Conformations and Cycloalkanes
Cyclohexane Conformations
Last updated: May 28th, 2026 |
Conformations of Cyclohexane (and Cyclopentane)
In the last post, we saw that ring strain of cyclopropane and cyclobutane were 27 and 26 kcal/mol respectively. They are the unhappiest of rings – constrained into uncomfortable angles, with hydrogens forced by geometry to grumpily line up side-by-side with their repulsive neighbours.
The situation for cyclopentane (ring strain: 6 kcal/mol) and cyclohexane (ring strain: 0 kcal/mol) is much happier.
Table of Contents
- Why Aren’t The Bond Angles Of Cyclohexane 120° ?
- The “Flat”, “Envelope” and “Twist” Conformations of Cyclopentane
- The Five Key Conformations of Cyclohexane
- The “Flat” Conformation of Cyclohexane
- The “Half Chair” Conformation of Cyclohexane
- The “Boat” Conformation of Cyclohexane (but it’s actually more like a hammock)
- The “Twist Boat” Conformation of Cyclohexane
- The “Chair” Conformation of Cyclohexane
- Energy Diagram: The 5 Key Conformations of Cyclohexane
- Notes
- Quiz Yourself!
- (Advanced) References and Further Reading
1. Why Aren’t The Bond Angles Of Cyclohexane 120 Degrees?
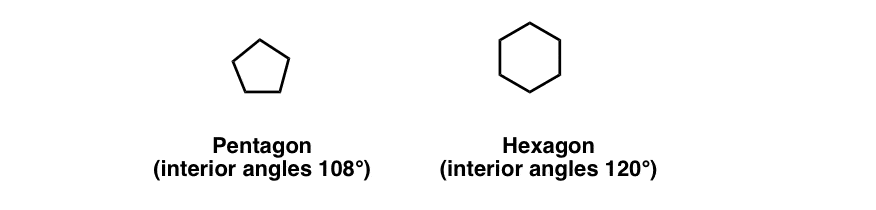
First, though, the answer to the riddle from the last post. At first glance, the fact that cyclopentane is more strained than cyclohexane might seem strange because pentagons have bond angle of 108° (very close to the ideal tetrahedral angle of 109°) and hexagons have a bond angle of 120°.
Shouldn’t we expect that cyclohexane is more strained than cyclopentane, not less? What’s going on?
You might see the key fallacy in this line of thinking. What’s the key difference between a pentagon and cyclopentane, or a hexagon and cyclohexane?
The key difference is that the molecules don’t necessarily have to be flat! In fact, as we saw for cyclobutane, “flatness” is disfavored, because it leads to eclipsed C-C bonds, which can have considerable torsional stain. In fact we saw that the dominant conformation for cyclobutane was the “puckered” conformation, where one of the carbons pokes out of the plane created by the other 3 carbons. This relieves torsional strain.
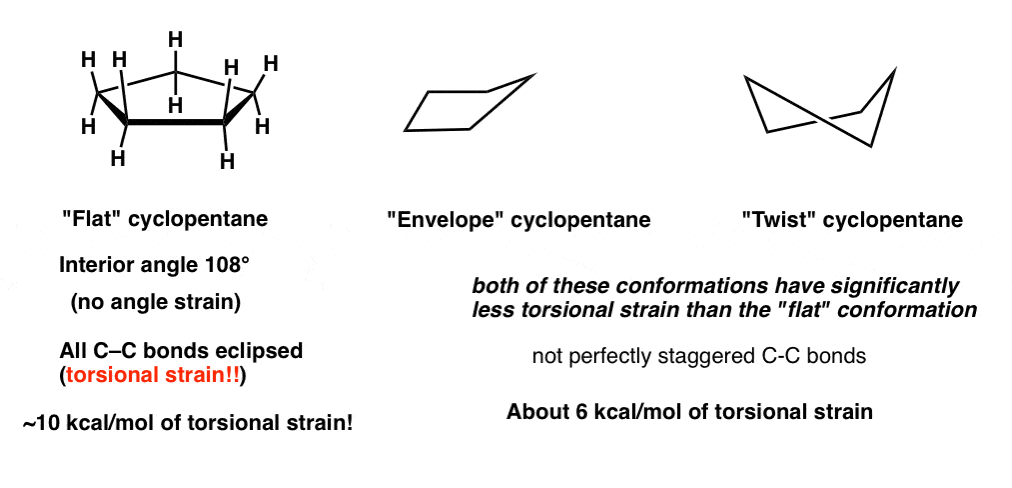
2. The “Flat”, “Envelope”, and “Twist” Conformations Of Cyclopentane
Similarly, for cyclopentane, the “flat” conformation has all of its C-C bonds eclipsed and has considerable torsional strain (about 10 kcal/mol). However, since there is some flexibility, what we in fact observe is two dominant conformations for cyclopentane (of comparable energy), the “envelope” and the “twist”. While each of these conformations has some torsional strain (the C-C bonds are not perfectly staggered), both of these have considerably less torsional strain than flat cyclopentane.
