lone pair on phosphorus is less tightly held than that on nitrogen, so it is more “easily” donated. [This is NOT a dumb question – the issue is somewhat complex and can depend on the type of electrophile. I haven’t gotten into Hard and Soft acids and bases but I suggest you look it up]
Hi I had a question. When taking inductive effects (electron withdrawin and electrons donating) into account are we looking at the conjugate base or the acid itself if we wanted to apply rules such as neutral N or O are considered electrons donors?
You’re looking at how well the stabilize (or destabilize) the conjugate base. Anything which stabilizes the conjugate base will result in a stronger acid (and vice versa). For example HOCF3 is a stronger acid then HOCH3 because those three fluorine atoms help to stabilize the conjugate base, -OCF3 through the inductive effect.
I had lot of problems with acidity. Now I have a better understand after reading this article. thank u very much!
OK! Glad to hear you find it useful Chanaka.
I have been looking for a thorough explanation like this for such a long time, being unable to find one. This is really helpful for a med student struggling with his Chem & Intro to Bio exam, thank you so much James.
I had a question if you have a moment! I understand the setup of the Ka formula, but for the life of me, I don’t understand what the numbers ARE. When you punch it into the formula, what numbers are you punching in for certain atoms and/or compounds in order to get the Ka, and then get the pKa. It’s not explained in my text, or any resources I may have. My professor is also not very great at translating our questions in class.
It’s obtained from measurement – from experiment, in other words. It can’t be just “figured out”, we have to physically do experiments that measure the equilibrium constant.
http://sasc-specialists.ucdavis.edu/jim/118A/Acidity_for_118A_Leivens_details.pdf
Basically the same thing explained in a more elaborate ( slightly better ) way.
Please know that I absolutely love what you’ve done. You’ve made organic chem a million times easier. This is just one of the VERY rare cases where I have found a better reference.
Thank you – that is indeed a nice reference.
Thank you so much for creating this easy to understand article. I was about to give up until I stumbled upon this today. This will be the FIRST place I check to help me through orgo! Cheers!
Hi James, firstly, a VERY big thank you to you for this amazing website. Its makes organic chemistry SO easy for me to understand!
I think there might be a slight error in the ‘Orbitals’ section of this article. In the graphic, the acidity increases from sp3 to sp (rightward, ascending order) while the signs ( < ) show otherwise. I might be missing something, so please correct me if I am!
Once again, thanks!
Utkarsh
oops. Thank you. Fixed!
Hi, I’m confused as to how hydrogen bonding in specifically the Halides, affects the acidity.
H-bonding makes the solution tightly binded. Like in case of HF (which shows H Bonding) , all the HF molecules are tightly binded to each other so it s not easy for HF to get ionised into H+ and F-( all molecules are already happy in their state of low energy )
this was so helpful thank you!
why staboilization of conjugate base enhance acidity ?
Equilibrium tends to proceed toward the more stable product, yes? So what happens to the equilibrium HA –> H+ A- as you make A- more stable?
Hey! These pages have been great, where can we find the answers to the problems you usually give at the end?
You must be used to all the gratitude by now but thank you so much! This just made everything fall into place :)
Why is it that HF is more acidic than HI (#2) , but when its connected to the carboxyl group it is I that is more acidic? Anyone?
Thank you so much!! You have made my MCAT studying more a light jog ;)
why is OH- more basic than SH-. “-” is minus sign
Thank you so much! Love reading through everything! You make it so understandable and interesting! I really appreciate the links to more topics too!
Fantastic! What an explanation…. Truely helpful and admirable….
and for factor #5… do we look at the hybridization of the acid or the conjugate base?
ex// ch3 has a hybridization orbital of sp3 but its CB ch2 has a hybridization of sp2
do lone pairs count when figuring out the hybridization of an orbital?
Just wanted to sincerely thankyou for transforming an extremely difficult subject into something comprehensible and futhermore enjoyable. There’s something to be said for that specific talent and it is greatly appreciated. Your use of acronyms and descriptive context have improved both my labs and test marks.
Thank you.
if benzene ring lost a proton it is not stabilizes by resonance because no way for resonance
When trying to choose a compounds with the highest acidity, according to CARDIO, how do you determine which factor you should prioritize first?
For example:
Suppose you’re trying to determine which compound is more acidic, CHCH or benzene ring?
When you remove the proton the benzene ring is stabilized by resonance but the HCC- has a lot of s character in its orbital. How would you determine which compound is more acidic?
i didnt get 5 point orbital as s character increases then t should be sp3<
sp2<sp
This was really clear and helpful Thank you so much!!
