Reactions of Aromatic Molecules
Intramolecular Friedel-Crafts Reactions
Last updated: May 28th, 2026 |
The Intramolecular Friedel-Crafts Reaction
We explore the intramolecular Friedel-Crafts reaction in this post. But first, a very quick refresher on intramolecular reactions in general.
Table of Contents
- Quick Recap: Intramolecular Reactions Can Be Tricky
- Intramolecular Friedel-Crafts Alkylation
- Intramolecular Friedel-Crafts Acylation
- Summary: Intramolecular Friedel-Crafts Reactions
- Notes
- Quiz Yourself!
- (Advanced) References and Further Reading
1. Quick Recap: Intramolecular Reactions Can Be Tricky
Here is an instant formula for an organic chemistry exam question.
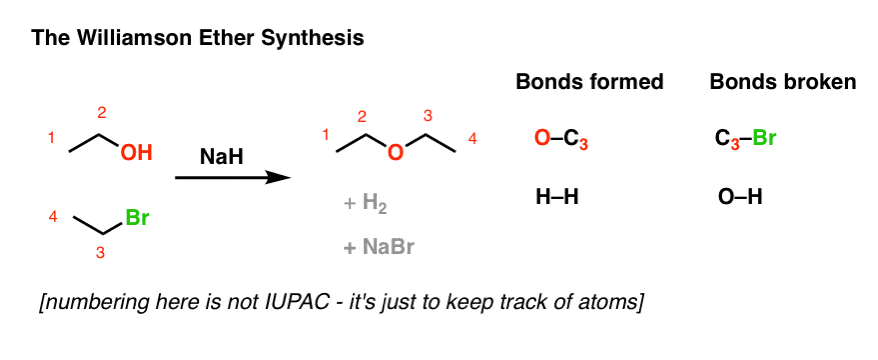
Start with a straightforward reaction that students understand fairly well, like the Williamson Ether synthesis….
(drawn weirdly, for a good purpose)
…now, just make a simple modification by adding one bond. Voila. Instant stumper!
The second question above is an example of an intramolecular reaction, where the nucleophile and electrophile are on the same molecule, and the result of their reaction is that a ring is formed. In past posts on this subject, I’ve used the analogy of a belt. It still works.
Why are intramolecular reactions good exam questions? Because they sort out the students who learn the reactions by memorizing a table of simple examples, and those who actually know (and most importantly, can apply!) the pattern of bonds formed and bonds broken. Furthermore, it involves no new concepts, which makes it totally fair game.
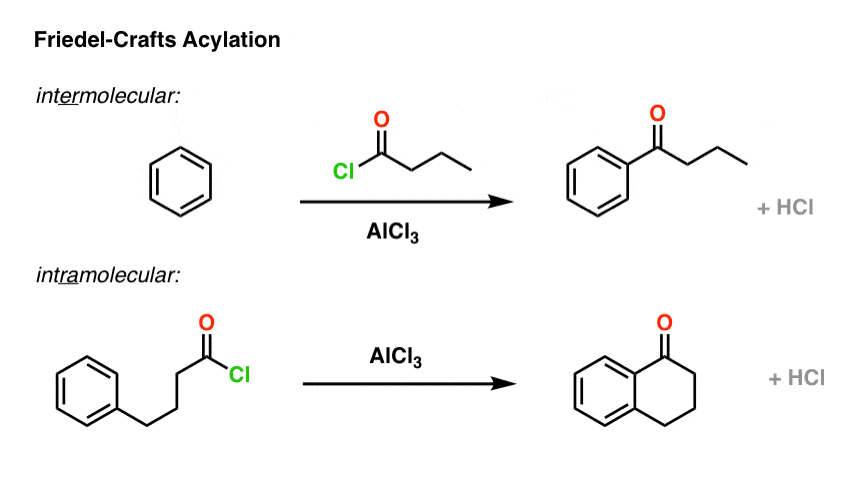
Intramolecular variants exist for a lot of different reactions, and it comes up so often that it’s worth mentioning separately. The Friedel-Crafts alkylation and Friedel-Crafts acylation reactions are no exception.