[By the way, these conformations of cyclopentane are not just of purely academic interest – ribose and deoxyribose are 5-membered rings whose conformations have tremendous biological relevance. See Note 1 at bottom]
3. The Five Key Conformations Of Cyclohexane
So that helps to explain why cyclopentane has at least some ring strain.
What about cyclohexane?
Glad you asked! Well, find a comfy thing to sit in – a chair, or a hammock if you will, because over the next few posts, we are going to talk and talk and talk about cyclohexane, and then talk some more. There will be no ring more important to us in this course than cyclohexane.
In this post, however, I just want to sketch out the basics. What are the different conformations (shapes) that cyclohexane has, how do they differ in energy, and why? For best results, I suggest making a model of cyclohexane.
[When many students hear someone say, “make a model”, it’s often like hearing, “go ask your mother”, so really, do what you want. But I promise that it will help as we go through this.]
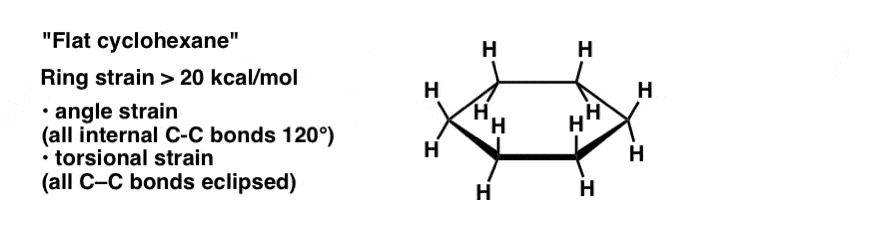
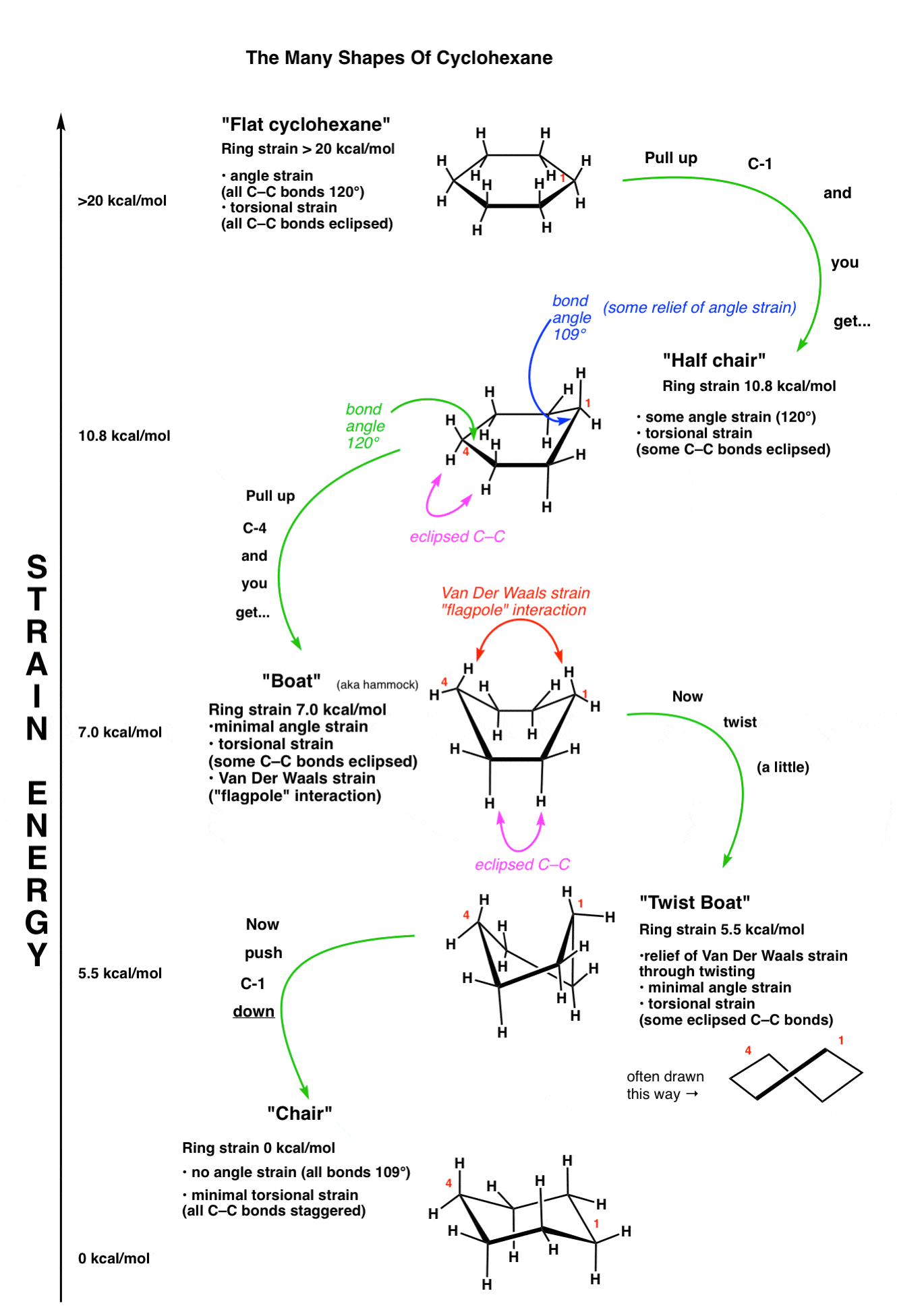
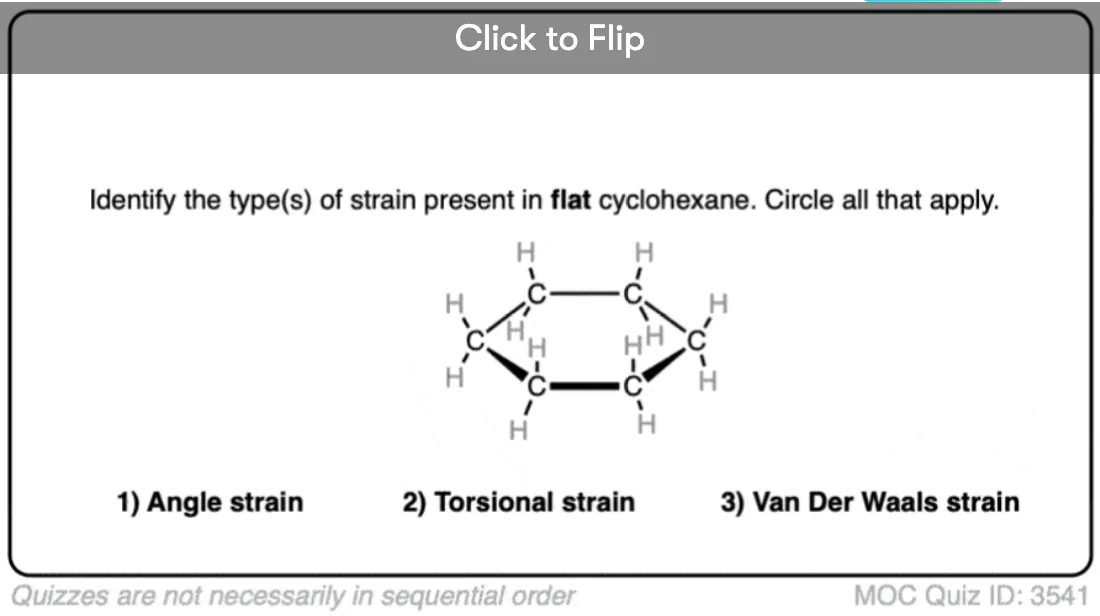
4. The “Flat” Conformation Of Cyclohexane
Flat cyclohexane is very strained (about 20 kcal/mol). Not only is there angle strain (with internal angles at 120°, above the ideal angle of 109°) but every C-C bond is eclipsed, so there is also tremendous torsional strain. In solution, the flat conformation of cyclohexane is generally of too high an energy to be observed.
