Stereochemistry and Chirality
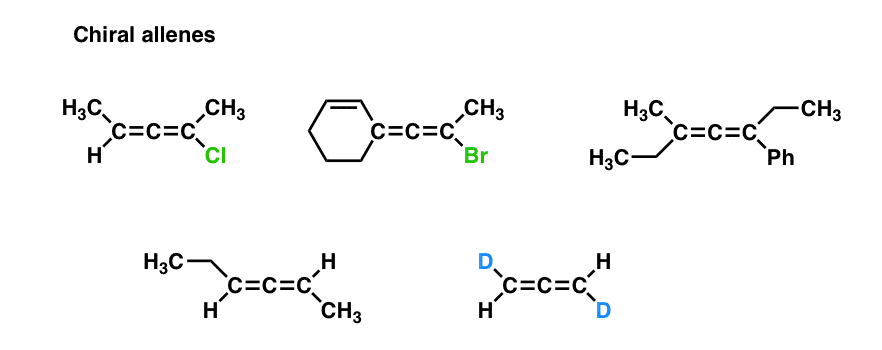
Chiral Allenes And Chiral Axes
Last updated: May 30th, 2026 |
All About Allenes, Chiral and Otherwise
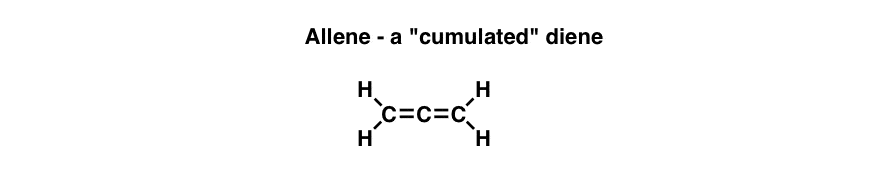
- Allenes are examples of cumulated dienes, molecules with consecutive (not conjugated) double bonds.
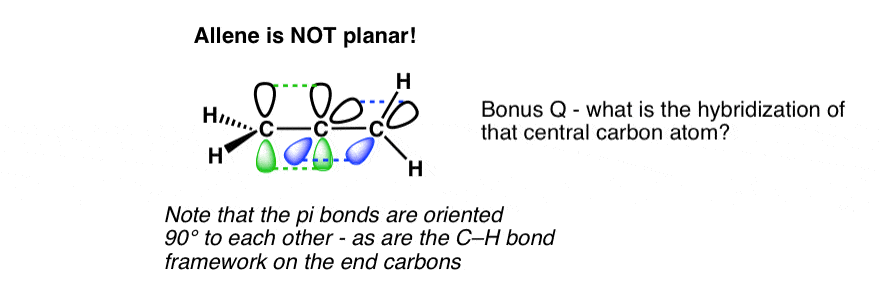
- Allenes are not flat. Due to the fact that the two pi-bonds of allene are at right angles to each other, the end substituents are also turned 90° as well.
- Allene itself has two mirror planes and is achiral.
- However, if both ends of the allene are each attached to two different substituents, the resulting molecule lacks a plane of symmetry and is chiral.
- This type of chirality is referred to as “axial chirality” because it resembles the handedness of screws.