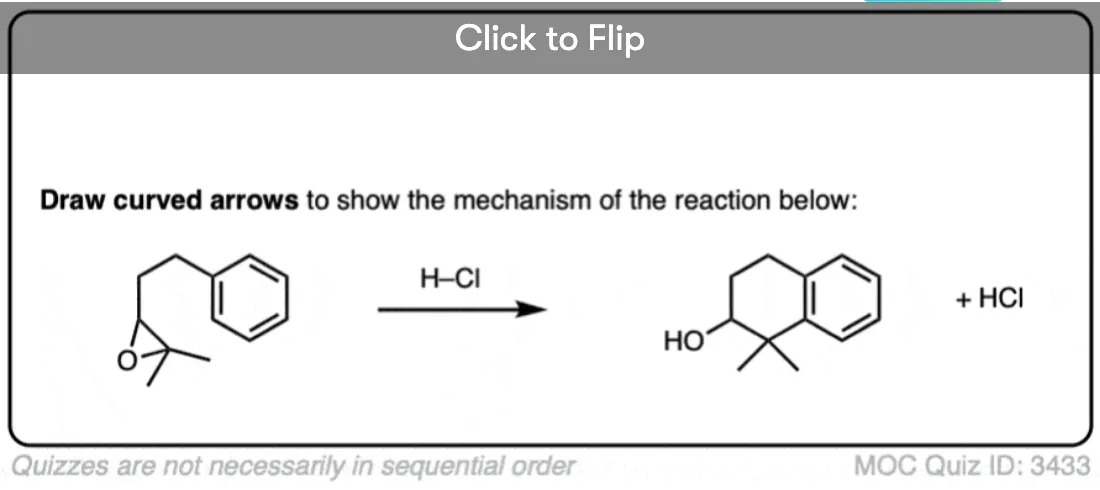
2. Intramolecular Friedel-Crafts Alkylation
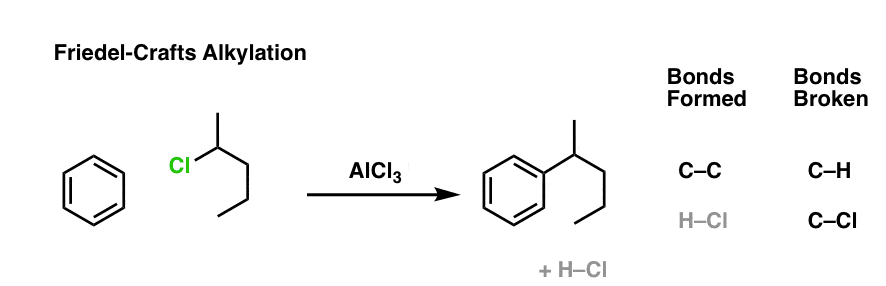
Here’s an example of an intermolecular Friedel-Crafts alkylation. The nucleophile is the aromatic ring, and the electrophile is the alkyl chloride. Add a little catalyst (AlCl3) and boom! electrophilic aromatic substitution.
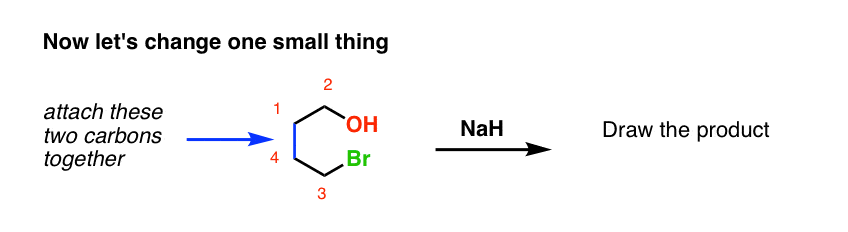
Now let’s change things up just a little bit. We’ll attach the alkyl halide to the ring via a new carbon-carbon bond, and then add the catalyst. What’s the product?
No new concepts! Same pattern of bonds that form and break. But if you haven’t seen an example like this before, it might throw you completely off your memorized notes. That’s the plan! [Cue recording of “evil laugh”].
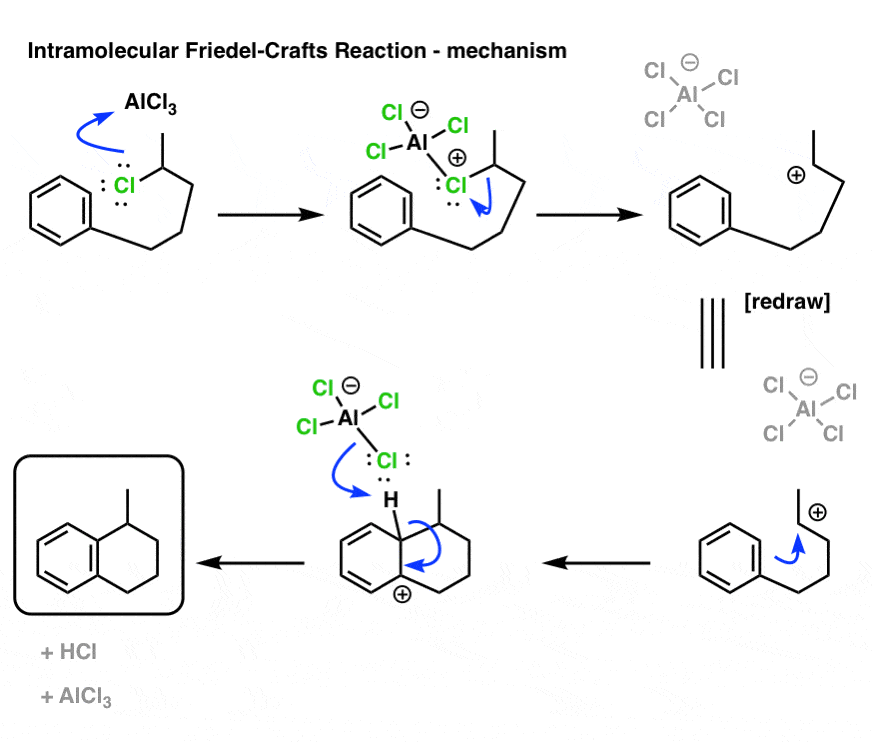
OK. Here’s the mechanism, and the final product is drawn out below left.