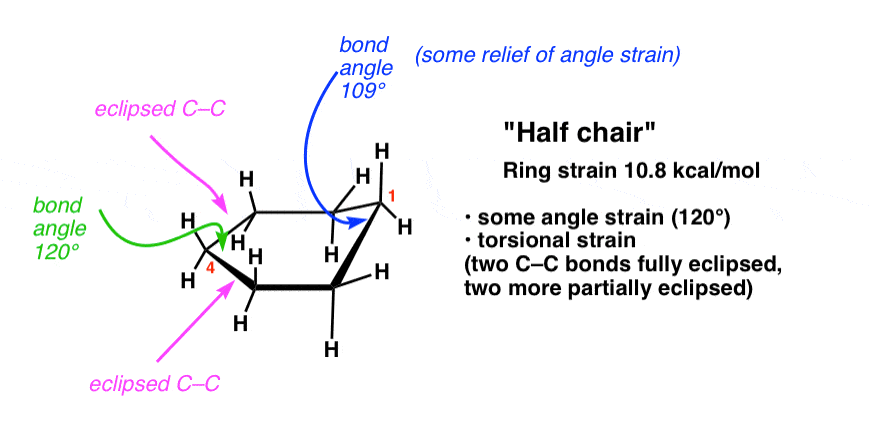
5. The “Half Chair” Conformation Of Cyclohexane
If one takes one end of the cyclohexane and “pulls” it up, then we will have a “half-chair” conformation of cyclohexane. If you try this with a model you will note that there will be rotations about 4 different C-C bonds, and this will result in them no longer being completely eclipsed. Furthermore the bond angles upon doing so will more closely resemble the ideal tetrahedral bond angles of 109°.
This results in significantly less torsional strain and angle strain. The strain energy relative to a theoretical “unstrained” ring is about 10.8 kcal/mol.
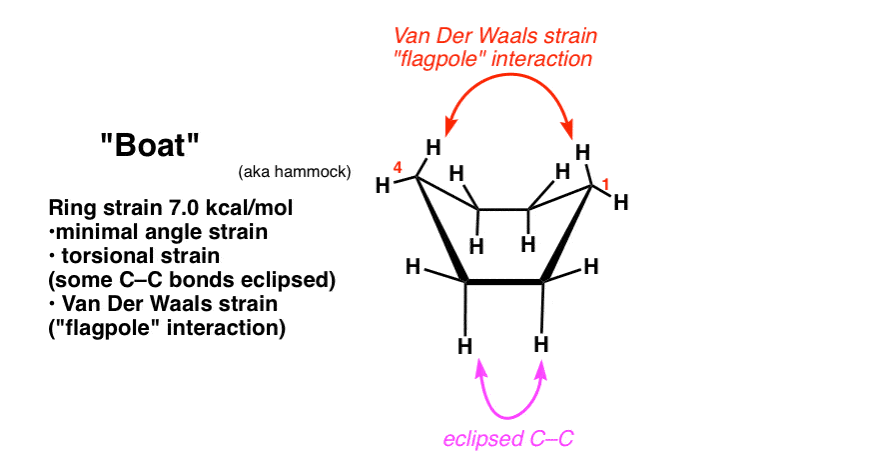
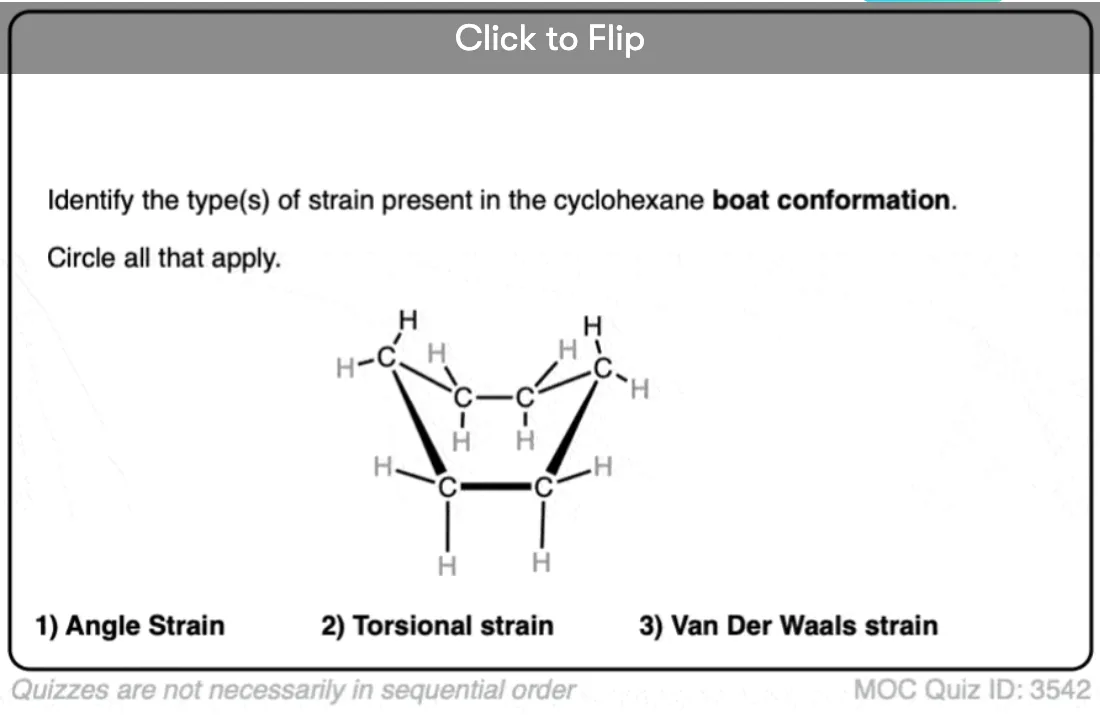
6. The “BOAT” Conformation of Cyclohexane
