Bonding, Structure, and Resonance
Evaluating Resonance Structures: The Fewer Charges, The Better
Last updated: May 29th, 2026 |
Evaluating Resonance Forms – The Rule Of Least Charges
- So far we’ve introduced resonance (See article – Introduction to Resonance) and introduced the curved arrow formalism (See article – Curved Arrows For Resonance) to show the movement of electrons.
- Importantly, we’ve talked about how the “true” picture of a molecule is a hybrid of its resonance forms (and not an equilibrium between forms).
- Here, we’re going to start evaluating the relative importance of various resonance forms and the contribution they make to the overall resonance hybrid.
- The first rule, we’ll discuss here, is that the resonance form with the least number of point charges will be the most important resonance form, and make the greatest contribution to the resonance hybrid.

Table of Contents
- Recap: The Three “Legal Moves” Of Resonance Curved Arrows
- The First Rule Of Evaluating Resonance Structures: Minimize Charges
- Evaluating The Resonance Structures Of Ethene (“Ethylene”)
- Evaluating The Resonance Structures Of The Allyl Cation
- Evaluating The Resonance Structures Of The Allyl Anion
- Notes
- Quiz Yourself!
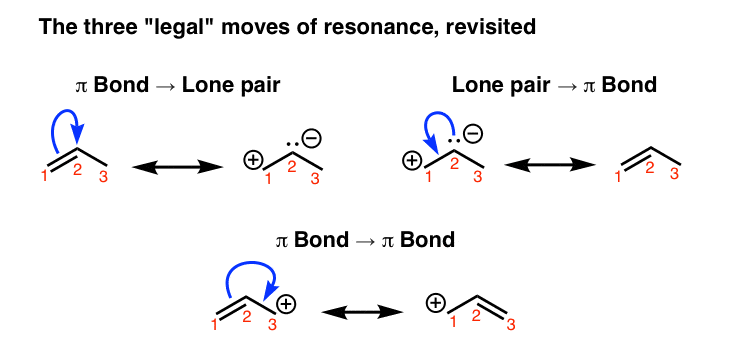
1. Recap: The Three “Legal Moves” Of Resonance Curved Arrows
Here’s a recap of the different “moves” we can perform on a molecule to get different resonance forms. Every resonance form we can draw for a molecule can be made through a combination of these three moves.
We can apply these “moves” so long as we don’t break the octet rule. The thing is that if we start looking at even simple molecules, we can soon get lost in a maze of potential resonance forms if we apply the arrow-pushing rules willy-nilly. So let’s cut to the main problem: how do we evaluate the importance of different resonance forms?
In other words, which make a greater contribution to the overall resonance hybrid (more significant) and which make less of a contribution to the overall hybrid (less significant)?
2. The First Rule Of Evaluating Resonance Forms: Minimize Charges
Here’s the punch line for today:
Let’s look at three examples. Really simple ones, but they’ll do.
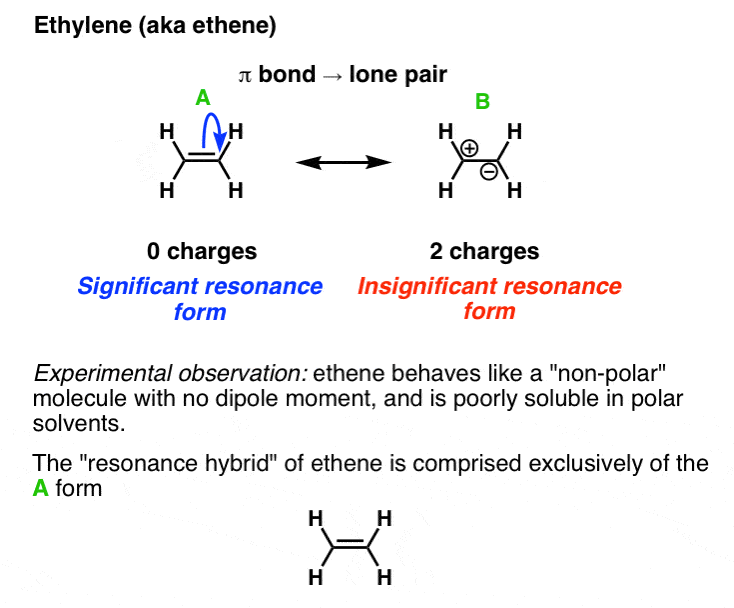
3. Evaluating The Resonance Forms Of Ethene (“Ethylene”)
The simplest molecule with a π bond is ethene. If we draw a resonance structure for it, we can move the π bond to the lone pair of one of the end carbons (doesn’t matter which one) to give a carbocation and a lone pair. This is a “legal” resonance form, since we’re not breaking the octet rule.