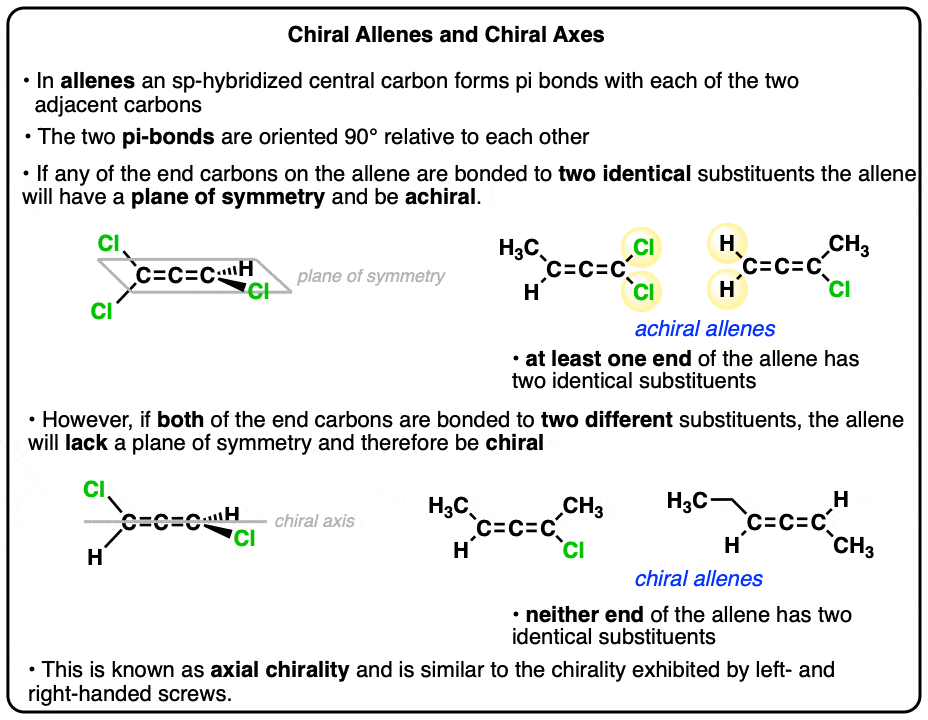
Table of Contents
- Allenes are “Cumulated” Dienes
- The Central Carbon In Allene Is sp Hybridized
- A 3-D “Fly-By” Of Allene Shows It Is Not Flat
- Examples of Substituted Allenes With Mirror Planes
- An Example of A Chiral, Di-Substituted Allene
- Chiral Allenes Have an “Axis of Chirality”
- Summary: How Do I Recognize A Chiral Allene vs. An Achiral Allene?
- Notes
- Quiz Yourself!
1. Allenes are “Cumulated” Dienes
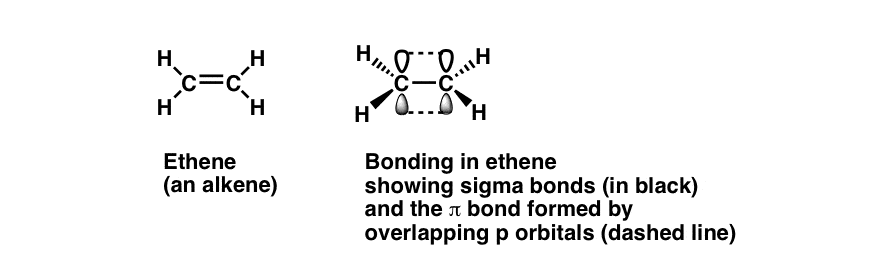
We’ve met alkenes before – right? Just in case you haven’t, let’s review the basics, because today’s post is going to depend on understanding a few of the most important concepts.
Alkenes contain both sigma [ σ] bonding formed through “head-on” orbital overlap [i.e. “single bonds”] and also pi [ π ] bonding where orbitals have “side-on” overlap [i.e. “multiple bonds”]
Alkenes like ethylene are flat [planar]. The p orbitals are at 90 degrees to this planar structure.
If you’ve ever used graphite lubricant, you might know it’s slippery because graphite is made up of layered sheets of flat, pi-bonded carbon, and the sheets have very little friction between each layer. Graphene is the same idea, only it’s a single layer.
Now let’s draw something weird. What do we get when we extend a second double bond directly adjacent to the first one?
Does this look strange to you? The first time you see this, you’ll probably think – yes! You might wonder – do these even exist? The answer is also – Yes! This moleule is called allene, and is part of a family of molecules called cumulenes, so named because the double bonds are cumulative (consecutive).
2. The Central Carbon In Allene Is sp Hybridized
Now let’s ask: what might the bonding in this molecule look like? In other words, what does it look like in 3D?
You might think – simple! It’s flat, like an alkene!
It’s actually not quite that simple! The key is that central carbon – it has TWO π bonds.
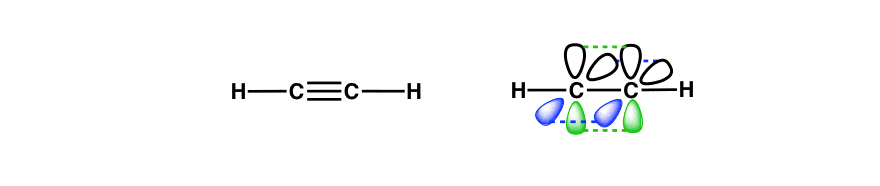
You might recall that acetylene has two π bonds as well, at 90 degrees to each other.
