Aldehydes and Ketones
What Is The Alpha Carbon In Carbonyl Compounds?
Last updated: May 29th, 2026 |
Alpha Carbon? Beta Carbon? Alpha, Beta Unsaturated Carbonyl?
Making it through the chemistry of carbonyl derivatives (ketones, aldehydes, carboxylic acids, esters, and more) there are at least two “weird” nomenclature issues that repeatedly come up to baffle students: Greek letters, and “1,2-” or “1,4-” addition reactions. In this post I’ll try to address them both.
Table Of Contents
- The “Alpha Carbon” Is The Carbon Adjacent To The Carbonyl.
- “1,2” and “1,4” Additions To “Alpha, Beta” Unsaturated Carbonyls
- Quiz Yourself!
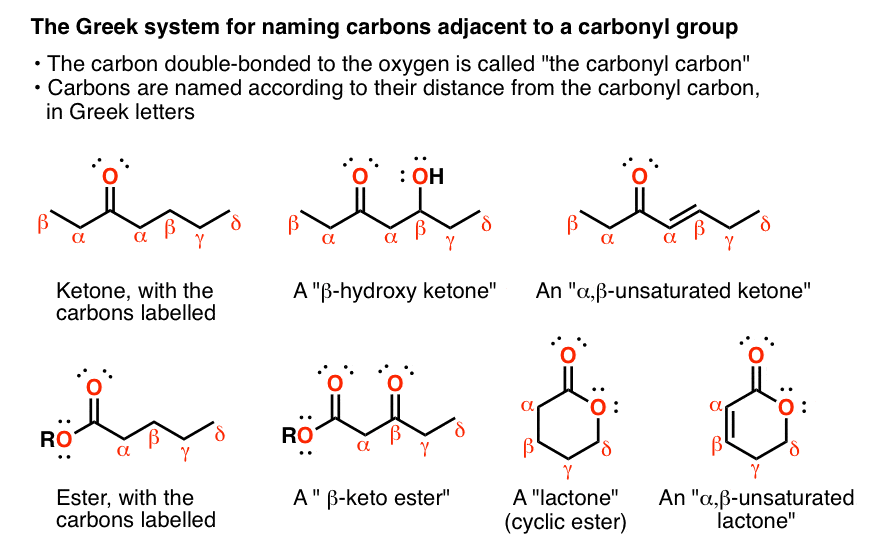
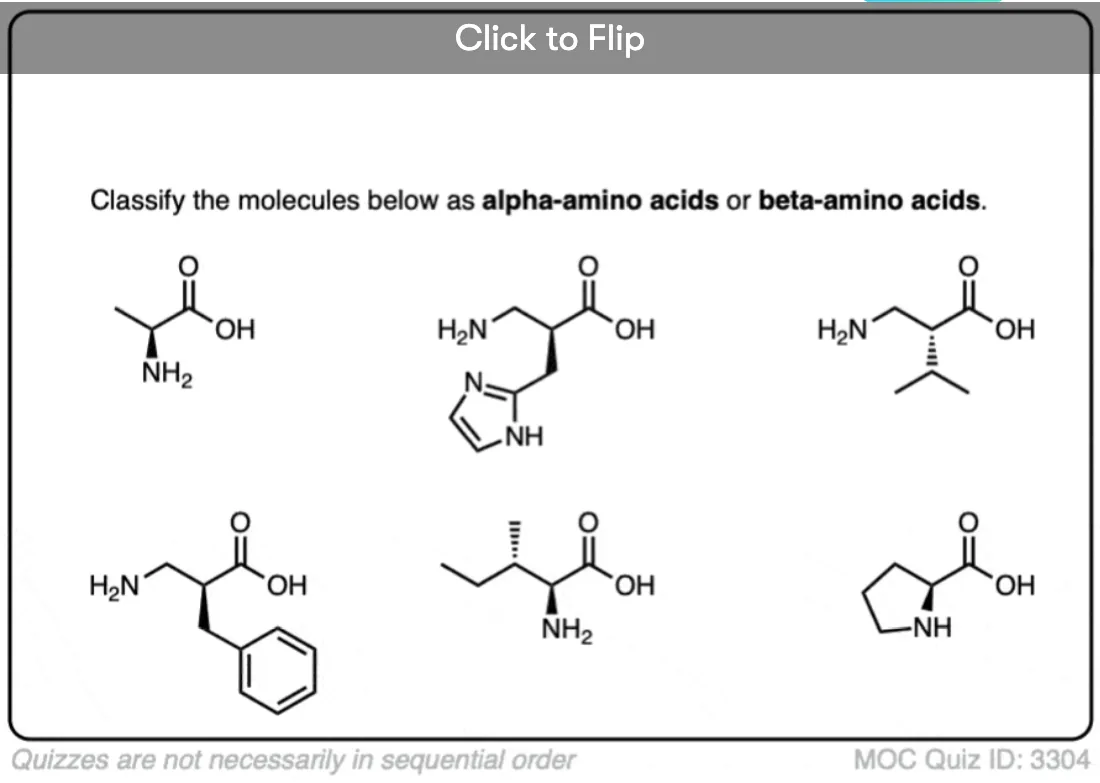
