Elimination Reactions
Bulky Bases in Elimination Reactions
Last updated: May 28th, 2026 |
Elimination Reactions Using “Bulky Bases” – When The Zaitsev Product Is Minor
We’ve recently talked about Zaitsev’s rule in elimination reactions, and how the transition state leading to the more substituted alkene is lower in energy. This post covers reactions involving “bulky bases” where less of the Zaitsev product is obtained.
Table of Contents
- “Normal” E2 Reactions Follow Zaitsev’s Rule, Giving The “More Substituted” Alkene
- “Bulky Bases” Tend To Give A Higher Proportion Of “Non-Zaitsev” Products
- Bulky Bases Give More “Non-Zaitsev” Products Due To Steric Interactions With The Alkyl Halide
- Two Common Bulky Bases Are The t-Butoxide Ion And Lithium Di-Isopropyl Amide (LDA)
- Notes
- Quiz Yourself!
- (Advanced) References and Further Reading
1. “Normal” E2 Reactions Follow Zaitsev’s Rule, Giving The “More Substituted” Alkene
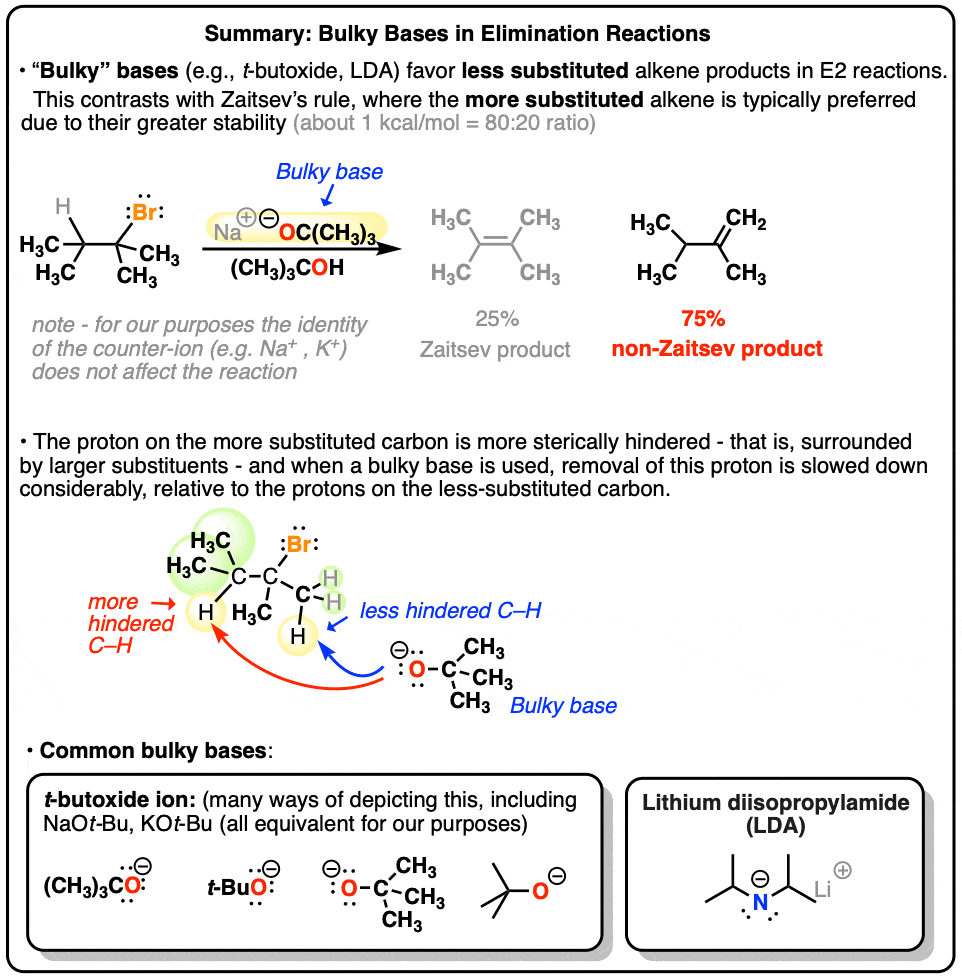
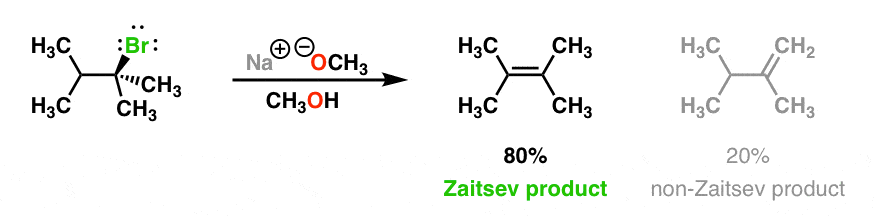
Most elimination reactions follow Zaitsev’s rule : you should expect that the “more substituted” alkene will be formed if at all possible. Like in the elimination reaction below, for instance, we get 80% of the tetrasubstituted alkene [“Zaitsev” – more substituted because there are 4 carbons attached to the alkene] and 20% of the disubstituted “non-Zaitsev” product.