I want you to note that the overall pattern of bonds that form and bonds that break is exactly the same in the intramolecular case as it is in the intermolecular case, namely: form C-C and H-Cl, break C-H and C-Cl.
Timeless advice for drawing out the mechanism for a reaction like this:
- number the carbons! it’s really easy “drop” a carbon in your drawings, and that will lose you points.
- draw the “ugly version” first, and THEN re-draw to make it look nice. Don’t worry about making it look pretty until you have drawn in the bonds that form and break.
In the case of the Friedel-Crafts, the intramolecular version works best for making 6-membered rings, but it’s also possible to use the Friedel-Crafts to make 5- and 7- membered rings as well (not shown).
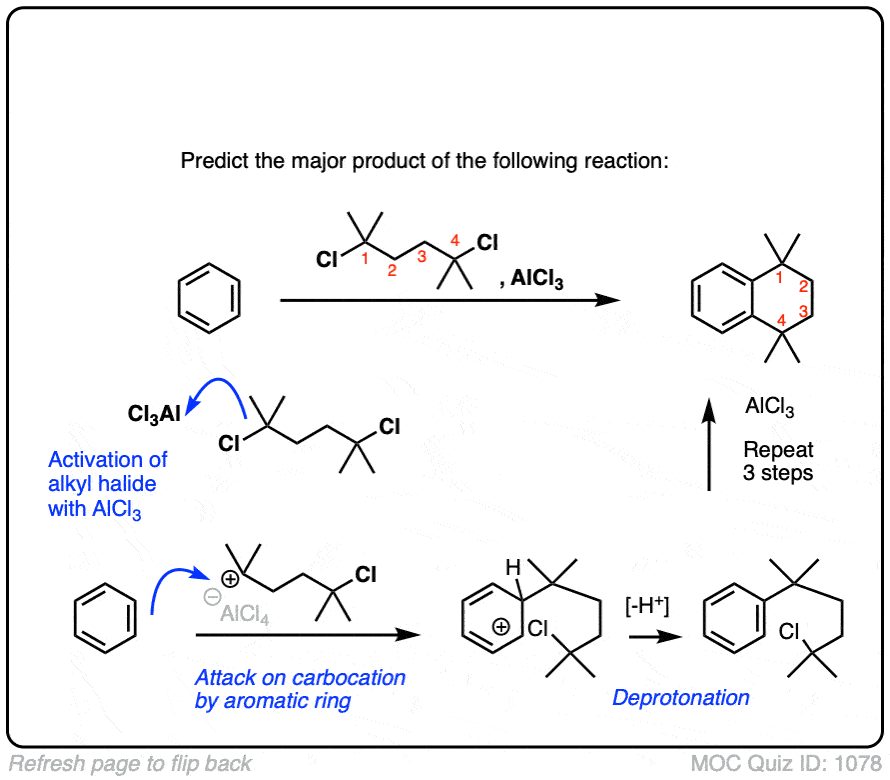
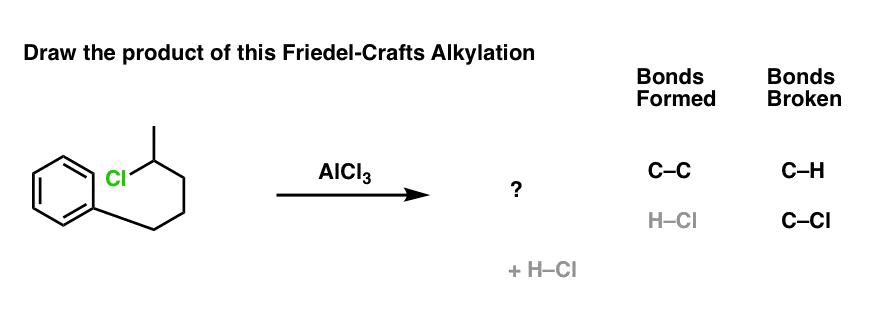
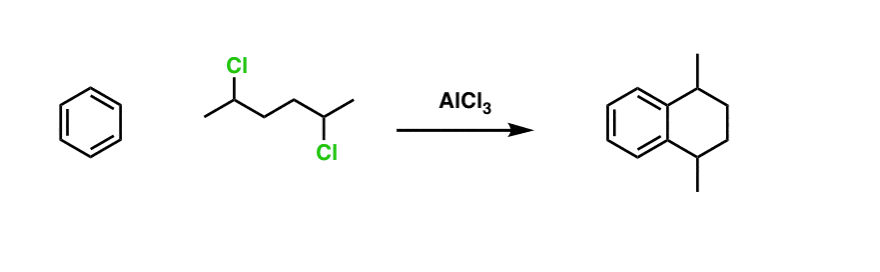