1. The “Alpha Carbon” Is The Carbon Adjacent To The Carbonyl
The functional group C=O is called a “carbonyl”. The carbon itself is called the “carbonyl carbon”, and the oxygen is called “the carbonyl oxygen”. But what do you call a carbon adjacent to the carbonyl carbon… or 3 carbons away?
- In organic chemistry, it’s common to use Greek letters to denote this. So the carbon adjacent to a carbonyl is called an “α (alpha) carbon”, two carbons away is called a “β carbon”, and so on.
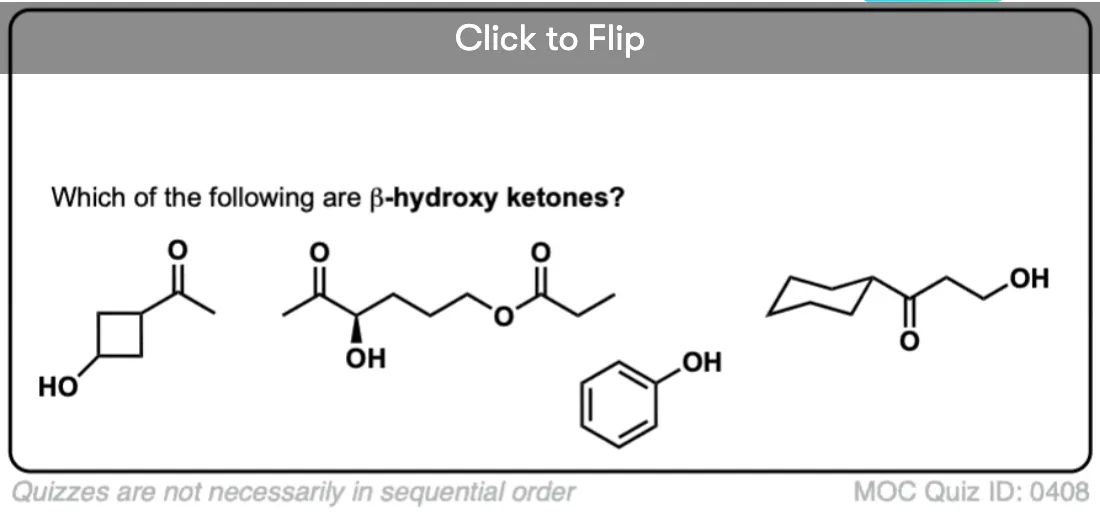
- This nomenclature can be used to depict different kinds of substituted carbonyl groups. For example a ketone with an OH on the beta carbon would be called a “β-hydroxy ketone”. If it was one carbon further down it would be a “γ hydroxy ketone”.
