Elimination Reactions
Antiperiplanar Relationships: The E2 Reaction and Cyclohexane Rings
Last updated: May 28th, 2026 |
Antiperiplanar Relationships Between C-H And The Leaving Group: The E2 Reaction and Cyclohexane Rings
Here we come to a very testable application of the E2 reaction – how to draw the products of E2 reactions in cyclohexane rings!
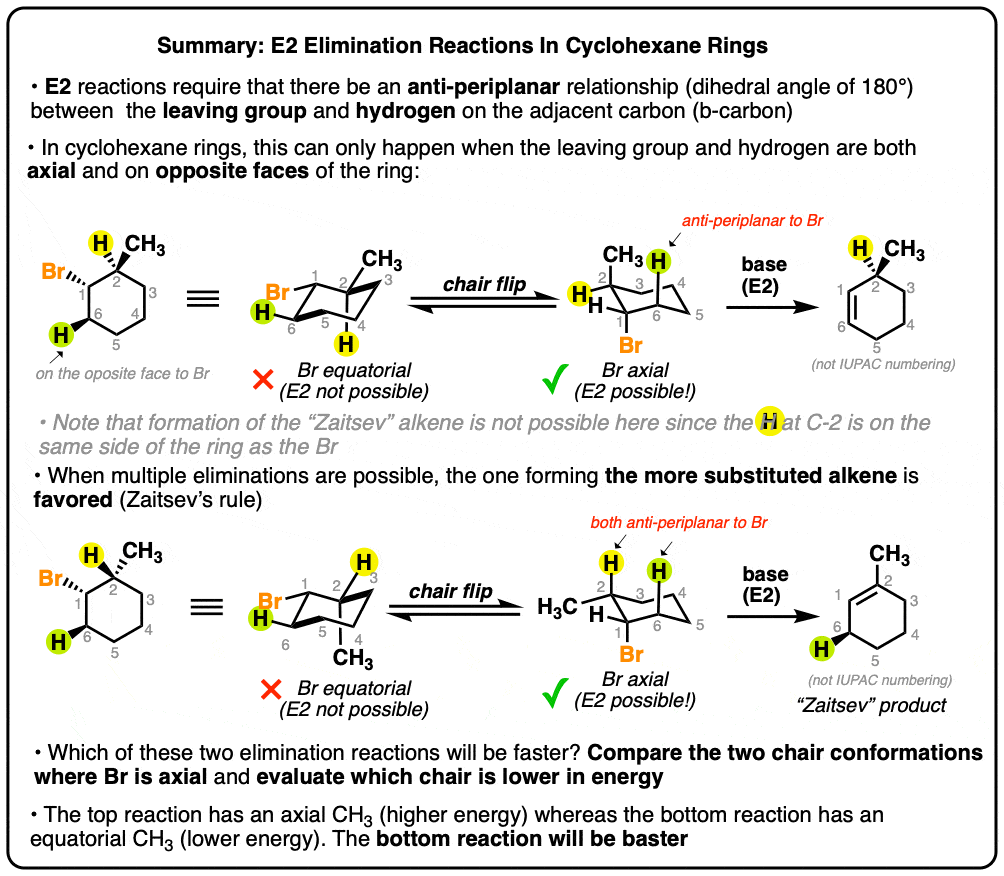
Table of Contents
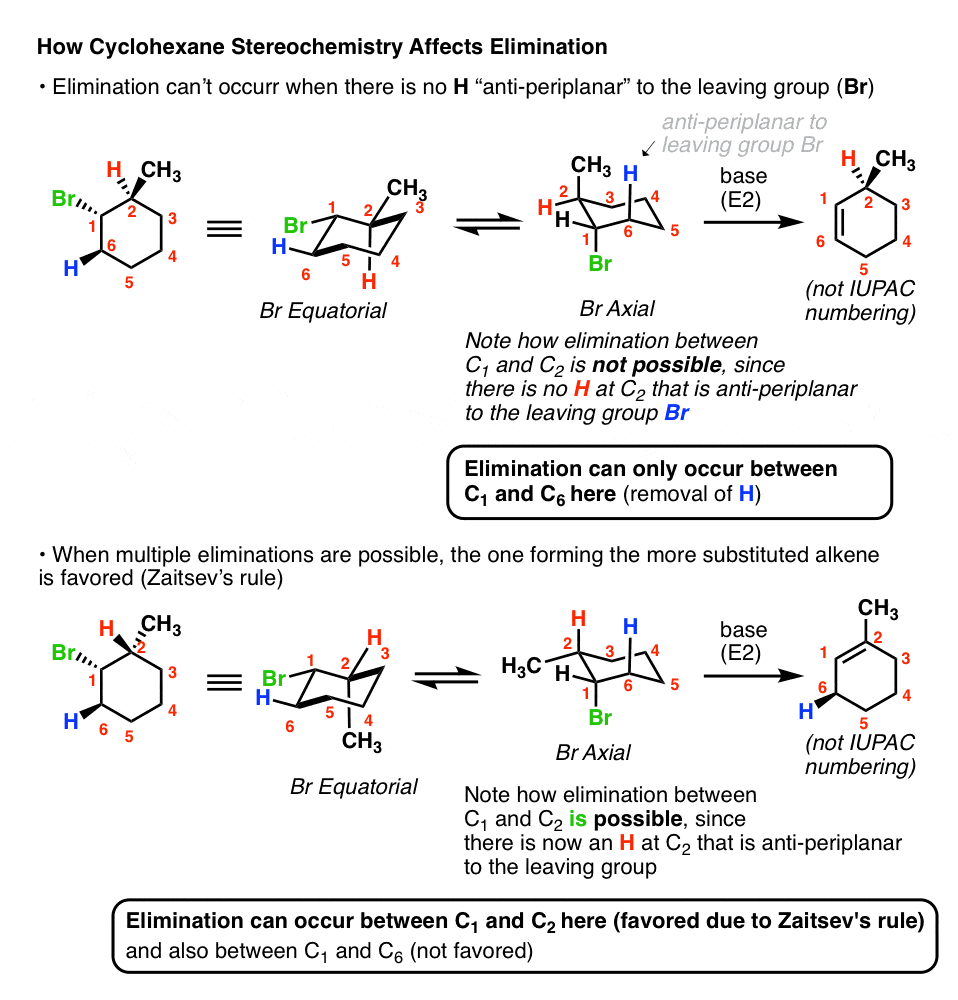
- In The E2 Reaction, The Leaving Group Is Always “Anti-Periplanar” To The Hydrogen That Is Removed On The Adjacent Carbon (“Beta-Carbon”)
- In Cyclohexane Rings, E2 Reactions Can Only Occur When The Leaving Group Is Axial
- In E2 Reactions Of Cyclohexane Rings, The Only Way The Leaving Group And C-H Bond Can Be Anti-Periplanar Is If They Are On Opposite Faces Of The Ring
- Some Examples: What Would Be The Major E2 Product In Each Case?
- Cyclohexane Substituents Can Affect The Rate Of E2 Elimination Reactions In Cyclohexane Rings
- The Higher The Concentration Of The Conformer With An Axial Leaving Group, The Faster The Elimination Will Be
- Notes
- Quiz Yourself!
- (Advanced) References and Further Reading
1. In The E2 Reaction, The Leaving Group Is Always “Anti-Periplanar” To The Hydrogen That Is Removed On The Adjacent Carbon (i.e. the “Beta-Carbon”)
Last time we compared the E1 and E2 reactions and mentioned one of the key differences was the stereochemistry of the E2 reaction. Remember that in the E2, the leaving group is always “anti” to the hydrogen that is removed on the adjacent carbon. [That means that they’re directly opposed to each other, or 180°; kind of like the minute hand and the hour hand when a clock reads 6:00].
This is an extremely important detail to be able to apply in reactions.