Awesome, glad to hear it.
Did you get the CARDIO acronym from a Dr. Christine Pruis or Chad’s Reviews from Arizona State University? Dr. Pruis is our Organic Chemistry professor and came up with this acronym 7-8 years ago, so perhaps that is the ‘credit’ you speak of? If so, that is awesome!
I am curious how you came up with this CARDIO acronym? And when? I have heard it from one other person a few years ago.
I shamelessly stole it from a comment thread on SDN.
Organic is my favourite part in chemistry. Your post and contents provided me a good quick revision before my exams and I did extremely well…..
Thanks to you….you have a good way of teaching organic chemistry..
I would definitely recommend this site to my friends who think organic is boaring subject….
Once again thanks…..
Can you please make these notes available as PDF?
Thanks.
you can use web2pdf for that……
google it…
it also has browser plugin..
This is awesome :D ! I never quite got a hold of this topic since the past 2-3 months and now after reading this page it’s all crystal clear to me ! And as for the mnemonic , when i told it to my chem teacher , he was impressed and asked me for the website :D ! Great job, keep it up :D
Great, glad you found it useful!
Oh wow, this is amazing. I was starting to think there was no site/book that had exactly this information, this compactly (and brilliantly, might I add) presented. Thank you so much! You’ve got no idea how much this helped (and how much it reduced my study time, god knows we can all do with extra time on our hands :) ). Thanks again!
This site is so helpful. It makes organic chemistry way easier. Thank you!
Glad you find it helpful Luis.
On the figure describing inductive effects on pKa of carboxylic acids you have bromoacetic acid with pKa of 2.86 on one line and 2.97 on the next. Why the difference?
Fixed. Thanks for the spot – Evans’ pKa table says 2.86. Not sure where I pulled the 2.97 from.
All these stuff were very useful to me. Got to know more things that I didn’t know before. Thanks a lot..! Similar article on basicity will be appreciated a lot..!
I feel sooo much more confident about orgo bc of u =)
I just don’t seem to understand why does ionic radius increase acidity? I mean, Binding Energy decreases and it’s more easy to lose an electron, if acids are compounds that accept electrons how does acidity increase?
Basicity is all about the stability of negative charge. With larger atoms, essentially the charge is more spread out over a greater volume. Lower charge densities are more stable. See also here: https://masterorganicchemistry.com/2012/04/25/walkthrough-of-acid-base-reactions-3-acidity-trends/
I have a problem where I have to determine the most stable conjugate base, which indicates the strongest acid. I just want to know, is it possible to have a strong acid according to its pKa value, but according to atom, resonance, etc. another acid is stronger?
pKa represents an experimental measurement. Experimental measurements are primary – the concepts we pull out of them, such as the factors mentioned, are secondary. So what you’re mentioning isn’t possible, assuming all other variables are the same.
I just wanted to thank you for all the work you put into this site. I’m an undergraduate at Yale and for the past semester, I’ve been afraid I’ll fail Orgo. Thanks to your site, I no longer feel as stressed because you’ve done such a good job of explaining things. So, yes, thank you!!
Wishing you the best!
Thanks Laina. Glad you find it useful. Let me know if theres anything I can do to make the site more helpful for your needs.
Thank you so much for this summary sheet, I was having so much trouble trying to figure out what made a molecule more acidic – and here it all is! Fully explained and easily understandable. Fantastic stuff.
I love this site… I hate reading organic chem txt books because most are boooooring but you make studying for O-chem the highlight of my friday evening… You are funny, and your delivery style is absolutely amazing!! I am in O-chem II, barely made it through the first but I am excelling in my second…. Thank you sir for doing this!! I appreciate it more than you know… Good day!
I was the most confused person who could not understand the concept. I read your note. I received 100% on Acidity part on my Exam. Thank you!
This is the kind of comment that makes my day. Thanks!
thanks for making org chemistry easier but i have a question about resonance, you didnt mention electron donation or withdrawal by resonance
That’s a thorny issue… it can be hard to separate the influence of inductive and resonance effects. Do you have a specific example that you’re thinking of?
I have a related question. I’ve read that in most cases, resonance stabilization decreases basicity. But is it true that in the cases of guanidines and amidines, resonance increases the basicity? If you could explain how, that’d be great!
I just wanted to say you are doing an absolutely fantastic job of teaching organic chemistry and making it comprehensible. I’ve been reading your posts for the last few days, and material which was alien to me before as finally started to make sense.
Thanks!
Thanks!