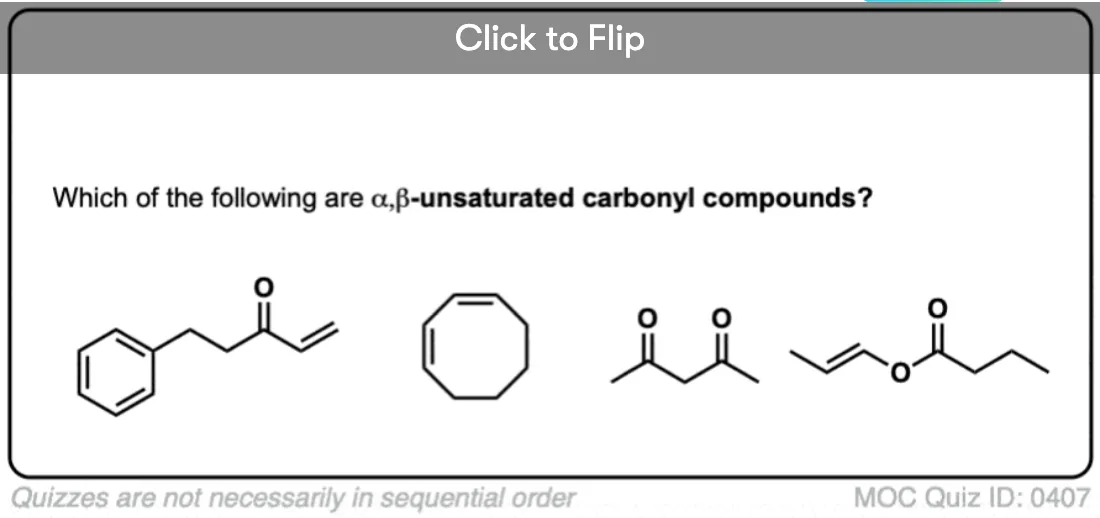
- If we have a double bond between the α carbon and the β carbon it’s common to call it “α,β-unsaturated”. So we can have α,β unsaturated ketones, aldehydes, esters, and so on.
- It can keep going beyond gamma, of course, but it’s rare to see it progress beyond ε (epsilon).
- Another thing: aldehydes, esters, carboxylic acids, and so on, can only have one “alpha” carbon each, wheras ketones can have two. Sometimes you’ll see one set of Greek symbols marked with ‘ (prime) symbols to distinguish them. The location of the prime is completely arbitrary.