The Zaitsev product generally forms because the more substituted alkene is generally more stable. (See article: Alkene Stability)
However, today we’ll talk about one interesting exception to this “rule” and how under certain conditions we actually end up with the “non-Zaitsev” alkene product instead.
2. “Bulky Bases” Tend To Give A Higher Proportion Of “Non-Zaitsev” Products
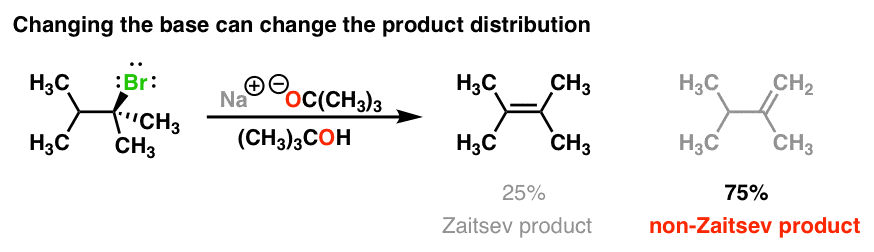
For instance, instead of using sodium methoxide, (NaOCH3) if you use the base NaOC(CH3)3 [or KOC(CH3)3, changing sodium for potassium doesn’t really matter here] you end up with an interesting reversal of products!
So what’s going on here? Why might we get less of the Zaitsev product here and more of the “non-Zaitsev” product?
3. Bulky Bases Give More “Non-Zaitsev” Products Due To Steric Interactions With The Alkyl Halide
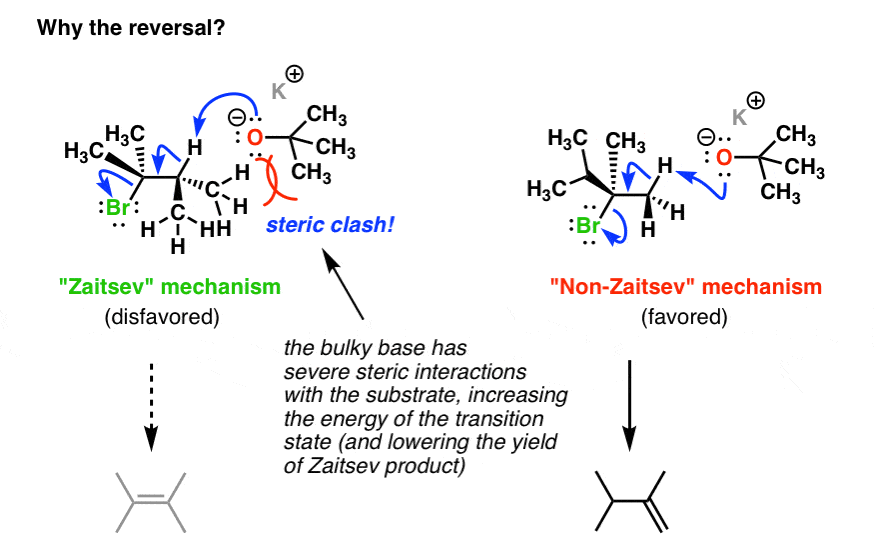
Well, if we draw out what the structure of the reactants might look like in their transition state, we can start to see why. [Note: this is not technically a transition state since we’re not drawing partial bonds, but you can at least see how the reactants are assembled].
The base in this instance – potassium t-butoxide – is an extremely bulky base, and the proton we remove to form the Zaitsev product is on a tertiary carbon. As the oxygen from the base draws nearer to this proton, a steric clash occurs.
In essence the electron clouds around the methyl groups are interacting with each other, and the repulsion between these clouds will raise the energy of the transition state [remember – opposite charges attract, like charges repel]. This will slow down the reaction.
Looking at the reactant assembly that produces the non-Zaitsev product, the bulky base is removing a proton from a primary carbon. Steric clash is considerably reduced in comparison to that for the Zaitsev product. Elimination is faster, and we therefore end up with the less substituted alkene as our major product.
This is one example of a reaction where the more thermodynamically stable product is not formed. [recall that alkene stability increases with the number of carbons directly attached to the alkene].