The question is, how significant is the resonance form with the two charges? The answer from experiment is: not very much because ethene behaves essentially like how we’d expect a neutral, uncharged molecule to behave – it has a low boiling point and doesn’t mix with water. [Note 1]
(One thing to note – the net charge of each resonance structure is the same here (i.e. they both have a net charge of zero) since on the resonance form on the right, the opposite charges cancel).
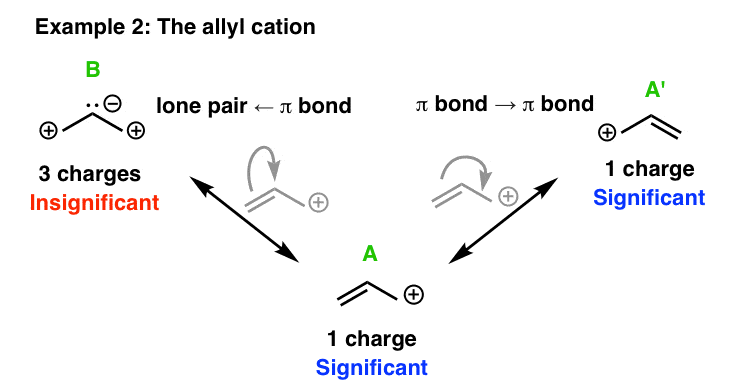
4. Evaluating The Resonance Forms Of The Allyl Cation
Let’s now look at the resonance structures of the allyl carbocation. The net charge on the allyl carbocation is +1.
When we apply the relevant arrow-pushing “moves” to it, we get something like this, with 4 resonance structures – A, A’ , B, and C. Resonance structures A and A’ both have one charge. Resonance structures B and C have three charges. [Although again, note that B and C each have a net charge of +1 when you cancel the opposite charges].
So what does experiment tell us? Our best experimental evidence for the structure of the allyl carbocation tells us that the positive charge is distributed equally between the two end carbons, with a bond length intermediate between that of a C–C single bond (1.54 Å) and a C–C π bond (1.34 Å).
That is to say, the resonance hybrid of fhe allyl carbocation is a 1:1 mixture of resonance forms A and A’.
Resonance forms B and C don’t contribute to the resonance hybrid to any significant extent.
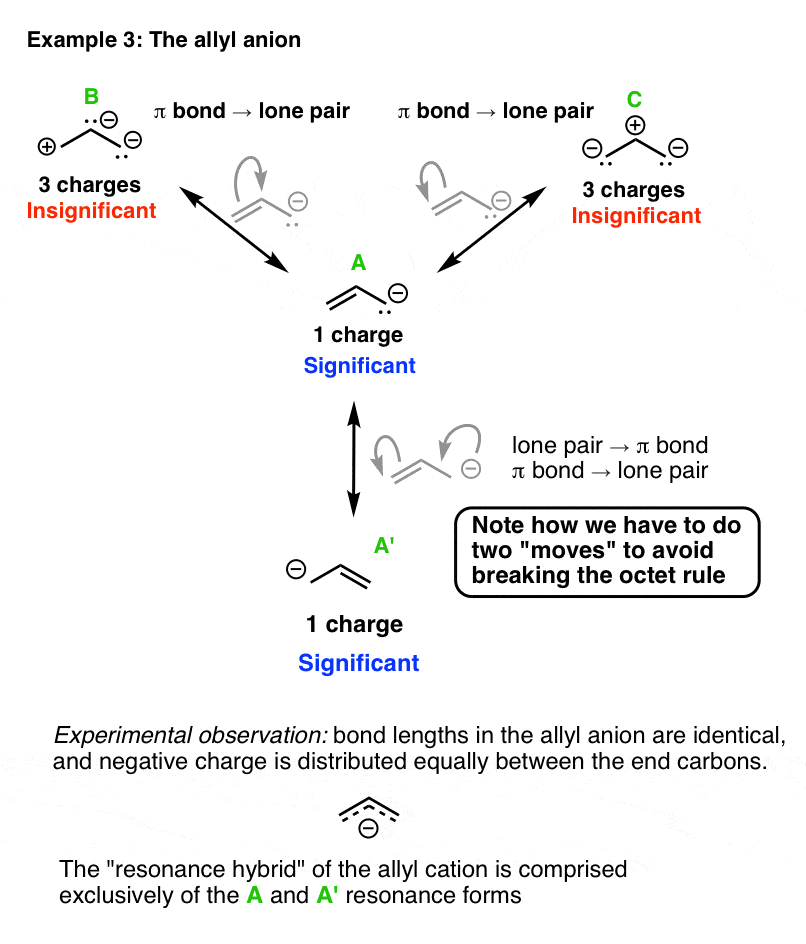
5. Evaluating The Resonance Forms Of The Allyl Anion
Finally, let’s look at the allyl anion. We can use a similar application of the arrow pushing rules to get the different resonance forms, although here’s a twist.