- For esters, the OR group is not denoted “alpha”. It’s usually just called the “alkoxy” group.
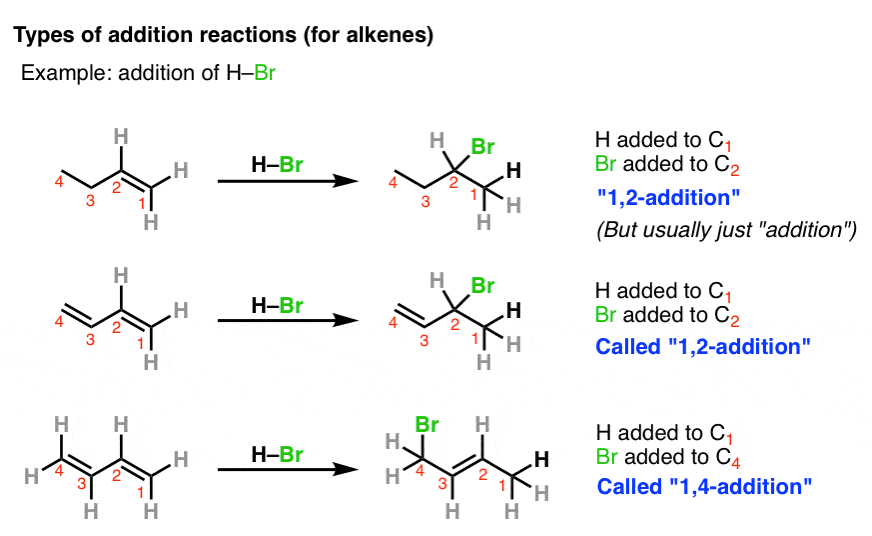
2. “1,2” and “1,4” Additions To “Alpha, Beta” Unsaturated Carbonyls
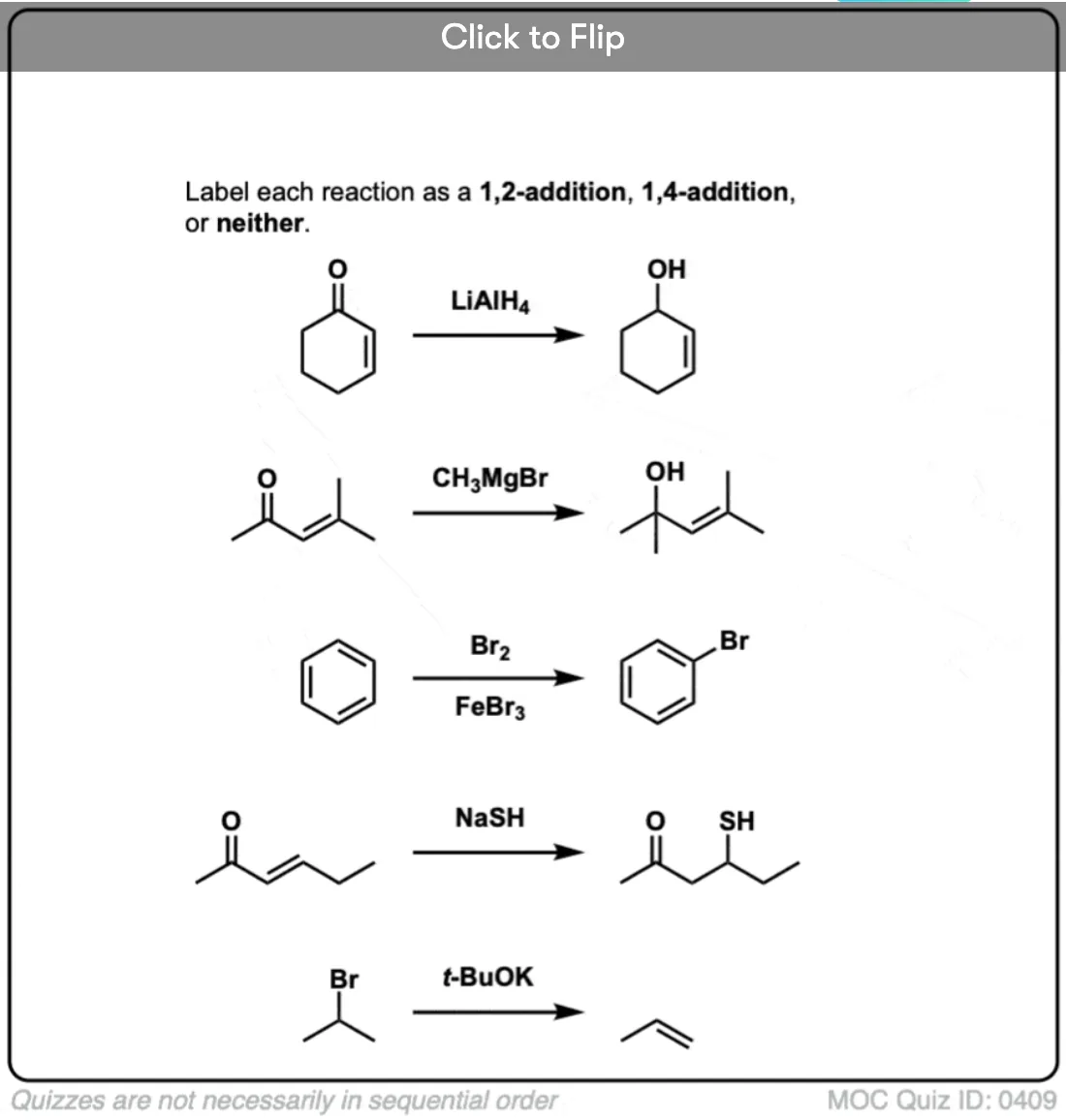
Another item of confusion are the terms “1,2-addition” and “1,4-addition”. This originates in the discussion of hydrogen halides to alkenes and dienes. Usually, addition to straight alkenes is just called “addition”. But when two or more alkenes are next to each other, at least two different products become possible. The numbers “1,2-” and “1,4-” are used to distinguish the two products from each other.