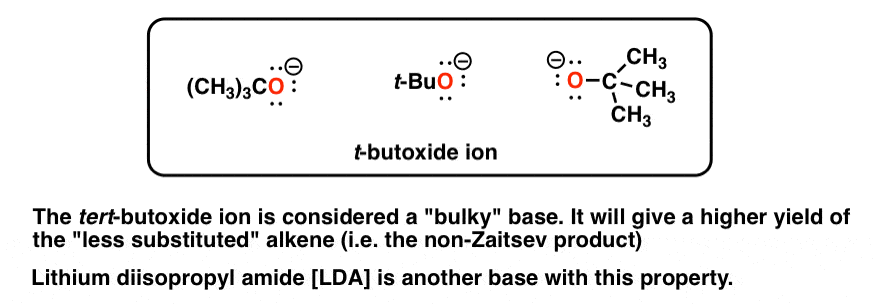
4. Two Common Bulky Bases Are The t-Butoxide Ion And Lithium Di-Isopropyl Amide (LDA)
So the bottom line for this post is that when performing an E2 reaction, using a bulky base will produce a greater proportion of non-Zaitsev alkene products relative to a less bulky base.
As far as we’ll see, the most common “bulky base” we need to consider is the t-butoxide ion, which can be drawn in many forms [see diagram]; occasionally you might see lithium di-isopropyl amide (LDA) used as well. For our purposes this completes the roster of bulky bases.
In the next post we’ll talk about an interesting observation we can make during certain E1 reactions.
Next Post: Comparing the E1 and SN1 Reactions
Notes
Quizzes
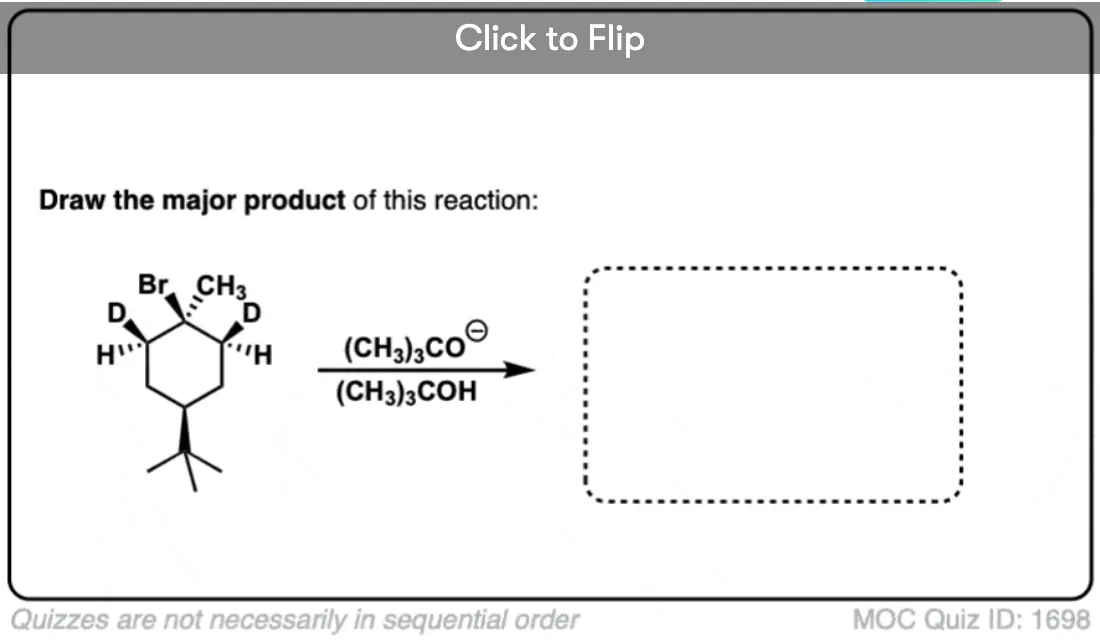
Become a MOC member to see the clickable quiz with answers on the back.
