Free Radical Reactions
Free Radical Initiation: Why Is “Light” Or “Heat” Required?
Last updated: May 7th, 2026 |
Free-Radical Reactions Require Heat Or Light For Initiation (Bond-Breaking)
If you come across just a few free-radical reactions, you should notice a familiar pattern. Every free-radical reaction that you’ll encounter is accompanied by either “heat” or “light”.
In fact, this is one of the most important clues to knowing you’re dealing with a free radical reaction!
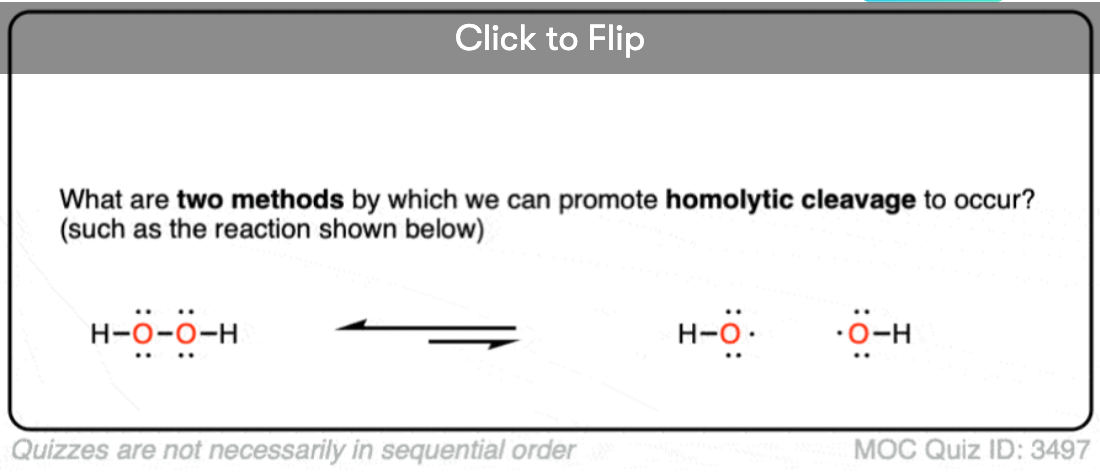
So why is that? Because the first step in free radical reactions is initiation – the homolytic cleavage of bonds to create new free radicals, and this requires an input of energy either from light or heat.
Table of Contents
- Bond Breaking Requires An Input of Energy
- The First Step In Free Radical Chain Reactions Is “Initiation”
- Notes
- (Advanced) References and Further Reading
1. Bond Breaking Requires An Input Of Energy
Free radicals are created when a bond undergoes homolytic cleavage – that is, the bond breaks such that each atom receives the same number of electrons.
It’s important to recognize that breaking a bond requires an input of energy to the molecule. [Note 1]. The energy required for bond cleavage commonly comes from two sources – you guessed it – heat, or light.
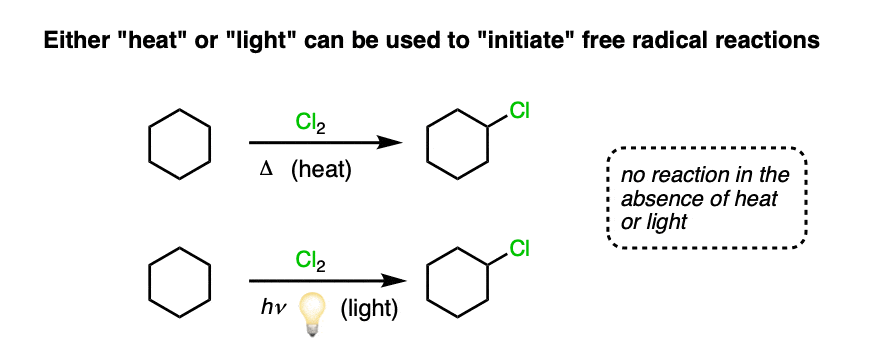
Take chlorine, for example. The heating of chlorine gas (actually – more commonly, of chlorine dissolved in a solvent) or exposure of chlorine gas to visible light results in significant cleavage of the Cl-Cl bond to deliver two free radicals, as shown below.