Using HBr as an example, in the first case, we’re forming C-H on carbon 1 and C-Br on carbon 2. Hence, “1,2-addition”.
In the second reaction, we form C-H on carbon 1 and C-Br on carbon 4. Hence, “1,4-addition”.
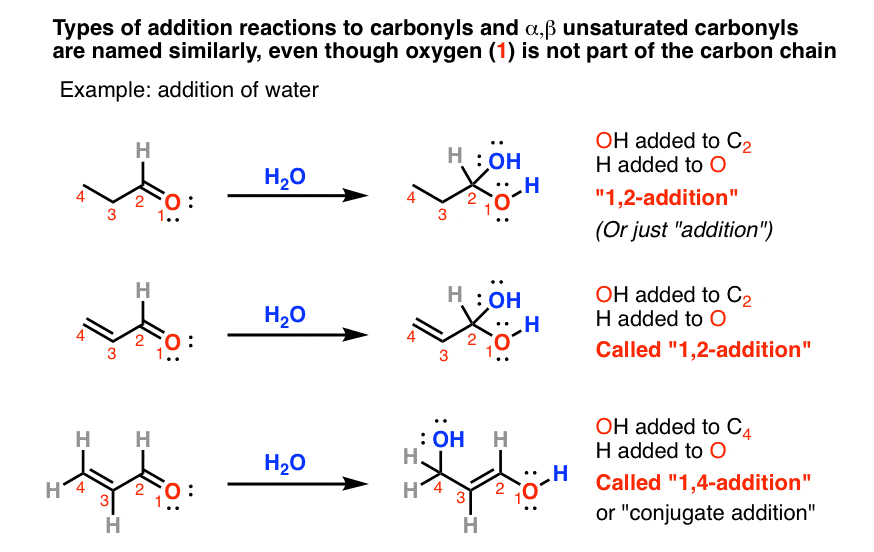
The same analogy holds for additions to carbonyls and to alpha-beta unsaturated carbonyls, but it’s a little bit confusing because the “1” in this case refers to oxygen: not an atom we’re used to numbering.
But if you can get over that little bit of weirdness, the analogy to “1,2-addition” and “1,4-addition” with alkenes is dead on. As an example, the 1,2-addition of water to an α,β unsaturated ketone gives a C-OH bond on position 2 and an O-H bond on position 1; the 1,4-addition of water to the same ketone forms C-OH on position 4 and O-H on position 1.
(It doesn’t just stop at 1,4-additions by the way. If you have a triene (3 adjacent alkenes), it’s possible to have “1,6-additions”. Rare, but it can happen.)
Notes
Quiz Yourself!