The same is true for allene, except that it’s only the central carbon that is involved in two pi bonds.
Here’s what the orbitals of this molecule look like.
The hybridization of that central carbon? It’s sp, just like the carbons in acetylene. The end carbons are sp2
3. A 3-D “Fly-by” Shows That Allene Is Not Flat
The line diagram of allene really does not do it justice.
So here I present a 3D “fly-by” of allene. Note how the two CH2 groups are “offset” with respect to each other by 90 degrees.
Allene is not flat! [those white balls – imagine they are hydrogen]
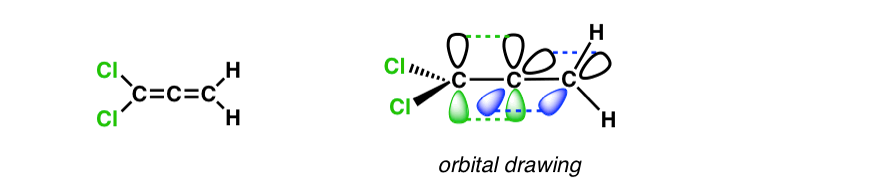
4. Substituted Allenes That Have Mirror Planes
Like so many things in organic chemistry, this simple fact leads to consequences that aren’t immediately apparent. Let’s pay attention to the mirror planes in this molecule. Note that there are two.
Recall that in order for a molecule to be chiral it must not be superimposable on its mirror image. If the molecule has a mirror plane (plane of symmetry) then it will be superimposable on its mirror image [and therefore achiral].
With two mirror planes this is definitely an achiral molecule.
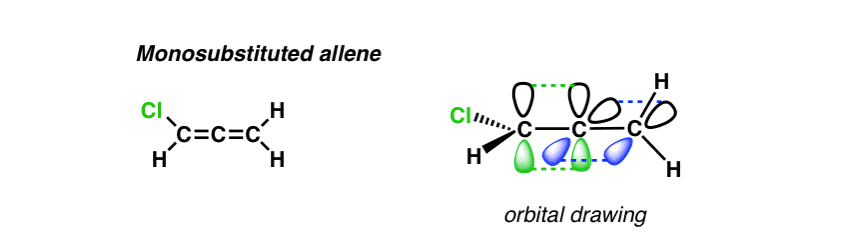
Now let’s start playing around a bit. What happens if we replace one of the hydrogens by another group? In the drawing I’ve arbitrarily made this atom Cl but in practice it can be anything except H in this case.
Here’s a line drawing of the molecule and a picture of its orbitals.
Now here’s what it looks like in 3D. Notice that we’ve lost one of the mirror planes! But it still has one, so it’s still achiral.
Now: what happens if we add a second group to the same carbon?
It’s still achiral! – in fact, we’ve gone back to having two mirror planes.
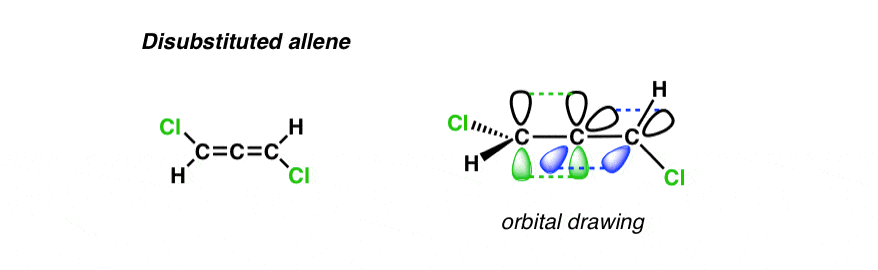
5. This Di-Substituted Allene Has No Mirror Plane – And is Therefore Chiral!
Now, finally, let’s change the position of that second group. Instead of making it so that there are two identical groups on the carbon, let’s change it so that there’s one on each carbon. Like this:
Here’s the 3D version.
Now I ask: where are the mirror planes?
THERE ARE NONE! If there are no mirror planes —> then we are looking at a chiral molecule.
6. Chiral Allenes Have An “Axis Of Chirality”
Wait. You might ask -what trickery is this? We have no chiral centres. Don’t we need something to have a chiral centre to be chiral?