Here’s a slightly more advanced practice problem that starts with benzene. Can you draw the final product? (answer below)
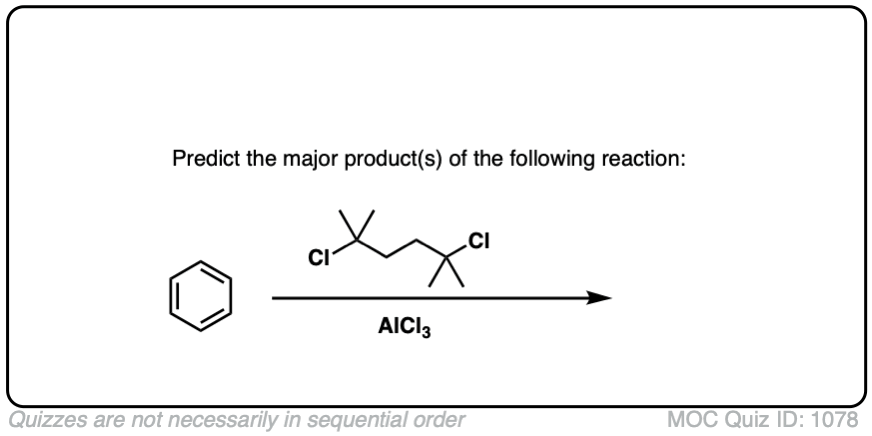 Click to Flip
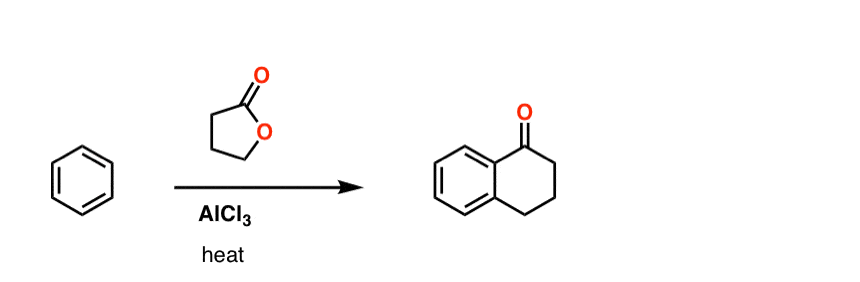
Click to Flip
3. Intramolecular Friedel-Crafts Acylation
Once you’ve seen the intramolecular Friedel-Crafts alkylation, the intramolecular Friedel-Crafts acylation is not exactly going to come as a surprise.
Again, I want you to verify that the bonds being formed and broken are exactly the same in each case. The only difference is that in the second case, the nucleophile and the electrophile are attached to each other through a tether.
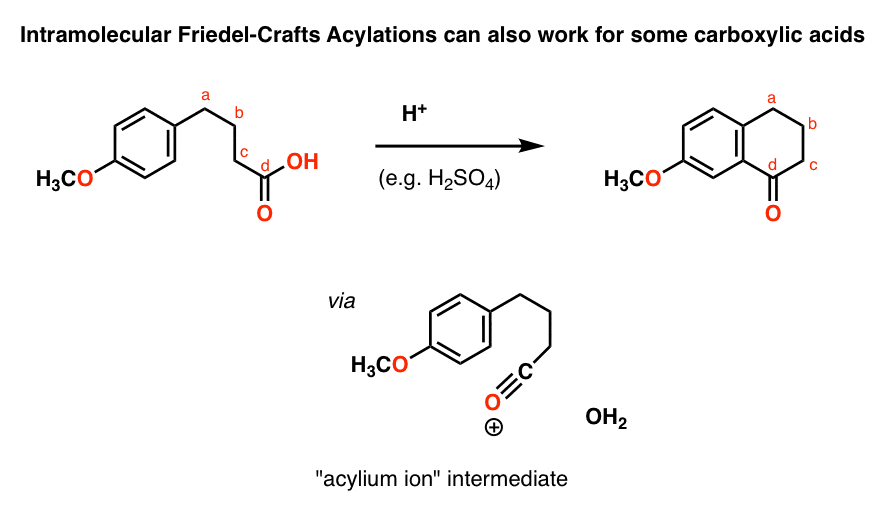
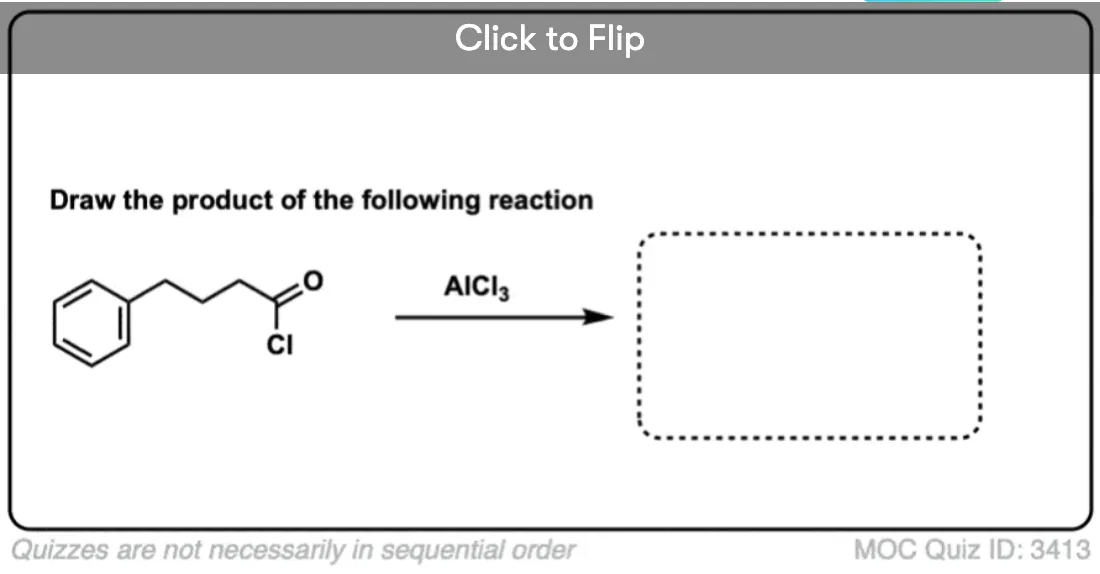
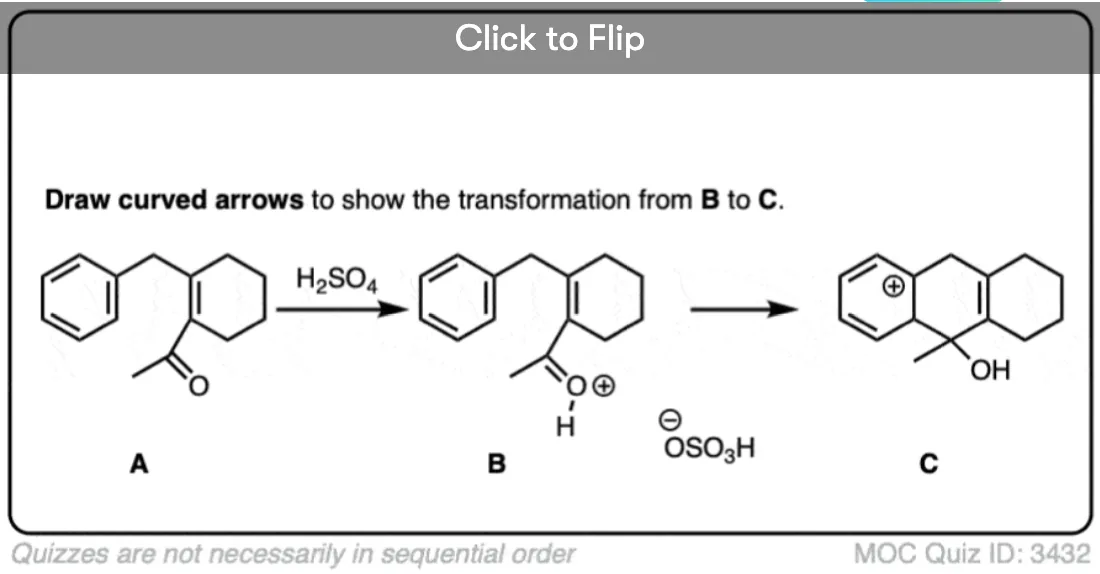