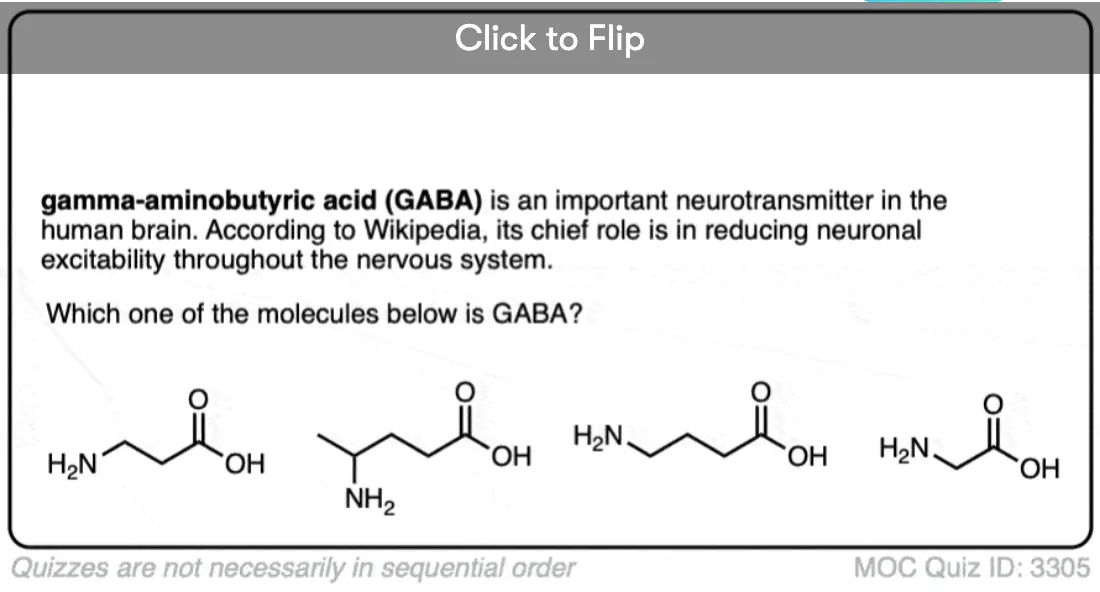
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Hi, what is the way of identifying whether a nucleophile will attack the alpha carbon or the carbonyl carbon? For example, in a claisen condensation, what is preventing the base from attacking the carbonyl carbon? Please let me know, thank you!
Hi – it can, and does, but it ends up not mattering.
For instance if you have CH3C(O)OCH3 and are using CH3O(-) as the base, one option is deprotonating the alpha carbon. The other is attacking the carbonyl carbon. However, if you attack the carbonyl carbon, you’ll eventually kick out CH3O(-), and then get…. CH3C(O)OCH3 . So you end up back where you started.
This is also why it’s smart to use a base that is identical to the OR group on the ester. Otherwise you get transesterification.
MOC QUIZ ID-0407
In the diketo compound, as the most stable tautomer will be 1-keto-2-enol form, so shouldn’t it be consider as alpha-beta unsaturated carbonyl compound?
That’s a valid question. Since one of the ketones is in the enol form, the OH has many properties of a carboxylic acid.
For this reason these compounds are sometimes called ‘vinylogous acids” since they undergo many reactions of carboxylic acids.
For example it’s possible to replace the OH group with Cl and perform addition-elimination reactions, much like one can perform nucleophilic acyl substitution reactions. https://en.wikipedia.org/wiki/Vinylogy
With regard to multiple functional groups, in the beta-keto acid example at the top of the page, I see the carbon labeled gamma as being alpha. It is both (gamma to the ester carboxyl carbon but alpha to to the ketone carbon). However for me, alpha is more relevant as in my biochemistry class, I often point out that our reactions involve the special nature of the alpha carbon (aldol and Claisen condensations, Citrate Synthase, etc…). Being gamma is not as relevant.
Good point Todd. Thank you.
Of course, alpha beta, and gamma are all relative terms. I thought about changing the diagram to put a blue “alpha” on the position where gamma is written to accomodate this point, but felt it would add some unnecessary clutter.
Furthermore, from a practical perspective with the beta keto ester, it is impossible to get deprotonation at the gamma carbon without deprotonating the alpha carbon first, and even then only after resorting to an extremely strong base to form the di-anion. So in this particular case I’m going to leave it as gamma.
Marvellous explanation, quite helpful. Especially the problems at the end are quite worthful.