Actually you are very familiar with a chiral object that has no chiral centre.
Have you ever screwed before?
No, no, no! I mean, screwed using a screwdriver. This is a chemistry blog. Don’t expect anything lascivious here.
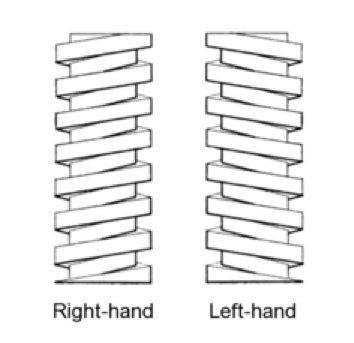
See, most screw threads are, to use a familiar mnemonic, “righty tighty, lefty loosey.” You have to turn the screwdriver clockwise to screw it in, and counterclockwise to loosen. Screws are chiral!
[By the way the enantiomer of that common screw thread would be… “lefty tighty, righty loosey” – those types of threads exist BTW – especially in gas barbecues, so as to prevent people from trying to connect things to them such as garden hoses or other idiotic things].
Here’s a representative picture of two chiral screws. You can also imagine these as spiral staircases. Imagine walking up each staircase. On the staircase labelled “left- hand” below, your right arm would be on the inside as you ascend. On the “right-hand” staircase, your left arm would be on the inside as you ascend. [see Note 2]
Instead of having a chiral centre, screws have what we refer to as a “chiral axis“. Other common examples of things with chiral axes: spiral staircases, snail shells and DNA.
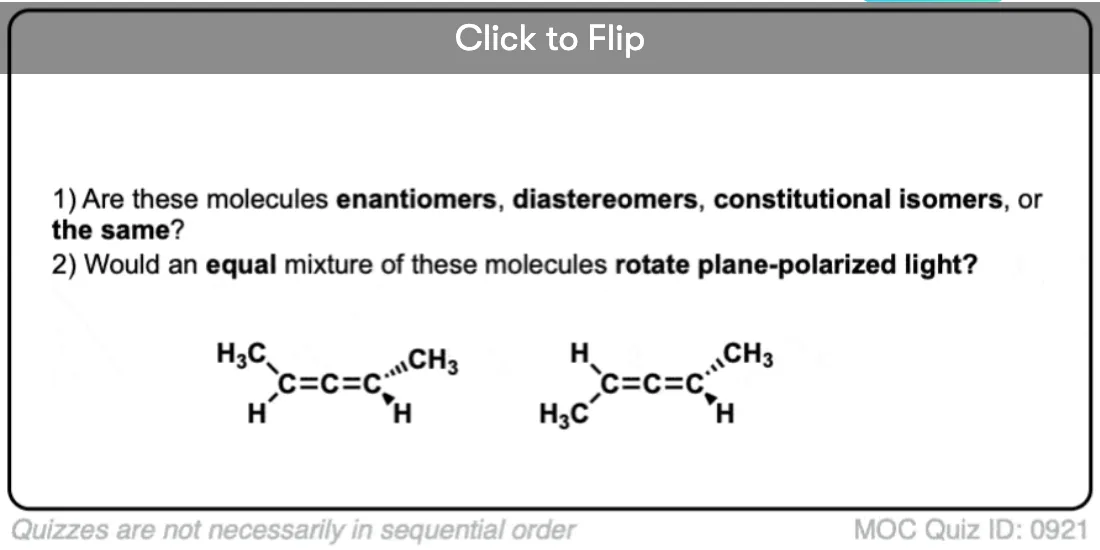
Our disubstituted allene has a chiral axis – just like a screw.
Screws are chiral – and chiral objects have enantiomers. And just as screws have enantiomers, so do chiral molecules with a chiral axis.
What would the enantiomer of the allene look like?
Here is an enantiomeric pair of allenes. Can you see now how they are not superimposable?