One little wrinkle that you probably won’t see, but what the heck. We’re used to seeing acyl halides (and anhydrides) in the Friedel-Crafts, but one interesting thing to note about the intramolecular Friedel-Crafts acylation is that carboxylic acids can participate too. For example, treating this carboxylic acid with a strong acid such as H2SO4 results in an intramolecular Friedel-Crafts acylation:
One reason why this works well for the intra- versus the intermolecular case is that the nucleophile is held so closely to the electrophile. This has the same effect as if the concentration of the electrophile was increased dramatically. You might see other examples where you can “get away with” using a poor nucleophile (or electrophile) in a given reaction if it’s done in an intramolecular fashion.
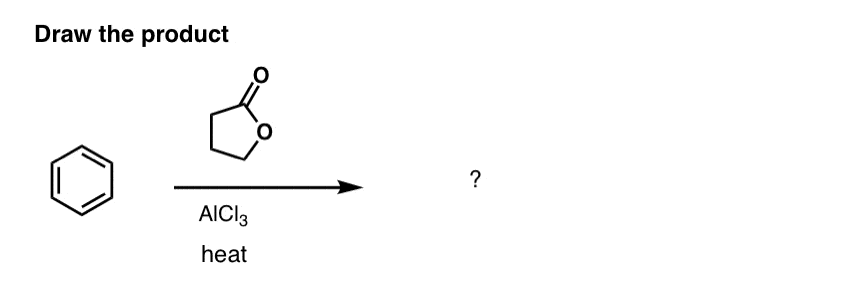
Here’s a challenge question for you. Can you draw the product of this sequence of Friedel-Crafts reactions?