Taking the other end of the half-chair and pulling it up creates a structure which resembles a boat (or, as I modestly propose, a hammock). There is no longer significant angle strain, since all internal angles can now be 109 degrees. There is still torsional strain, however, as if you look carefully you will see that along two different C-C bonds, the C-H bonds are all eclipsed [there’s a total of 4 eclipsing H-H interactions].
Furthermore, there is an additional source of strain in the hydrogens of the “prows” of the boat. In the boat conformation these hydrogens are held together in such close proximity that their electron clouds come close enough together to repel, an effect known as Van Der Waals strain. Sometimes this interaction is known as the “flagpole” interaction. The ring strain in the boat is about 7.0 kcal/mol.
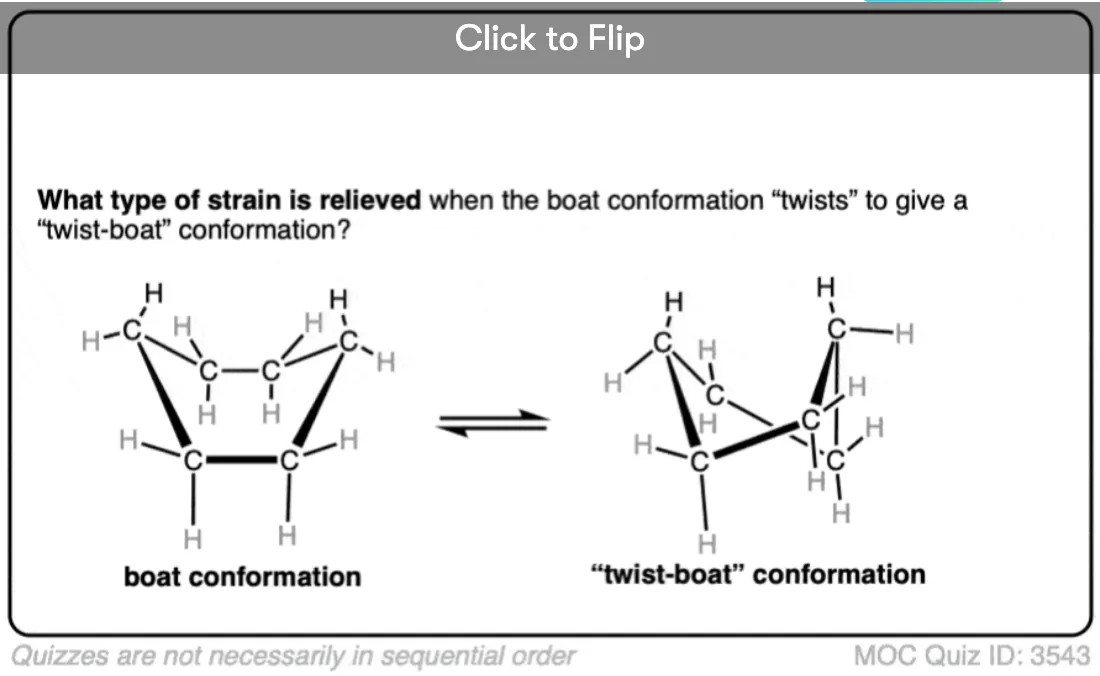
7. The “Twist Boat” Conformation Of Cyclohexane