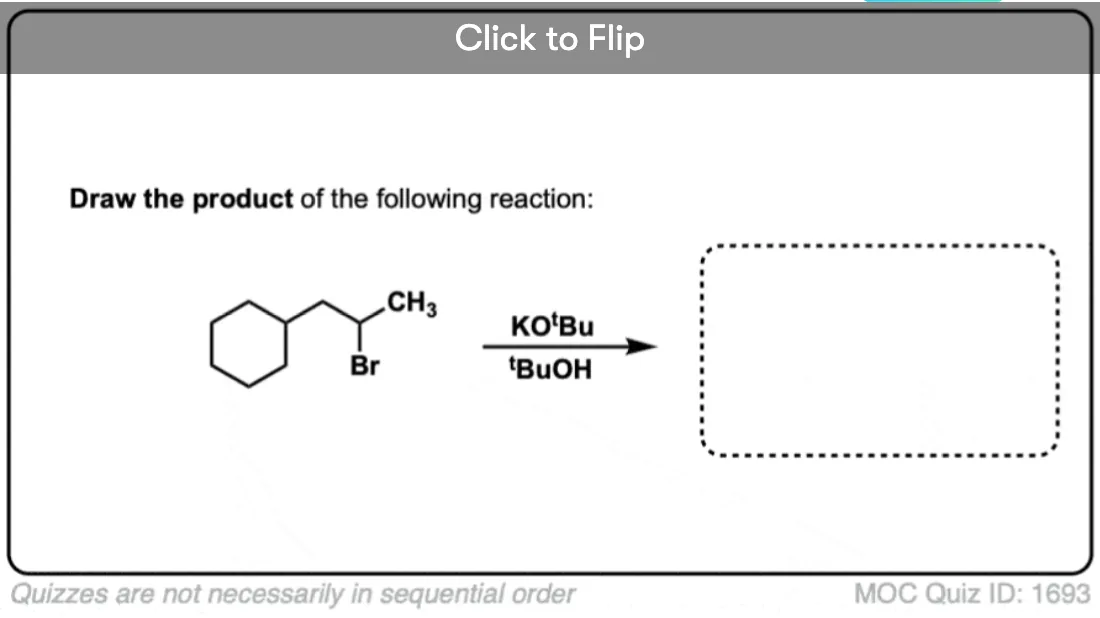
Become a MOC member to see the clickable quiz with answers on the back.
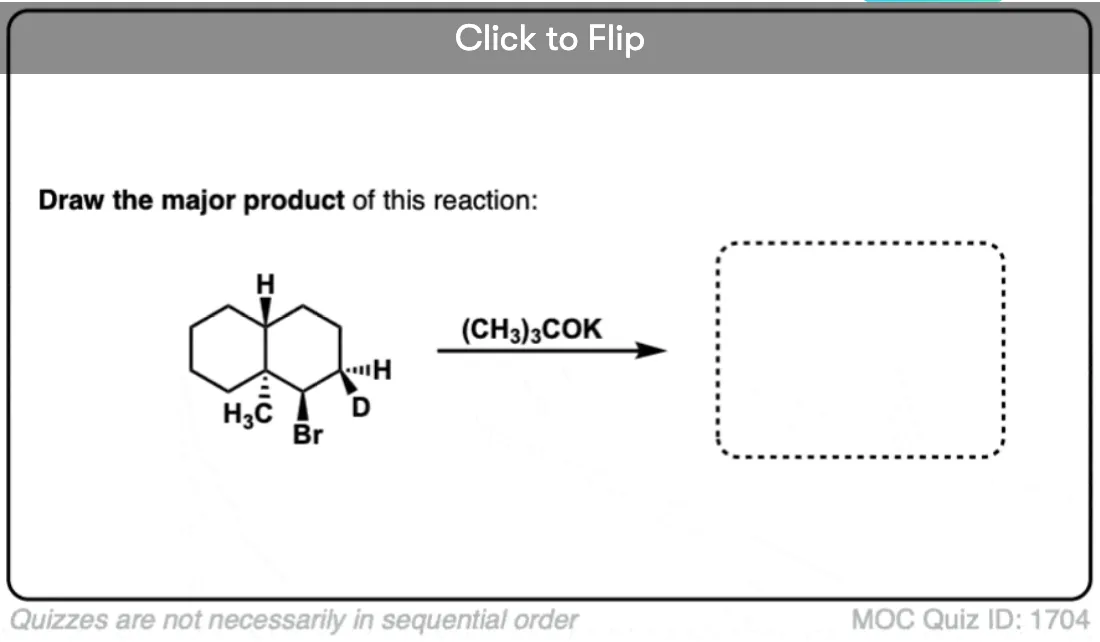
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
(Advanced) References and Further Reading
- For an example where a bulky leaving group can lead to “non-Zaitsev” (aka “Hofmann”) products, see this post on the Hofmann Elimination.