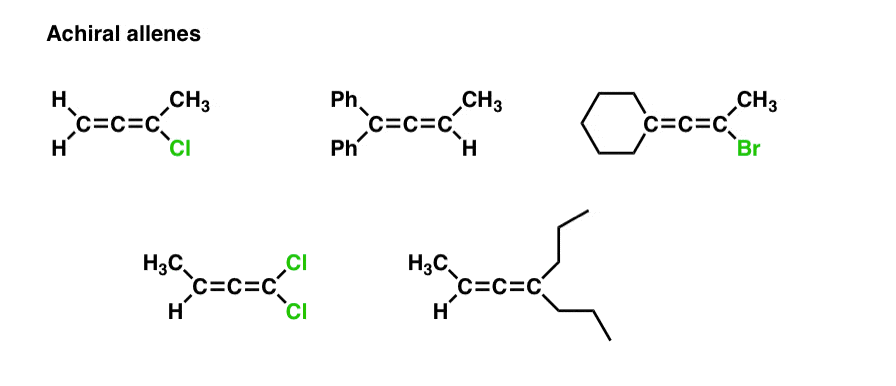
7. Summary: How Do You Recognize A Chiral Allene As Opposed To An Achiral Allene?
[In other words, an allene with a chiral axis vs. an allene without a chiral axis]
You might ask, is there some kind of short cut for recognizing a chiral allene? Sure.
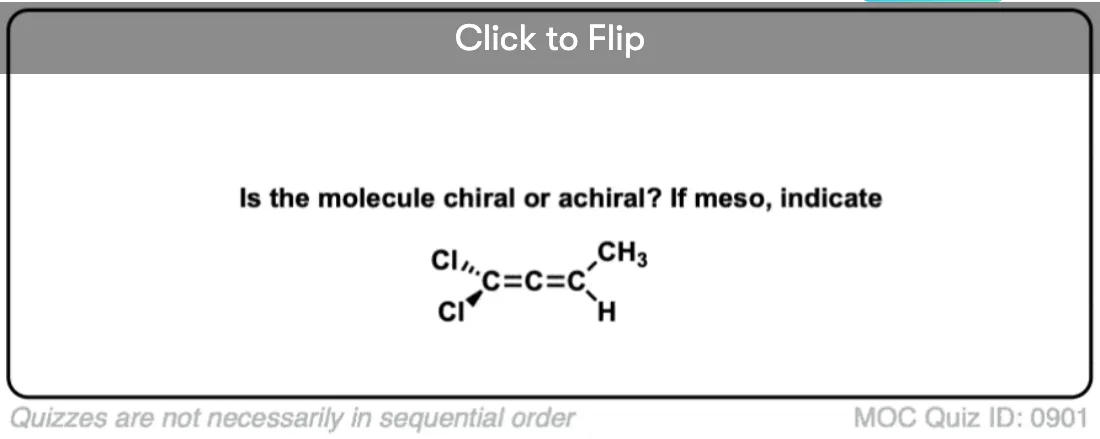
Examine both “ends” of the allene. If either of those ends is attached to two identical substituents, it is achiral – because it will have a mirror plane.
If neither of the ends are attached to two identical substituents, then it is chiral.
Notes
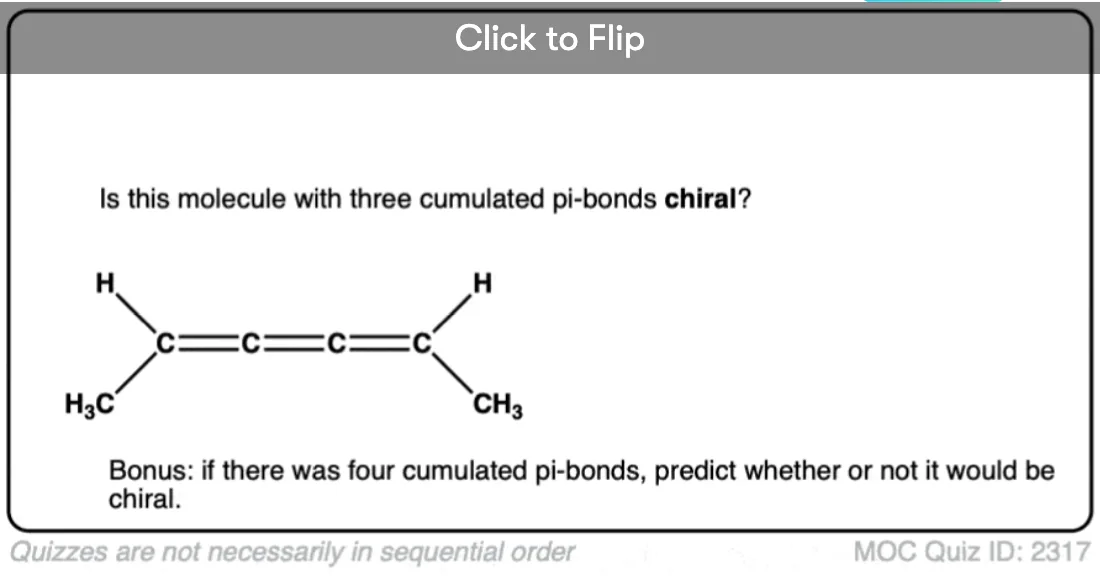
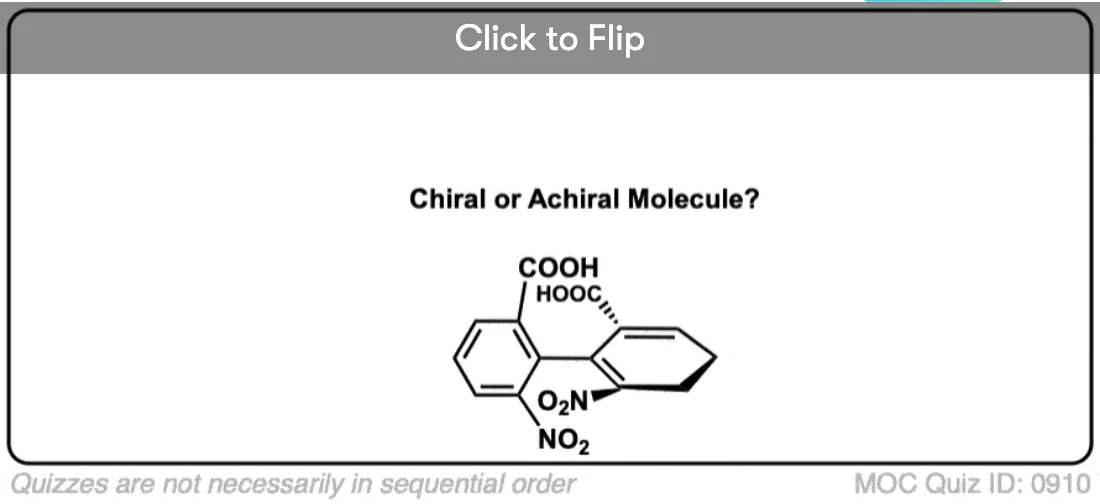