. A significant portion of Van Der Waals strain of the boat can be relieved by a slight rotation of the bonds, resulting in a conformation known as the “twist boat”.
Drawings don’t do it adequate justice – for this one you really need to build a model to see how a slight twisting of the structure can result in less strain. There is still significant torsional strain since there are two C-C bonds about which the hydrogens are not perfectly staggered.
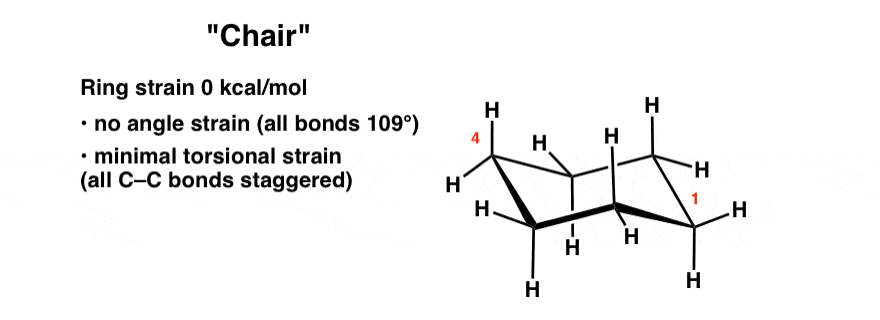
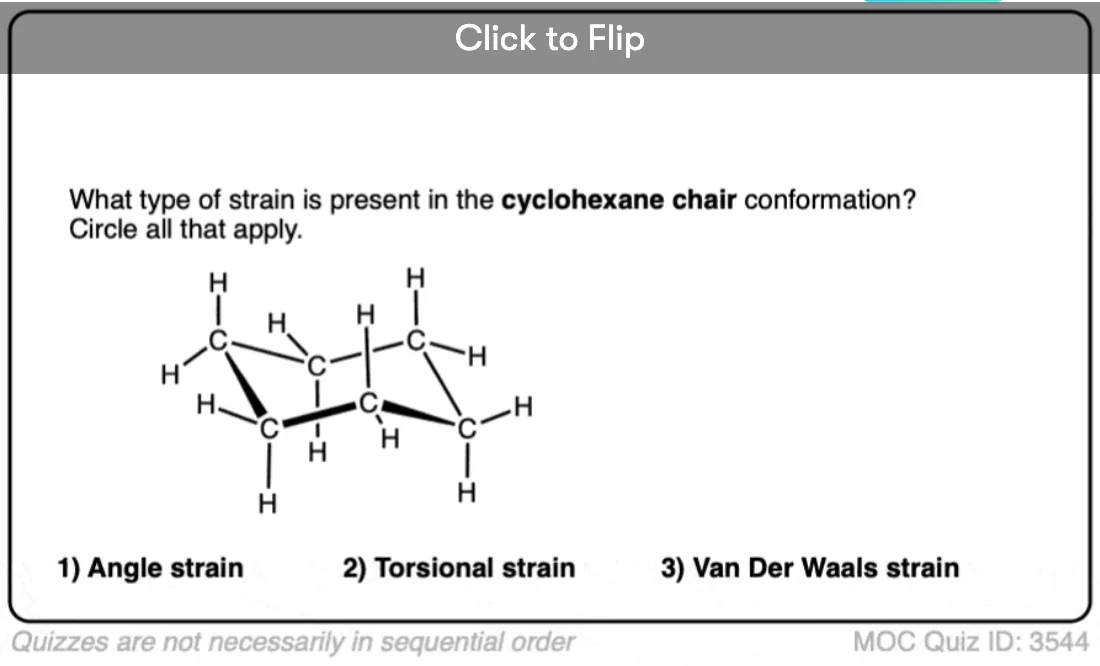
8. The “Chair” Conformation Of Cyclohexane
If one starts with the “half chair” and then pulls the “flat” end in the opposite direction of the carbon that is poking out of the plane, the result is a structure with 4 carbons in the same plane and one carbon each poking above and below the plane. This resembles a deck chair, so it goes by this name.