- Stereochemical and base species dichotomies in olefin-forming E2 eliminations
Richard A. Bartsch and Jiri Zavada
Chemical Reviews 1980 80 (6), 453-494
DOI: 10.1021/cr60328a001
A pretty comprehensive review containing many examples of Zaitsev and Hofmann-selective elimination reactions. - Mechanism of elimination reactions. Part X. Kinetics of olefin elimination from isopropyl, sec.-butyl, 2-n-amyl, and 3-n-amyl bromides in acidic and alkaline alcoholic media
M. L. Dhar, E. D. Hughes, and C. K. Ingold
J. Chem. Soc. 1948, 2058-2065
DOI: 10.1039/JR9480002058
Table I in this paper shows that solvolysis of 2-bromobutane with 1 M NaOEt in ethanol gives 82% yield of alkene at 25 °C, but similar solvolysis at 80 °C gives 91.4% yield of alkene. - Beiträge zur Kenntniss der flüchtigen organischen Basen
Aug. Wilk. von Hofmann
Just. Lieb. Ann. Chem. 1851, 78 (3), 253-286
DOI: 10.1002/jlac.18510780302
Early paper on Hofmann eliminations by its discoverer. Eliminations of quaternary ammonium salts favor loss of ethylene over larger groups. - —The nature of the alternating effect in carbon chains. Part XVIII. Mechanism of exhaustive methylation and its relation to anomalous hydrolysis
Walther Hanhart and Christopher Kelk Ingold
J. Chem. Soc. 1927, 997-1020
DOI: 10.1039/JR9270000997
Prof. Ingold mentions in this paper, “It follows from the basic hypothesis that the ease of removal of the b-proton (reaction A) depends (a) on its vulnerability, (b) on the proton-avidity of the attacking anion” - Steric Effects in Elimination Reactions. VII. The Effect of the Steric Requirements of Alkoxide Bases on the Direction of Bimolecular Elimination
Herbert C. Brown, Ichiro Moritani, and Y. Okamoto
Journal of the American Chemical Society 1956, 78 (10), 2193-2197
DOI: 1021/ja01591a047
As Nobel Laureate Prof. H. C. Brown (Purdue) states in this paper, “[…] with increasing basic strength of the alkoxide bases (C2H5O– < (CH3)3– < CO–) there is observed not a decrease, but rather an increase in the selectivity of the reagent. It may be concluded therefore that the increase in base strength does not play any major role in altering the isomer distribution in the present reaction.”
Check out Table IV. For 2-bromobutane, 1.0 M KOtBu gives a 53:47 ratio of 1-butene to 2-butene. For 2-bromopentene, it’s 66:34 for 1-pentene vs 2-pentene. The ratio of “anti-Zaitsev” alkenes gets higher when the bromide is tertiary; for 2-bromo-2-methylbutane and 1.0 M KOtBu the ratio is 72:28 for the “anti-Zaitsev”. [30:70 when the base is KOEt].
If you’re really interested see this chart (thanks, Ben!) Olefin Product Distribution - Steric Effects in Elimination Reactions. IX. The Effect of the Steric Requirements of the Leaving Group on the Direction of Bimolecular Elimination in 2-Pentyl Derivatives
Herbert C. Brown and Owen H. Wheeler
Journal of the American Chemical Society 1956, 78 (10), 2199-2202
DOI: 1021/ja01591a049
In this paper, Prof. Brown shows that, all things being equal, bulkier leaving groups also lead to formation of less substituted olefins. 2-bromopentane gives 31% yield of 1-pentene upon solvolysis with KOEt, but 2-(trimethylammonium)-pentane gives 98% of yield of 1-pentene. - Steric Effects in Elimination Reactions. X. Steric Strains as a Factor in Controlling the Direction of Bimolecular Eliminations. The Hofmann Rule as a Manifestation of Steric Strain
Herbert C. Brown and I. Moritani
Journal of the American Chemical Society 1956, 78 (10), 2203-2210
DOI: 1021/ja01591a050
This is Prof. Brown’s paper wrapping up the topic of steric effects in E2 eliminations. Prof. Brown used various pyridine bases varying in steric hindrance around the nitrogen (pyridine, 2-methylpyridine (2-picoline), and 2,6-dimethylpyridine (2,6-lutidine). Increasing the steric bulk of the base does increase the yield of the less substituted olefin. - Mechanism of elimination reactions. Part XIX. Kinetics and steric course of elimination from isomeric menthyl chlorides
E. D. Hughes, C. K. Ingold, and J. B. Rose
J. Chem. Soc. 1953, 3839-3845
DOI: 10.1039/JR9530003839
This is an example of the Zaitsev rule in a cyclohexane system. Neomenthyl chloride gives 78% 3-menthene and 22% 2-menthene with EtO– in ethanol. - Eliminations in Cyclic cis‐trans‐Isomers
Dr. W. Hückel and Priv.‐Doz. Dr. M. Hanack
Angew. Chem. Int. Ed. 1967, 6 (6), 534-544
DOI: 10.1002/anie.196705341
Study on E1 and E2 eliminations in cyclic systems. E2 eliminations will give the “Hofmann” product if anti-arrangement of H and leaving group is not possible, whereas E1 will give Zaitsev regardless.