Note that we can’t move the end lone pair to form a new π bond without breaking the octet rule! So in this case we actually have to draw two arrows to make it legal. This gets us to the equivalent resonance form, A’.
On the other hand the other resonance structures (B and C) can be obtained through a fairly straightforward process (move a π bond to form a lone pair on either carbon.)
Again, however, these resonance forms don’t contribute much to the overall resonance hybrid. Experiment tells us that the electron density on the allyl anion is on the ends, and the bond lengths are again intermediate between a C–C single bond and a C–C π bond.
The best interpretation of this data is that the allyl carbanion is a 1:1 mixture of the resonance forms A and A’.
So again, the bottom line is that the most stable (and significant) resonance structures will be those with the fewest charges.
You’ve probably noticed however that these examples are pretty simple – we’re just dealing with carbon atoms. What happens when we deal with unsymmetrical resonance structures? We’ll need to introduce a new principle for that!
Next Post: Evaluating Resonance Forms (2): Applying Electronegativity
Notes
Note 1. If you measure the electronic and structural properties of ethene, it’s clear that the carbon-carbon bond length is shorter than that of a normal carbon-carbon single bond, implying a strong interaction between the two atoms.
Furthermore, if the resonance structure on the right were significant, we’d expect ethene to have a relatively high boiling point (since it’s charged) and be quite soluble in polar solvents like water. In fact, ethene boils at a very low temperature (–88 °C) and is practically insoluble in polar solvents. So it’s safe to conclude that the resonance form on the right is quite insignificant.
Quiz Yourself!
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.

In the allyl anion resonance form C, we too have equal bond lengths and even distribution of negative charge over the end carbon atoms aw in the hybrid of forms A and A’. So there must be other experimental observations based on which this form is rejected as insignificant.
Yes, this is insignificant because it does not show aa pi bond between any of the carbons. The bond length for a C-C single bond is about 1.50 angstrom, whereas that for a C-C double bond is about 1.40 angstrom. The meaasured bond length is more consistent with the presence of a C-C pi bond.
Thanks for a great post (and website).
I wonder about ranking resonance contributors stability.
You treat the rule of least charges as superior.
However, many textbooks state that the demand of full octet prevails.
For example, see ‘Organic chemistry’ by Bruice or here:
http://www.chem.ucla.edu/~harding/tutorials/resonance/imp_res_str_key.html
Could you please refer me to any relevant text (article?) which resolves the issue?
For practical purposes, full octets is implied in the rule of “least charges” , simply because the only commonly encountered atom which will ever have less than a full octet is positively charged carbon.
For example two, I created 5 resonance structures. I believe I’m wrong though because my 5th resonance structure had a lone pair of electrons on the 3rd carbon in allyl cation would imply a resonance structure of A’?
Hi James! I’ve really been enjoying this series (even though I’m working on one very similar — just don’t have the time!)
Your ethylene example confuses me a bit… wouldn’t there also be a possible B’ resonance form where the pi bond electrons move to the left carbon as a lone pair? And if so, wouldn’t these two resonance forms (insignificant though they may be) essentially “cancel” each other out, so we wouldn’t expect there to be a measurable dipole anyway?
Thanks!
thanks Jess… you’re right re: the dipole. there’s probably a better way to word this. Will edit!
I noticed the new curved arrows :) Lookin’ good, Billy Ray!
MANY thanks, I’m an idiot for not figuring out how to do this earlier. Very happy with the result.
nonono, you’re supposed to say ‘Feelin’ good, Lewis’
:)