The chair is a perfectly unstrained molecule. There are no eclipsing interactions; the conformation along each C-C bond is staggered. Furthermore the orientations of the atoms about each carbon are perfectly tetrahedral. Since it lacks ring strain and is of lowest energy, the chair conformation is the dominant conformation that cyclohexane will be found in solution.
Time for some bold type: The chair conformation is the most important conformation of cyclohexane to understand.
9. Energy Diagram: The 5 Key Conformations Of Cyclohexane
Putting all of these conformations together, we can obtain this chart, with highest energy at the top:
If many of these conformations and shapes still seem strange to you, don’t worry! In the next post we are going to talk in more detail about the cyclohexane chair conformation, including how to draw it properly.
Notes
Note 1. On sugars and conformations. Quote from my friend Jon, who is much more knowledgable than myself about these matters:
“In double helices – B–DNA (the classic double helix) basically has sugars with a south pucker. Double-stranded RNA, and also A-DNA (found in other sequences/conditions) basically has sugars with a north pucker.
Interestingly, kinases that are needed to activate nucleoside drugs (AZT and many others) recognize the puckers and so the pucker of a nucleoside can be key to allowing it to be activated as the triphosphate and incorporated into DNA. Victor Marquez of the NCI (NIH) is the authority in this area of nucleoside conformations and their biological activity.”
Quiz Yourself!
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
(Advanced) References and Further Reading
Cyclopentane and cyclohexane are significantly less strained than other alicyclic hydrocarbons, because they are at the ‘sweet spot’ where they are flexible enough to avoid unfavorable diaxial or syn-pentane interactions.
- THE STRUCTURE OF ALICYCLIC COMPOUNDS
G. Aston, S. C. Schumann, H. L. Fink, and P. M. Doty
Journal of the American Chemical Society 1941, 63 (7), 2029-2030
DOI: 10.1021/ja01852a508
An early paper that suggested cyclopentane has a non-planar structure based on vibrational (IR or Raman) data. - Conformations and Strain Energy of Cyclopentane and its Derivatives
Kenneth S. Pitzer and Wilm E. Donath
Journal of the American Chemical Society 1959, 81 (13), 3213-3218
DOI: 1021/ja01522a014
An early paper which calculates the strain energy of cyclopentane to be 8.5 kcal/mol. - Vibrational Spectra and Structure of Cyclopentane and its Isotopomers
Esther J. Ocola, Leslie E. Bauman, and Jaan Laane
The Journal of Physical Chemistry A 2011, 115 (24), 6531-6542
DOI: 1021/jp2032934
A more recent paper which does a thorough combined experimental and theoretical analysis of the potential energy surface of cyclopentane. Interestingly, it is extremely small – the difference in energy between the twist and bent forms is less than 0.1 kcal/mol. - Ueber Polyacetylenverbindungen
Adolf Baeyer
Ber. 1885, 18 (2), 2269-2281
DOI: 10.1002/cber.18850180296
The original paper on ring strain by the legendary chemist Adolf von Baeyer. Even though this paper is titled on a completely different topic, ring strain is discussed at the very end of the paper. - Evaluation of strain in hydrocarbons. The strain in adamantane and its origin
Paul von R. Schleyer, James Earl Williams, and Blanchard K. R.