Note 1. Bonus question: Would you expect this molecule to be chiral? Why or why not?
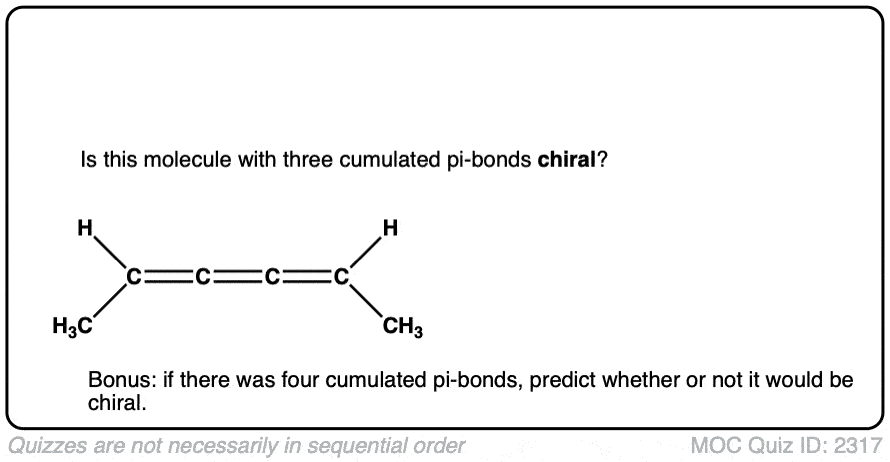 Click to Flip
Click to Flip
Note 2. Nerdy note about staircases, possibly a myth. This may or may not be true, but I recall reading a book in my childhood about castles where it was said that castles were built with staircases that forced someone ascending them [an attacker, presumably] to have their right (sword) hand on the inside and a descending defender to have their sword hand on the outside. The idea being that there is much more freedom of motion if your sword hand is on the outside because you have to swing it across your body.