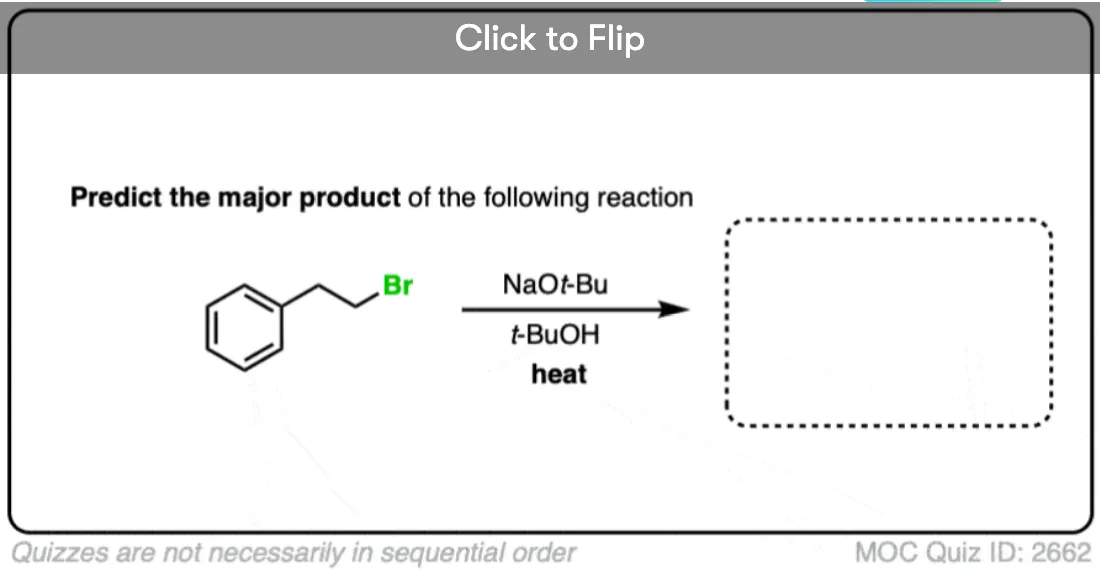
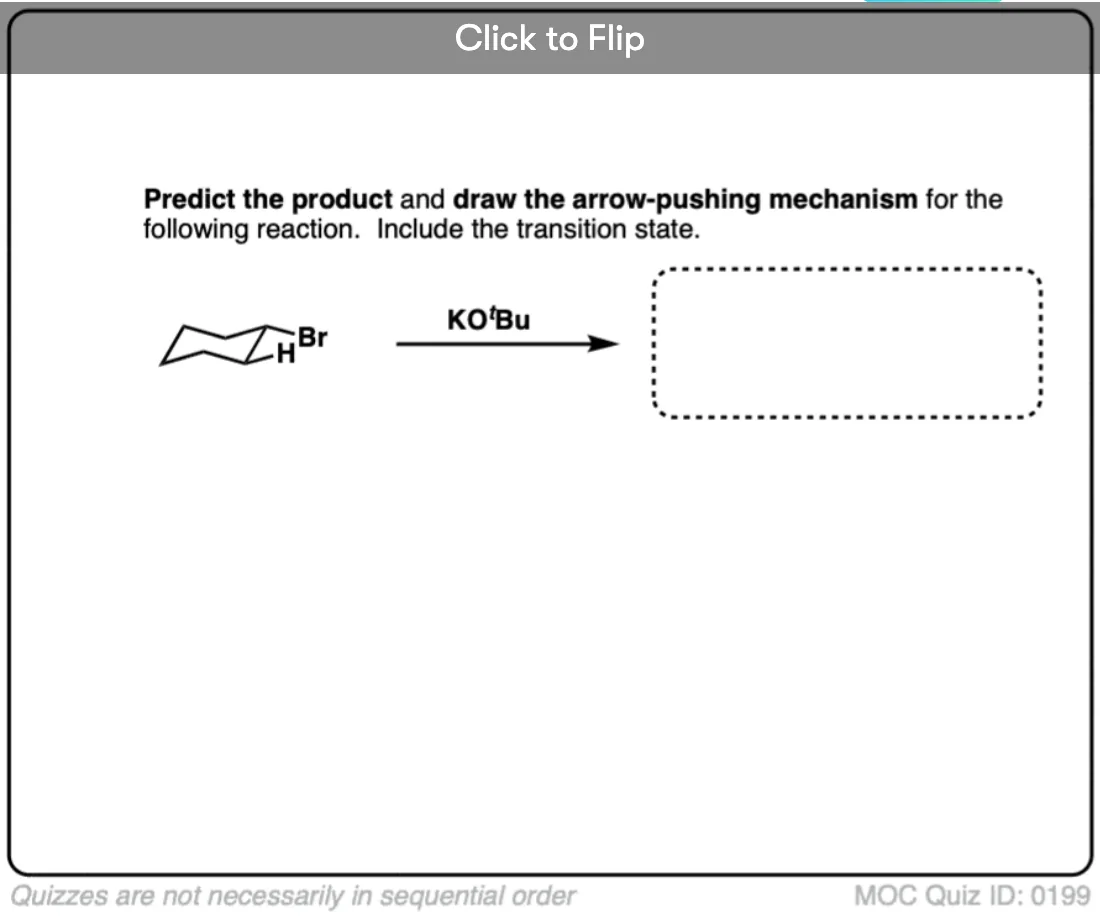
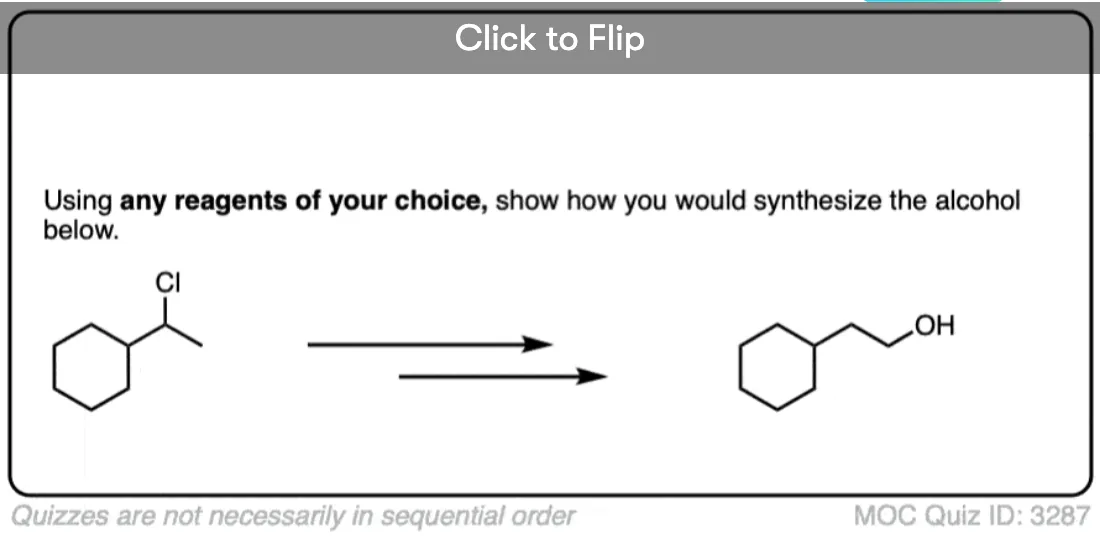