Journal of the American Chemical Society 1970, 92 (8), 2377-2386
DOI: 1021/ja00711a030
An early paper by Prof. P. v. R. Schleyer before he moved to Germany in the 1970’s which uses a different set of calculations than those employed previously, arriving at a value of 1.35 kcal/mol for the strain of cyclohexane. Table
VII in this paper has a large collection of strain energies of various hydrocarbons, including cyclopentane, cyclohexane, and adamantane (7.19. 1.35 and 6.48 kcal/mol, respectively), especially noteworthy since up to this point adamantane was thought to be relatively “strain-free”. (Adamantane was a pet topic of his, as one of his most highly-cited papers is a 1-page communication in JACS on the simple synthesis of adamantane.) - The Concept of Strain in Organic Chemistry
Kenneth B. Wiberg
Angew. Chem. Int. Ed. 1986, 25 (4), 312-322
DOI: 10.1002/anie.198603121
Ring strain can also be called ‘angle strain’, resulting from the distortion of bond angles, increasing the energy content of the molecule. This paper also discusses the propellanes, an interesting class of small strained molecules. While [1.1.1]propellane can be isolated, [2.2.1] has not been obtained as a pure substance yet. This is due to the strength of the central bond towards homolytic cleavage, which provides a path for decomposition. This energy is strongly influenced by the difference in the strain energy between the reactant and the resulting diradical. In [1.1.1]propellane, the difference is 65 kcal/mol, while in [2.2.1]propellane, it is 5 kcal/mol. - Ueber die geometrischen Isomerien der Hexamethylenderivate
Sachse
Chem. Ber. 1890, 23 (1), 1363-1370
DOI: 10.1002/cber.189002301216 - Über die Konfigurationen der Polymethylenringe
Sachse
Z. Phys. Chem. 1892, 10U (1), 203-241
DOI: 10.1515/zpch-1892-1013
Sachse was the first to have the insight to recognize that cyclohexane does not need to be flat, and furthermore adoption of a bent shape (which we now call the “chair”) alleviates its angle strain. Unfortunately he passed away in 1893 before being able to further elaborate his ideas.


James – What does this bold line in cyclo-hexane really means ? Usually bold lines are referring to the atoms that point towards us and if that being the case we should have four bonds towards us but the literature shows only three lines or I am misunderstanding something here.
What can be said about rotation around a C-C single bond in a ring molecule?
It is constrained by ring size. Building a model will make this apparent. The reason that geometrical isomerism is possible in cyclic molecules (e.g. cis 1,2-dimethylcyclohexane and trans 1,2-dimethylcyclohexane) is because rotation to interconvert these molecules is impossible without destroying the ring.
Once the ring gets large enough (8+) such interconversions become possible.
The work is very relevant and helpful. Thank you so much, keep it up.
I can literally survive college!!!!
I found it really helpful. Thanks a lot :D
This website is amazing and I appreciate that you guys keep a lot of information accessible to everyone!
Thank you Samima! Please tell your friends :-)
Thank you so much. I’ve never had so much clarity before.
This is a brilliant website!
I just have one doubt regarding the chair conformation of cyclohexane: Shouldn’t there be 3 carbons in one plane and the remaining three in another plane?
Thanks again for all the help that I received from the content in this website :)
Yes, in the chair conformation there are three carbons in one plane and the remaining 3 in another plane. That is the case in the drawings above, although it might not be obvious from the perspective that is chosen.
I got so much help from this website studying Chemistry. Thank you so much
Glad to hear it HaEun. Thanks for the comment.
The last bit on puckering differences between ribo- and deoxyribonucleosides is fascinating. Here’s a nice computational study on the topic: http://pubs.acs.org/doi/abs/10.1021/jp9818683.
Thank you for your neat and helpful description of cyclohexane conformations:D
I learned a lot from your material. Thank you once again!
Glad you found it helpful Kyuwon!
thanks a lot, this site is awesome!
even i do agree that this site is too awesome that we can’t get our eye off it isn’t it alan walker o sorry alan kovacik