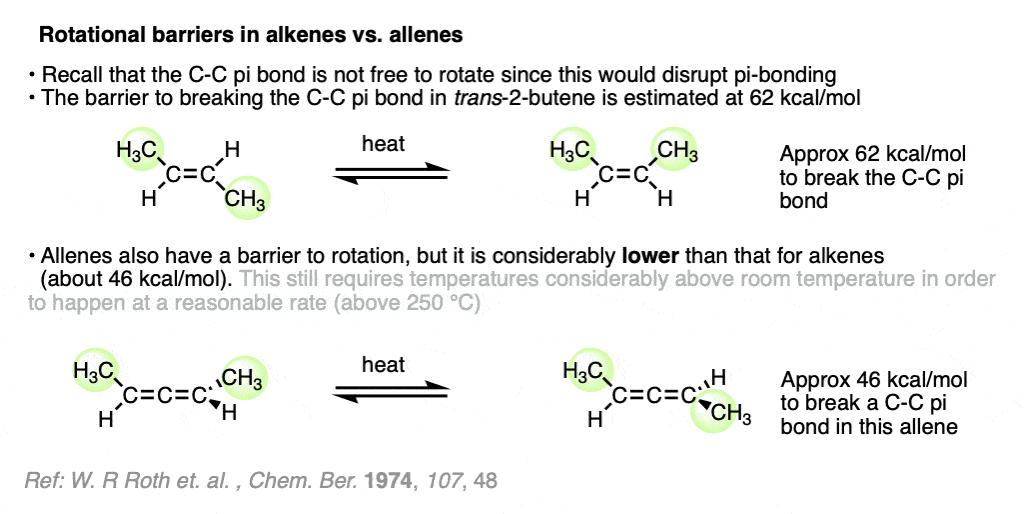
Note 3. Recall that free rotation about the C-C pi bond in alkenes is not possible without breaking the C-C pi bond. This requires about 62 kcal/mol in energy (for trans-2-butene). What about allenes? Considerably less energy is required – about 46 kcal/mol for the allene below. Still, this requires heating the molecule at about 260°C to get any meaningful rate of isomerization.
Quiz Yourself!
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.



can we assign R and S nomenclature for 3 cumulative diene between 4 carbon atoms. for eg C=C=C=C type systems.
For molecules with an axis of chirality, one looks along the chiral axis and numbers the substituents on the near carbon (1) or (2) according to the CIP rules. One then looks at the substituents on the far carbon and numbers them (3) or (4) according to the CIP rules. One then notes whether 1,2,3 goes clockwise (r) or counterclockwise (s). See this article (wikipedia) for an example:
https://en.wikipedia.org/wiki/Axial_chirality
So can we generalize the fact that if the number of double bonds are even with different groups at the ends then the molecule has to be achiral?
Yes, that’s a valid answer
Does a chiral allene contain stereogenic centres? Can we say that the two carbons at the ends are the stereogenic centres?
No, it is referred to as a chiral axis since chirality is not confined to the configuration of a single carbon.
Kind of like how a screw is chiral (left handed or right handed) but you can’t pinpoint an individual center of chirality; instead, it has an axis of chirality as well.
( H cl c=c=c=c H cl ) absolute configuration of it . And why it is optically inactive
See bottom of post.
Great work as always James. In Section 4, right after the line “Now: what happens if we add a second group to the same carbon?” you show a disubstituted allene structure with two Cl substituents on different ends of the allene, when I believe you meant to show them on the same carbon (to represent an achiral disubstituted allene). Keep up the good work.
Darnit. Will fix. Thank you Rich. [EDIT: fixed]
You’re funny. Love the analogies :)
In allene double bonds are shorter than normal C=C bond in ethene.Explain why?
What’s the hybridization of the central carbon? How do bond lengths change as one changes the relative amount of p-character in a bond (sp3 vs sp2 vs sp) ?
Very marvellous presentation sir. Thank you very much. But this is only initial part of chirality in allene. It will be very good if include configurational study of allene
I’m not sure what you mean by “configurational study of allene”.
That’s useful to understand (3D structure)*
Would the bonus question be considered meso because it has an internal plane of symmetry?
Not meso, just achiral.