Thank you, glad you found it helpful shailya. James
what if different functional groups are attached to the same chain……which Carbon is to be named as the alpha.
eg: CH2CH(Cl)CH2CH2OH or CH3C=OCH2CH2CHO
Whats the priority ?
It wouldn’t apply to the first molecule because there’s no carbonyl.
For the second, it’s a relationship issue.
So in CH3C=OCH2CH2CHO, the CH3 is alpha to the C=O; the first CH2 is alpha to the C=O but beta to the CHO; the next CH2 is beta to the C=O but alpha to the CHO.
I am confused by this as well. Even though it goes trough an intermediate enol to stabilise the negative charge, the addition (bonds formed) are 1,2 from each other.
If I (nucleophilically) attack para-vinyl-benzaldehyde at the terminal methylene, and I stabilise the negative charge of an intermediate all the way through an aromatic ring and up the para-carbonyl on the far side, then brought it back through the ring to add vicinally to the initial attack, is that a 1,8-addition? Is the numbering not based on where the new σ bonds are being made?
Thanks
yes – technically it would be 1,8-addition. The term, “1,8-addition” has even been used: https://pubs.acs.org/doi/10.1021/jo00038a053
James, are you sure the second last example with the 2-Oxo-hex-3-ene is an 1,4 addition? If this was the case, the product IMHO should be 2-Hydroxyhexane-4-thiol. I’d vote for 1,2 therefore.
Yes – it goes through an intermediate enol.
1,2 addition in this case would be addition to the carbonyl, without affecting the double bond.
Thanks for this website.
I was unknow of the term addition but now organic is my best friend. Thanks for making organic my friend. Credit goes to u.
First of all, I should appreciate your “magnanimity” in sharing your knowledge. I have a question about Alpha, Beta, and gamma hydrogen. I have seen a definition for these nomenclature in a paper available in below link:
http://www.sciencedirect.com/science/article/pii/S0016236102003538
As you can see, the author define Alpha, Beta, and gamma hydrogen as:
H alpha: Aliphatic hydrogen on C alpha to aromatic rings.
H beta: Aliphatic hydrogen on C beta and the CH2, CH beyond the C beta to aromatic rings.
H gamma: Aliphatic hydrogen on C gamma and the CH3 beyond the C gamma to aromatic rings.
What do the CH2, CH beyond … and CH3 beyond …mean?
Please consider an aliphatic chain with 10 or more carbons attached to an aromatic ring. According to these definitions, gamma hydrogen atoms are those bond with penultimate carbon as well as those belong to the CH3 at the end of chain.
Accordingly, the gamma carbon is not the third carbon next to aromatic ring, but the penultimate one. Moreover, based on the definition presented above, we have only two and five alpha and gamma hydrogen atoms,respectively whereas there may be several beta hydrogen atoms.
The main question is that whether my inference about those definitions are true?
Thank you very much.
I am not familiar with that journal and I cannot gain access. Looks like they are doing an NMR study and want to group the hydrogens according to where they show up in the spectra. I would not take what you read in this paper as any official statement of proper nomenclature.
Oh my
thank you soooooooo much for making this website!!! really really really!
Doesn’t change the weirdness of numbering an oxygen, but at least its clearly explained. Good work James, thanks.
Some other odd terms (which I think I’ve seen you explain elsewhere) include ‘geminal’ and ‘vicinal’ which refer to the same and adjacent carbon respectively. Geminal means twin and refers to two atoms attached to the same carbon. Vicinal means neighbor and refers to two atoms attached to adjacent carbons. These terms are usually used in reference to alkene addition/elimination.
And to think, this is easy compared to what the alchemists had to deal with. I should be able to list Organic Chemistry as a foreign language on my resume
heteroatoms are numbered all the time in organic chemistry and are often given higher priorities than their adjacent carbon atoms. think of the carbon-oxygen double bond as similar to a carbon-carbon bond but with higher priority
Yes, that’s correct. For example in pyrrole the nitrogen is numbered “1” and the same is true in furan and other heterocycles.