4. Summary: Intramolecular Friedel-Crafts Reactions
No really groundbreaking new concepts here, but it’s always helpful to keep alert for intramolecular examples of reactions. This concept never goes away. You will see it again!
In our next post in this series we’ll cover a completely different class of substitution reactions on aromatic compounds that works well with electron-poor aromatic groups. It’s called Nucleophilic Aromatic Substitution.
Notes
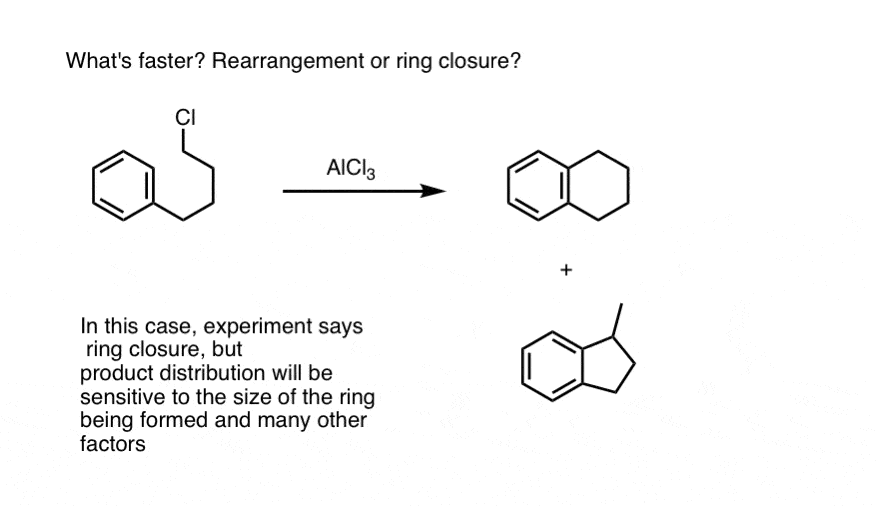
Note 1. Rearrangement vs. ring closure. We’ve seen that primary alkyl halides can rearrange via hydride (and alkyl) shifts to give the “more stable” carbocation intermediates. So what happens when a primary alkyl halide is involved in an intramolecular Friedel-Crafts alkylation reaction? Does rearrangement happen first, or is ring closure faster?
This is not the kind of question you can answer simply by thinking about it. When there are competing reaction rates, the only way to know for sure is through experiment.
This was studied, and the full paper on the study of rates of rearrangement versus ring closure is here. [J. Org. Chem, 1966, 31, 89]. This would indicate that closure to the 6-membered ring is faster than closure to the 5 membered ring in this case.
Quiz Yourself!
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
(Advanced) References and Further Reading
Friedel-Crafts acylation and alkylation can be intramolecular, and this is useful for the synthesis of bicyclic or polycyclic compounds.
Intramolecular alkylation:
Intramolecular F-C alkylations are most successful for the preparation of 6-membered rings, although 5- and 7-membered rings have also been closed in this manner. It is somewhat easier to form 6-membered than 5-membered rings in these reactions. 4-phenyl-1-butanol gives the cyclized tetralin in 50% yield in phosphoric acid, whereas 3-phenyl-1-butanol is mainly dehydrated to alkenes.
If a potential carbocation intermediate can undergo a hydride or alkyl shift (or Wagner-Meerwein rearrangement), this occurs in preference to ring closure of the 5-membered ring. This reflects a rather general tendency for 6 > 5,7 in ring closure by intramolecular Friedel-Crafts reactions.