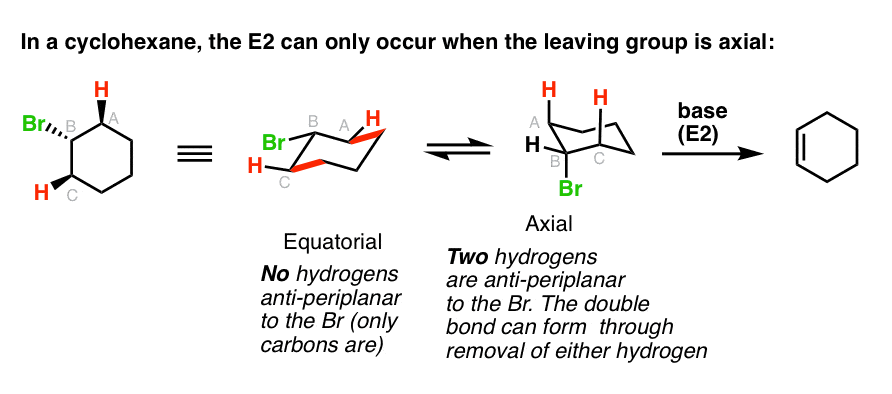
2. In Cyclohexane Rings, E2 Reactions Only Occur When The Leaving Group Is Axial
One way this often comes up is in discussions of cyclohexane rings. If you’ll recall, in the cyclohexane chair conformation, groups can either be axial (pointing straight up or down) or equatorial (pointing “somewhat up” or “somewhat down”).
In order for a hydrogen to be “anti” to a leaving group, it’s required that both groups be axial. Look closely at the cyclohexane ring on the left, where the leaving group is equatorial – see how the group that is “anti” is the C-C bond [highlighed in red]?
That E2 is never gonna work!
So if you draw the leaving group equatorial in a cyclohexane chair, you’ll have to do a chair flip so that the leaving group is axial. That’s shown in the right hand example, where an E2 can actually happen.
3. In E2 Reactions Of Cyclohexane Rings, The Only Way The Leaving Group And C-H Bond Can Be Anti-Periplanar Is If They Are On Opposite Faces Of The Ring
This brings us to the second point. If the leaving group is, let’s say, on the “top” face of the cyclohexane, you can only form an alkene to adjacent carbons where the hydrogen is on the opposite face.