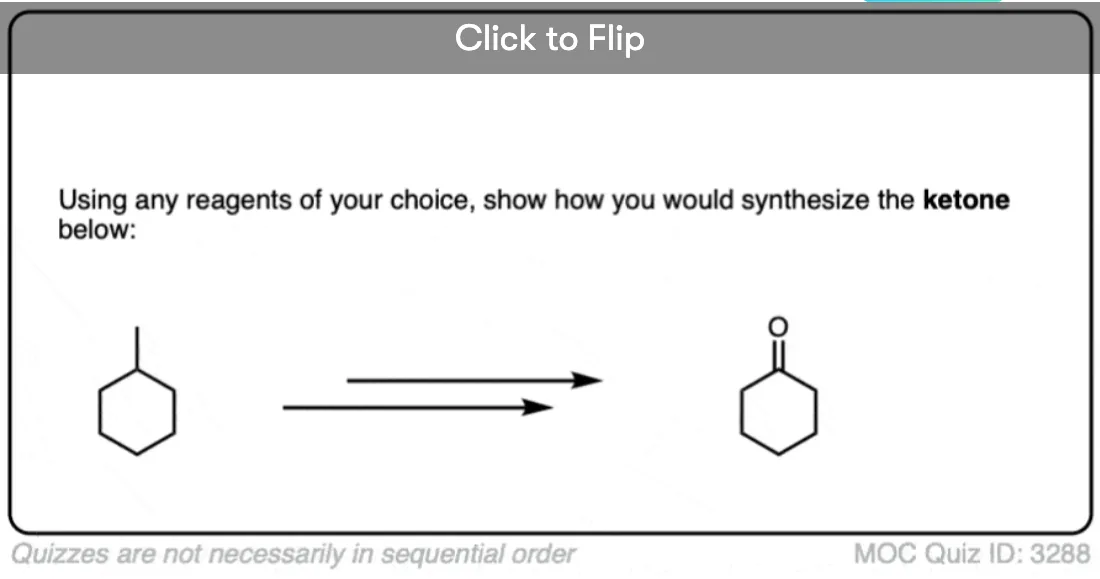
2 bromo butane on reaction with tert butoxide give trans 2 butene as major product?