- New Friedel—Crafts Chemistry. XVI.1 A Reconsideration of Cyclialkylation and Competing Reactions of Certain Phenylalkyl, Benzoylalkyl, and Acetylphenylalkyl Chlorides
Ali Ali Khalaf and Royston M. Roberts
The Journal of Organic Chemistry 1966, 31 (1), 89-95
DOI: 1021/jo01339a018 - New Friedel-Crafts chemistry. XIX. Cyclialkylations of some phenylalkanols
Ali A. Khalaf and Royston M. Roberts
The Journal of Organic Chemistry 1969, 34 (11), 3571-3574
DOI: 1021/jo01263a075 - Friedel-Crafts cyclialkylations of certain mono- and diphenyl-substituted alcohols and alkyl chlorides
Ali Ali Khalaf and Royston M. Roberts
The Journal of Organic Chemistry 1972, 37 (26), 4227-4235
DOI: 1021/jo00799a001 - Cyclization of 2-[N-(methylsulfonyl)anilino]acetaldehyde diethyl acetals to indoles. Evidence for stereoelectronic effects in intramolecular electrophilic aromatic substitution
Richard J. Sundberg and Joseph P. Laurino
The Journal of Organic Chemistry 1984, 49 (2), 249-254
DOI: 10.1021/jo00176a007
This paper has a section discussing the differences in the transition state geometries for 5- and 6- membered intramolecular ring closure. 5-membered transition states are significantly more strained, especially if it is assumed that the electrophilic carbon attacks from a direction perpendicular to the plane of the ring.Further examples of intramolecular Friedel-Crafts alkylations: - Experiments directed toward the total synthesis of terpenes. XVII. Development of methods for the synthesis of pentacyclic triterpenes based on a mechanistic interpretation of the stereochemical outcome of the Friedel-Crafts cyclialkylation reaction
Robert E. Ireland, Steven W. Baldwin, and Steven C. Welch
Journal of the American Chemical Society 1972, 94 (6), 2056-2066
DOI: 10.1021/ja00761a044
This is an example of a polycyclic ring system, where the product is a 3:1 mixture of b:a methyl isomers at the new ring junction, reflecting a preference for the orientation of the groups in the transition state. - A systematic study of benzyl cation initiated cyclization reactions
Steven R. Angle and Michael S. Louie
The Journal of Organic Chemistry 1991, 56 (8), 2853-2866
DOI: 10.1021/jo00008a049
This paper examines intramolecular F-C alkylation onto a benzylic position. 6-membered rings are formed more efficiently than 5- or 7-membered rings. - Enantiospecific synthesis of (+)-(R)-1-phenyl-3-methyl-1,2,4,5-tetrahydrobenz[d]azepine from (+)-(S)-N-methyl-1-phenyl ethanolamine (halostachine) via arene chromium tricarbonyl methodology
Steven J. Coote, Stephen G. Davies, David Middlemiss, Alan Naylor
Tetrahedron Lett. 1989, 30 (27), 3581-3588
DOI: 10.1016/S0040-4039(00)99447-4
The intramolecular F-C alkylations done with standard conditions (e.g. H2SO4/TFA or HBF4·OMe2) lead to racemic product, as expected. Chirality can be induced through an unusual (but advanced) methodology involving organometallics – reversibly forming an arene-Cr(CO)3 complex.Intramolecular acylation: - The Synthesis of 2-Hydroxy-17-equilenone
E. Bachmann and W. J. Horton
Journal of the American Chemical Society 1947, 69 (1), 58-61
DOI: 10.1021/ja01193a014
A classical reagent for intramolecular cyclization of phenalkyl carboxylic acids is PPA (polyphosphoric acid), which can be made by adding P2O5 and phosphoric acid. This is not used all that much anymore, since it is a pain to handle – very corrosive, and extremely viscous. - Methanesulfonic acid. A useful cyclizing acidic reagent
Alberto A. Leon, Guido Daub, and I. Robert Silverman
The Journal of Organic Chemistry 1984, 49 (23), 4544-4545
DOI: 10.1021/jo00197a047
MSA (methanesulfonic acid, CH3SO3H) is an alternative for PPA in F-C cyclization reactions. It is cheap, readily available, and is a easily handled liquid, comparable in acidity to PPA. - Spectroscopic and Other Properties of Large Ring Mono- and Dimeric Benzocyclanones Prepared by a High-dilution Friedel-Crafts Reaction
M. Schubert, W. A. Sweeney, and H. K. Latourette
Journal of the American Chemical Society 1954, 76 (21), 5462-5466
DOI: 10.1021/ja01650a060
While intramolecular F-C acylation is mainly used to close 5-, 6-, and 7-membered rings, even larger rings can be closed by high-dilution techniques. - Efficient synthesis of selected indenones
Brawner. Floyd and George Rodger. Allen
The Journal of Organic Chemistry 1970, 35 (8), 2647-2653
DOI: 10.1021/jo00833a036
Intramolecular F-C acylation can also be done with acyl chloride/AlCl3.Tandem reactions with both acylation and alkylation are also possible; these are termed cycli-acyalkylations. Unsaturated acids or lactones can be used. - α-TETRALONE
Cecil E. Olson, Alfred R. Bader, and G. Dana Johnson
Org. Synth. 1955, 35, 95
DOI: 10.15227/orgsyn.035.0095
The first procedure, submitted by A. R. Bader, uses a lactone to efficiently do a tandem F-C cycli-acylalkylation. A. R. Bader went on to found the Aldrich Chemical company, which made him extremely wealthy and forever changed the course of chemical research – chemists now do not have to spend their efforts resynthesizing common starting materials, as these are available off the shelf at high purities. - The scope of the Haworth synthesis
Israel Agranat and Yu-Shan Shih
Journal of Chemical Education 1976, 53 (8), 488
DOI: 10.1021/ed053p488
The first part of the Haworth synthesis involves F-C acylation with an anhydride, followed by intramolecular F-C alkylation after reducing the ketone.
I wondered that the activated lactone by Alcl3 in answer #2 is which part in lactone reacted with Alcl3? Is the oxygen in lactone ring or the oxygen in carbonyl group? How should I choose from two of them?