You might remember the example from last time where we couldn’t form the “Zaitsev” alkene because the Br was a wedge and there was an alkyl group on the carbon next door that was on the opposite face. In this case we can only form the less substituted alkene. If the methyl group is switched, however, then the E2 to give the Zaitsev product becomes possible:
The bottom line here [and trust me, this comes up in tests, a lot!] is that you always want to pay attention to what side of the ring your leaving group is on, and make sure that the E2 you draw is indeed possible.
4. Some Examples: What Would Be The Major E2 Product In Each Case?
Here are some more examples to think about. What would be the major E2 product in each case?
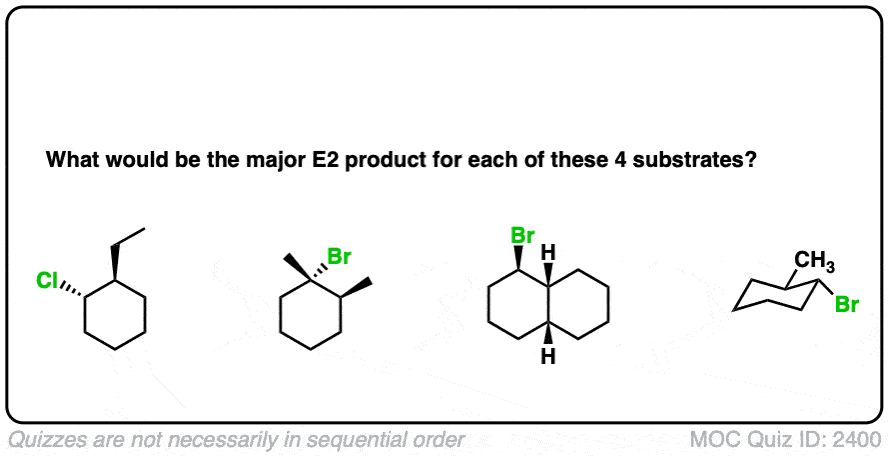 Click to Flip
Click to Flip
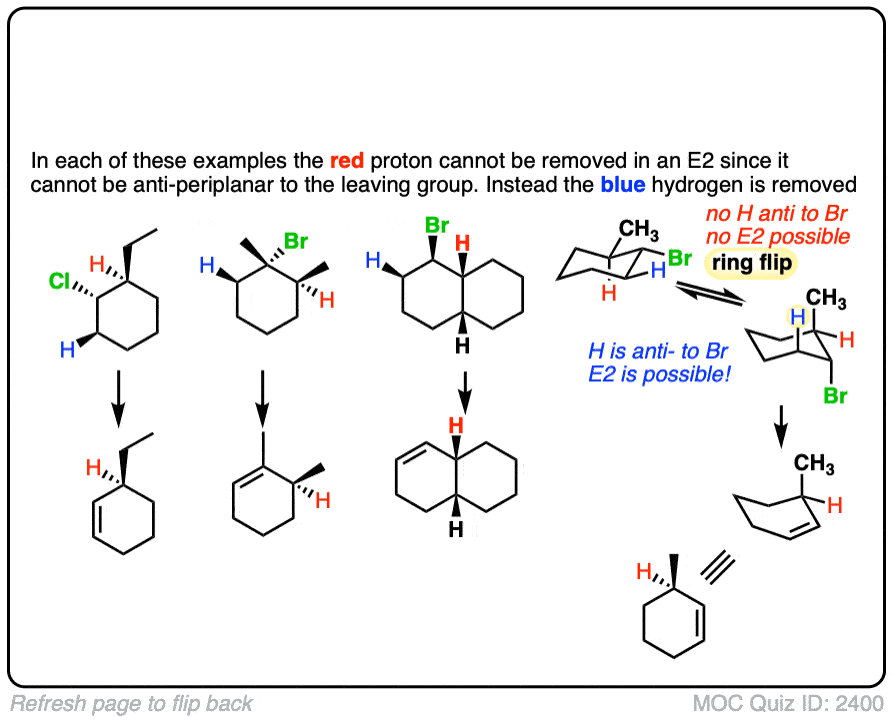
Now, let’s talk about a very interesting application of what we just discussed. This is a little more advanced, but see if you can follow it through. It ties together what we’ve discussed about the E2 with what you’ve previously learned about cyclohexane chair flips.
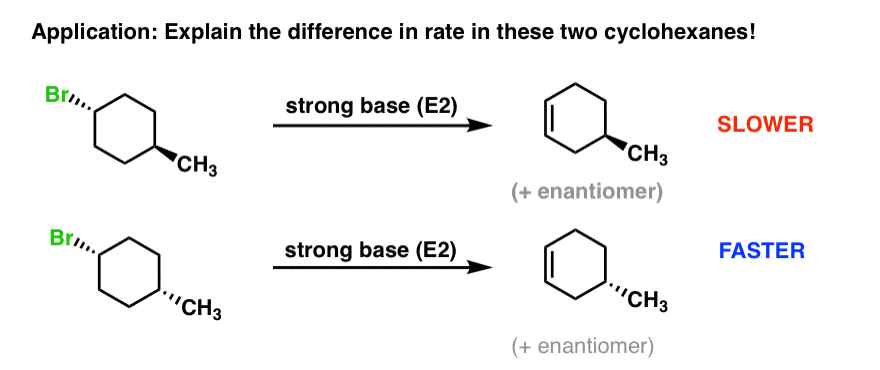
5. Cyclohexane Substituents Can Affect The Rate Of E2 Elimination Reactions In Cyclohexane Rings
Imagine you’ve got two alkyl halides, and they’ve got slightly different structures. We make the following observation: E2 with the second starting material is significantly faster than E2 with the first product. Question: why might this be?