No, 2-butene is the minor product.
hi, I have a question, in E1 reactions, the transition state leading to the more substituted alkene isn’t a part of the rate determining step right , so what drives formation of zaitsev product as major product in E1 reactions?
Does DBU and DBA considered bulky base?
DBU, yes. Not sure what you are referring to with DBA.
These articles, infact this whole website has been invaluable to my studies. I can’t thank you enough!
Glad to hear it Matthew – Please let me know if there’s anything missing that you’d like to see. – James
NaOH is not bulky base,is it?
No, it most certainly is not.
Hello,
I just recently did a test where we had an elimination reaction with t-butoxide, and the teacher marked me wrong saying it was Zaitsev’s rule. However, when I went to explain to her that bulky bases give Hofmann elimination products, she told me she needed some academic proof. Are there any textbooks that refer directly t-butoxide as a strong base which yields Hofmann as a major ? I cannot seem to find anything online. Thanks
Yes, I would refer any questioners to this H.C. Brown paper, particularly table 4: Brown, H. C.; Moritani, I.; Okamoto, Y. J. Am. Chem. Soc. 1956, 78 (10), 2193–2197.
DOI: 10.1021/ja01591a047
I believe that the student could be asking the questions, such as what would happen in the situation that a big bulky base was used in an E2 reaction. One thing I learnt most of all being a student, was to not totally rely on the corriculim and instead think more for myself. I’m not wanting to appear being a smarta$$ by making such a comment, other than my grades improved when I took more responsibility. I love what is being shown here, and for an undergraduate that had to pull out due to health issues, this resource is the bomb.
Thankyou
Thanks Brooster – I appreciate your comment and your kind words.
This is a neat example. Since the non-Zaitsev product appears in a 3:1 ratio but could have arisen from any of six beta proton removals compared to only one on the tertiary carbon, isn’t the path to the Zaitsev product still mechanistically favored?
That’s very similar to my own question — in the case of e.g. 1-bromo-1-methylcyclohexane doing an E2 with KOtBu, is there still enough steric bulk to favor the less-substituted methylenecyclohexane product over the more-substituted 1-methylcyclohexene?
Thanks!
Dear James:
I am studying for MCAT. and things are very confusing to me regarding this subject. Organic Chemistry by Smith does not recognize hofmann product at all and gives zaitsev product with tertiary butoxide even with tertiary. Organic Chemistry by Klein and Solomon give hofmann product with both tertiary and secondary alkyl halides (but no example of what happens with primary), and Organic chemistry by Bruice explicitly says tertiary butoxide with only tertiary gives hofmann but says secondary gives still zatisev. what am I supposed to do on a national exam when I don’t know which reference is used? what is the product with primary and secondary alkyl halide with tertiary butoxide?
Thanks
I don’t know what to tell you other than this is another issue which is a rat’s nest. It’s not taught consistently.
That’s an easy question to answer.
#1 rule is that the MCAT writers are ALWAYS RIGHT. So on a passage they will tell you what you need to know for the ochem passages and you use that info along with your background knowledge. The MCAT would never in a million years force you to rely on varying types of information for controversial topics. They will tell you what you think on the test for those types of questions.
Do you have a primary literature reference for this? I sometimes clash (see what I did there?) with my colleagues who don’t teach the t-butoxide non-Zaitsev elimination. I’d love to prove them wrong once and for all :)
I am stealing my data from Reusch’s site. My version of March talks about leaving groups and base strengths re: Zaitsev/ non-Zaitsev, but does not mention t-butoxide. It’s unevenly taught across the country, I can tell you that.
Someday when I have my own chemistry lab I am going to go through all these substitution and elimination reactions and churn out real data so I don’t have to feel like I’m talking ex recto all the time.
Gotcha. That’s where I always wind up, too. It’s frustrating for me, because students change professors at semester break, so I can’t very well hold new students responsible for a reaction their fall semester prof didn’t teach them… so I can’t really hold anyone in the class responsible for it 2nd semester… so why teach it at all in 1st semester?