Just like acyl chloride is the chloride react with the Alcl3
Good question. The carbonyl oxygen is activated by AlCl3. This sets up the carboxylate to act as a leaving group when the ring attacks at the sp3 hybridized carbon. That’s the first Friedel-Crafts. The second Friedel Crafts is an acylation reaction where O-AlCl3(-) is the leaving group.
I’m most of the way through the tetralone mechanism, but I can’t figure out the final dissociation of the aluminium complex. I had the bridging oxygen in the ester complex initially with the aluminium trichloride first, break the ring and the acylium formation. F-C acylation and rearomatization like normal, but here is where I’m confused. The aluminium centre has a negative charge, and the oxygen is no longer positively charged, so if the C-O bond breaks to alkylate, I have a doubly negative aluminium species. Where did I go wrong?
Thanks!
Right. Try the alkylation first – your leaving group will be RCO2Alcl3, which will then be well primed for FC acylation
Sir please tell the mechanism when acid anhydride react with 2 eq of alcl3 with benzene
Answer #2??? I need an explanation!!!
It’s a sequence of friedel-crafts acylation and then friedel-crafts alkylation. First step: AlCl3 reacts with the oxygen of the lactone, resulting in formation of the acylium ion which is attacked by the aromatic ring. Now you have an oxygen coordinated to AlCl3 (a good Lewis acid). The AlCl3 converts the oxygen into a good leaving group. So what can happen next is attack of the aromatic ring at the carbon bearing the oxygen, resulting in formation of a new six-membered ring.
In the example given for intra mole ular freidal crafts reaction ,acylium
Cation attacks meta to OCH3 group which is ortho para directing .It is not the major product.
You are proposing that a seven membered ring would form containing a trans double bond. That is not going to be a stable molecule.
The article is very nice. I would like to know source or reference for the particular example where you intramolecular acylation of carboxylic acids as I have some confusion as to why the ether has not been hydrolysed.
The reference is linked at the bottom, but it’s Organic Syntheses, Coll. Vol. 4, p.898 (1963); Vol. 35, p.95 (1955).
What happens with the alcohol is that it eventually coordinates to AlCl3 which activates it towards attack by the aromatic ring, and it departs as a leaving group.
But an R group is an ortho para director, and the major product is para, so why isn’t it close and connect to the para position of the aromatic ring?
The answer is that the para product would be very strained. The carbonyl can’t reach the para position in a way that leads to non-strained bond lengths and bond angles. If you are unsure about this I invite you to try to build a model!
In practice problem #1 it appears that you changed the reactant from a seven carbon chain in the practice problem to a six carbon chain in the answer key.
Shoot. Thank you Kirk, will fix.
Fixed, finally.
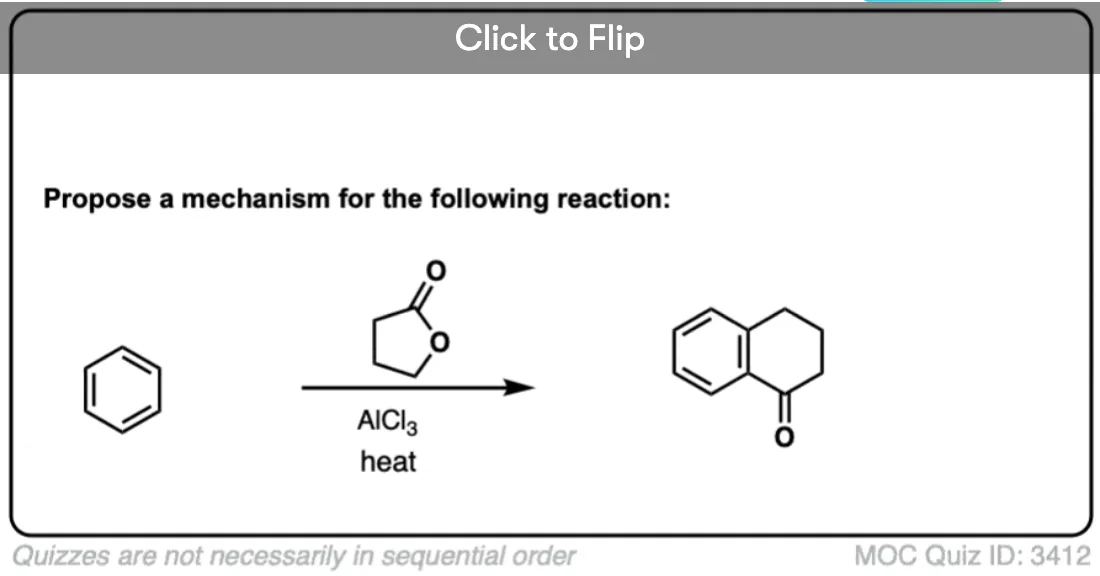
I wonder if there would be any takers to propose the mechanism for the tetralone formation via the above mentioned Friedel-Crafts :) I used to put that one on an exam for like 3 or 4 years. Students didn’t appreciate the challenge…
Yeah, that would definitely not win any fans.