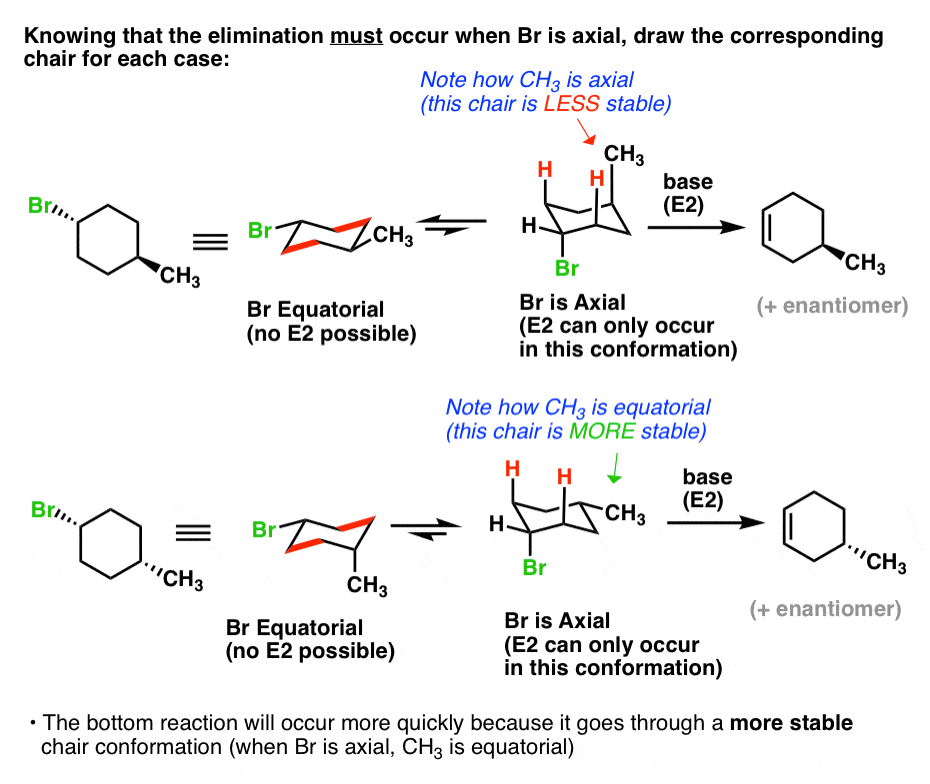
In order to understand what’s going on, it would help to draw the cyclohexane chair forms of both of these molecules. So let’s do that and then have a closer look.
What’s going on? Each molecule will have an equilibrium between two chair forms.
In the top molecule, the left-hand conformation is favored, because the bulky methyl group [CH3 is actually bulkier than Br, with an A-value of 1.75 vs. 0.4 – see A-Values] is equatorial. So equilibrium will favor the left hand molecule.
In the bottom molecule, the rightmost conformation is favored, because the bulky methyl group is equatorial. So equilibrium will favor the right-hand molecule.
6. The Higher The Concentration Of The Conformer With An Axial Leaving Group, The Faster The Elimination Will Be
Notice something interesting? Remember that in order for E2 to occur, the leaving group must be axial.
So there’s only one conformation where this will be possible for each ring. However, in the top example, Br is axial only in the least stable conformation, whereas in the bottom example, Br is axial in the most stable conformation. Since the bottom example will have a higher concentration where Br is axial, it will be faster.
Isn’t it interesting how it all ties together? Concepts you learn in one chapter can come back and be applied in later chapters!
In the next post we’ll talk about another example where Zaitsev’s rule doesn’t apply.
Next Post: Bulky Bases in Elimination Reactions
Notes
Quiz Yourself!