Spiro[3.3] heptane has two different groups attached(both in same plane) at C1 position and two different groups attached(both on different plane) , would there be a chiral axis?
For spiro[3,3]heptane, it has a plane of symmetry (just like allene). But it could certainly have an axis of chirality if methyl groups were placed on the 3- and 6- carbons, for example. That would be exactly like a chiral allene.
1.)Wat if all the four groups attached to Allen’s are different . Wolud there be a choral axis?
2.) If dibenzene has four different groups attached to the nearest carbons ( two groups on one benzene and two groups on other benzene) then where would be the chiral axis?
If single carbon has four different groups attached ( two in one plane and two in different different planes) , would there be a chiral axis?
1) Yes, there would be a chiral axis in this case.
2) Depends on if the rotation is restricted. If the groups are bulky enough, then rotation is restricted, and the atropisomers can be isolated. But if they are small, then rotation can occur, and conformers will interconvert.
3) Single carbon with four different groups attached does not have a chiral axis, it has a chiral center (asymmetric center).
So if both substituenta are identical on the same side, it can’t be EZ either, I’m assuming?
That is correct.
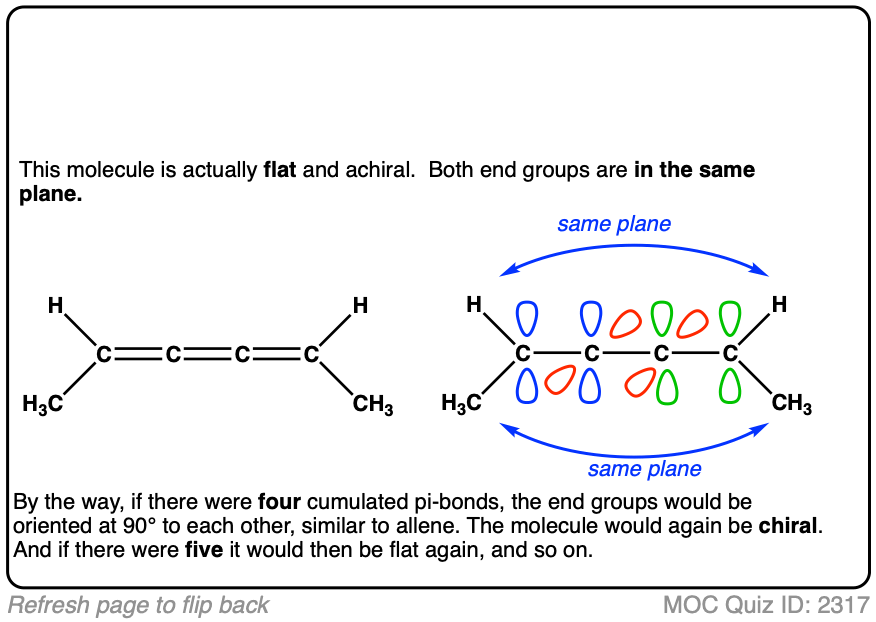
I understand that the bonus molecule is flat, so it’ll be achiral.
But what if it is something like, FCl-C=C=C=CH2 will it still be achiral?
Yes. There’s no “twist” to it – it’s completely flat. So long as the molecule is completely flat, it will always have a plane of symmetry.
Hi James! Do you have any recommendations on molecular kits? I have searched online yet cannot decide on which brand is best. Thanks!
I loved this article. Thanks.
Regarding that bonus molecule, I understand that its achiral. But what if we exchange the hydrogen and methyl group on one of the terminal carbon? Would it still be achiral?
Still flat, and still has a plane of symmetry, so still achiral.
Answer to bonus question here nicely explained by reddit user gtchemist
A possible correction?
Your pictures of screws is confusing, especially when compared with the text. On the left-hand side of the picture, a right-hand screw is shown, and I believe that when ascending it, my left-hand would be inside (contrary to your text).
Repeat the previous paragraph with “left” and “right” switched ;-).
Either your text is wrong, the picture is mislabeled, my 3d visualization skills are faulty, or I’m confusing left/right. But something’s not right.
Thanks – I was ignoring the label and referring to “left-hand” meaning literally the one on the left – fixed the text to make it less confusing! Nice avatar BTW
If you consider descending, then those are right . So instead of changing many things , you could simply change ascend to descend.