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
(Advanced) References and Further Reading
- Abspaltungsreaktionen und ihr sterischer Verlauf
Walter Hückel, Werner Tappe, Günter Legutke
Lieb. Ann. Chem. 1940, 543 (1), 191-230
DOI: 10.1002/jlac.19405430117
An early study of E2 stereochemistry on menthyl derivatives. - Mechanism of elimination reactions. Part XVIII. Kinetics and steric course of elimination from isomeric benzene hexachlorides
E. D. Hughes, C. K. Ingold, and R. Pasternak
J. Chem. Soc. 1953, 3832-3839
DOI: 10.1039/JR9530003832
The name of this paper is a little confusing since ‘benzene hexachloride’ does not refer to hexachlorobenzene, but 1,2,3,4,5,6–hexachlorocyclohexane. Hexachlorocyclohexane has a few isomers depending on the orientation of the Cl atoms, as explained in the paper. Depending on the orientation of the Cl atoms in the isomer, different products are obtained, based on whether an E2 pathway is possible at those position(s). - Mechanisms of Elimination Reactions. X. Deuterium Exchange in Base-Promoted Dehydrochlorination of β-Benzene Hexachloride
Stanley J. Cristol and Delbert D. Fix
Journal of the American Chemical Society 1953, 75 (11), 2647-2648
DOI: 1021/ja01107a027
Classic study of hexachlorocyclohexane isomers – all chlorine atoms on adjacent carbon atoms are trans to each other, so it is impossible to bring adjacent hydrogen and chlorine into an anti relationship without causing ring strain. - Mechanism of elimination reactions. Part XIX. Kinetics and steric course of elimination from isomeric menthyl chlorides
E. D. Hughes, C. K. Ingold, and J. B. Rose
J. Chem. Soc. 1953, 3839-3845
DOI: 10.1039/JR9530003839
This paper illustrates Zaitsev’s rule in a cyclohexane system. Neomenthyl chloride gives 78% 3-menthene and 22% 2-menthene with EtO– in ethanol . - Mechanism of elimination reactions. Part XXII. Anomalous elimination from the trimethylneomenthylammonium ion
E. D. Hughes and J. Wilby
J. Chem. Soc. 1960, 4094-4101
DOI: 10.1039/JR9600004094
This paper perfectly illustrated the concepts explained in this section. Menthylammonium ion cannot undergo elimination to give the 3-olefin, since the anti position on C4 is occupied by the isopropyl group. Only the 2-olefin is obtained from elimination with menthyl-LG (LG = leaving group, chloride, trimethylamine, etc.). In neomenthyl-LG, the isopropyl group is not anti, and so the 3-olefin is accessible. However, the yield of the 3-olefin decreases from 88% to 65% when EtO– is used instead of OH–, and it would be likely to decrease further when using bulkier bases (e.g. tBuO–). - Neighboring Carbon and Hydrogen. XIX. t-Butylcyclohexyl Derivatives. Quantitative Conformational Analysis
S. Winstein and N. J. Holness
Journal of the American Chemical Society 1955, 77 (21), 5562-5578
DOI: 10.1021/ja01626a037
This is the classic paper by Prof. Winstein introducing the concept of A-values. There is a brief mention of the effect of axial/equatorial orientation of the leaving group on reactivity. When the leaving group is axial, an E2 pathway is favored, but when the leaving group is equatorial, a mix of E1 and SN1 takes place.






Hey!Got stuck in a point,you say that the Br needs to be axial in order to go through E2,but crap in the 4th question of your quiz the methyl cyclohexane without any axial Br does goes through E2.I understand that it works out because of being anti-periplanar,but please,I think it will be best to be less specific about the Br being axial in some parts of the Reply is highly appreciated.
Thanks for this. Sorry for the absurdly late reply, I fixed quiz 2400 so that it shows a ring flip before the elimination reaction. Hopefully it’s more clear now!
For an axial CH3 and Br group in cyclohexane ring, the E2 is slower, and the reason you why is because “equilibrium position lies more to the other chair conformation (with LG and CH3 in equatorial), not the required chair (with LG and CH3 in axial)”, so by kinetics, rate is slower as concentration of required chair is lower.
But in other places, the reason said is because the “transition state for the chair having CH3 and Br in axial position is more unstable, so activation energy is larger”.
Which explanation is correct?
Wow that was very helpful thanks a lot!
Your explanation and pictures are very helpful, thank you. But with the elimination products, the other unshown product is not enantiomer but a constitutional isomer, isn’t it? I’m afraid if I understood something totally wrong. :(
Draw it out. It would still be 4-methyl cyclohexene, so it can’t be a constitutional isomer. Does it look superimposable on the *other* 4-methyl cylohexene? : – )
Can you explain dehydrohalogenation in 1-bromo-2-methyl-4-tertiarybutyl cyclohexane
It’s safe to assume the t-butyl group will prefer to be equatorial. With respect to the 1-bromo group, E2 will only occur when the Br is axial. So you’ll have different rates of reaction for cis- and trans- 1-bromo-4-t-butylcyclohexane, since one will have an axial Br when t-butyl is equatorial, and the other will only have an axial Br when the t-butyl is axial. You can determine for yourself which elimination will be faster. The situation is further complicated with 1-bromo-2-methyl-4-t-butyl cyclohexane depending on the stereochemistry of the methyl group.
Sir,is there any significance of heating,like for eg: if 1 bromo(wedge position) 2,2 dimethyl cyclohexane treated with NaOMe and MeOH without heating ,what wil be d product and if heated will the product change.like without heating will it undergo SN2 and by heating will it undergo E2??plz reply as soon as possible.thanking u
On a secondary alkyl halide, especially one adjacent to two methyl groups, elimination will be by far the dominant product.
Hi James,
Just want to thank you for such a wonderful website! I am a Biochemistry major, and Organic Chemistry has been my passion; however; there are times where some of the concepts get confusing, and your website helps to remind myself that everything in chemistry should make sense!
Thank you!
Glad you find the website helpful Nata!
Dear James:
I want to ask why in the last example, why both reactions give enantiomers? For example, the cyclohexane ring with both methyl and bromine on dashed lines. After elimination, shouldn’t the methyl group stay on the dashed line? How can it be enantiomers?
thanks a lot!!
The double bond can form in both carbons adjacent to the carbon with the leaving group (beta carbons). If you draw both products and flip one over you’d see that they are enantiomers: both would have the double bond between the same carbons but the methyl group would be either a dash or a wedge.
Excellent work James. I thought I was decent at organic chemistry but these lessons make everything even more simpler. I discovered that I still have a lot to learn.
Hello James
Thank you for the wonderful clarification
Two questions (apologies if these sound too naive):
1. To my knowledge anti periplanar conf simply means that dihedral angle between two groups is 180 degrees, and that this is required for e2. Now why can’t this be true if both, the leaving group and alpha-hydrogen are equatorial?
2. In trying to acquire this conf so that e2 can take place, can we convert from cis to trans, and vice versa?
1. There’s no substitute for making a model and seeing this for yourself.
2. Ring flips can never convert cis to trans or vice versa. Ring flips only switch axial to equatorial. Again, make a model and show this to yourself.
Hi James. For the 4 examples you gave us to try, I worked out the 2nd example (1-bromo-1,2-dimethylcyclohexane) and I get my major product as 1,2-dimethylcyclohex-1-ene (where the double bond is between c1-c2). You have the major product having the double bond between c1-c6. Can you PLEASE explain why that is the major product? Doesn’t Zaitsev’s rule tell us to have a MORE SUBSTITUTED alkene (my answer) versus least substituted (your answer)? I would really appreciate a response!
P.S. Your website is the reason I’m doing well in Organic and I cannot thank you enough. :)
-Anum
For the second example, which is E2, the bromine needs to be on the opposite side of the ring from hydrogen for the elimination to occur. This isn’t possible with C2, because the hydrogen is on the SAME side of the ring as the bromine. Therefore we can’t form an alkene here – it forms between C1 and C6.
2nd scheme, 2nd reaction, 3rd structure: it would be really helpful if you showed the C2-attached hydrogen, as it is directly involved in the reaction. The same for the 1st reaction probably wouldn’t hurt, either.
Otherwise excellent work, as usual.
Thank you again, I very much appreciate your keen eye.
Yes, that is a valid point.