[Why light? recall from general chemistry, E =hν ; there is a relationship between the frequency of photons of electromagnetic radiation (“light”) and their energy. Photons that collide with molecules impart energy to them; this can be sufficient to break bonds if sufficient conditions are met – see below for more]
The energy associated with a quantum of light, 48 kcal/mol at 600 nm (orange), 96 kcal/mol at 300 nm (near-UV) is of the same order of magnitude as that of most covalent bonds. [Ref]
2. The First Step In Free Radical Chain Reactions Is “Initiation”
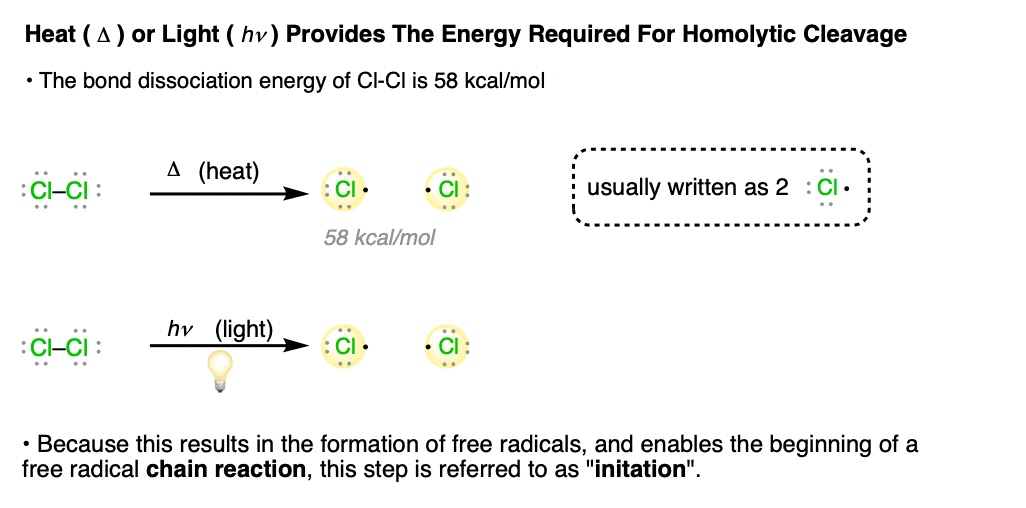
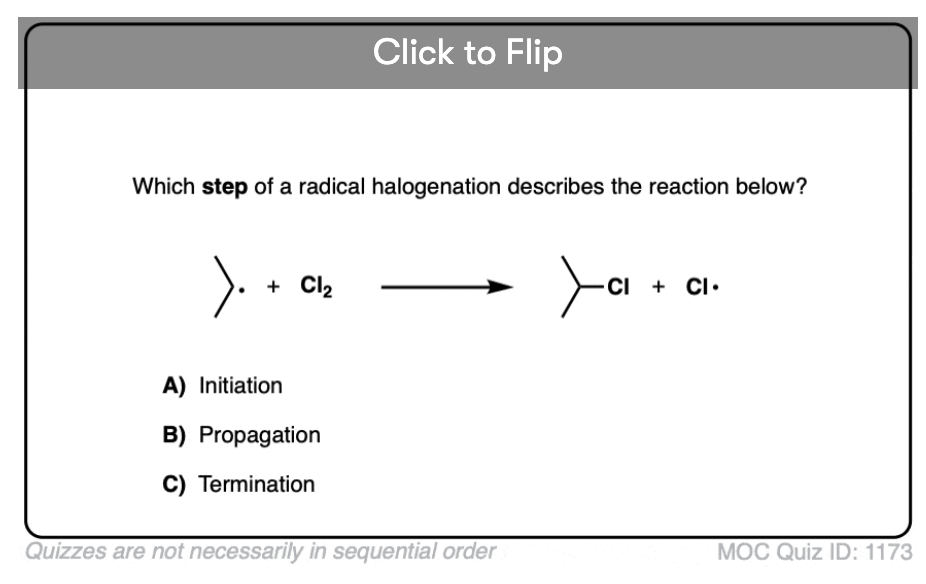
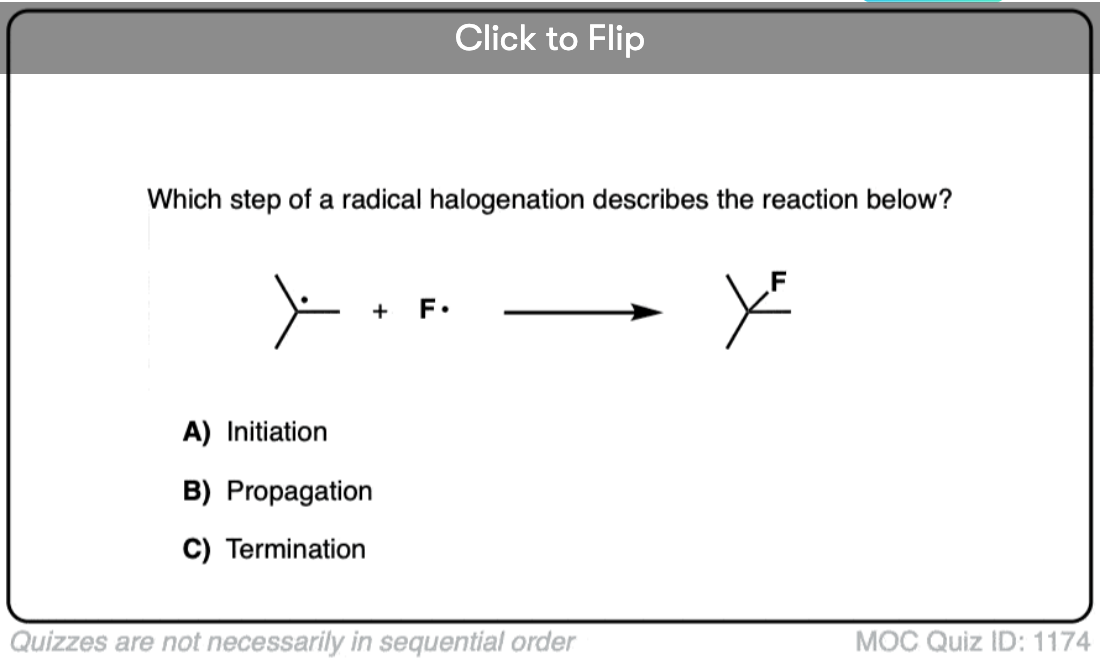
In free radical reactions, this first step – homolytic cleavage of a bond to yield free radicals – is referred to as “initiation”. In an “initiation step, the number of free radicals is always increased”.
You might ask: once a bond breaks into two radicals, what’s to stop it from re-combining? Good point! In fact – and this is often neglected in textbooks – it’s more proper to think of bond-breaking and bond-forming as being in equilibrium with each other. As we’ll go into details in the next post, only a very small concentration of free radicals is required for a reaction to take place.
This first step – initiation – is often quite slow. In fact, free radical reactions are often observed to have an induction period. That is, after all the reagents are mixed together, there is often a variable period of time where no reaction is observed, followed by a sudden – sometimes explosive! – acceleration of the rate.
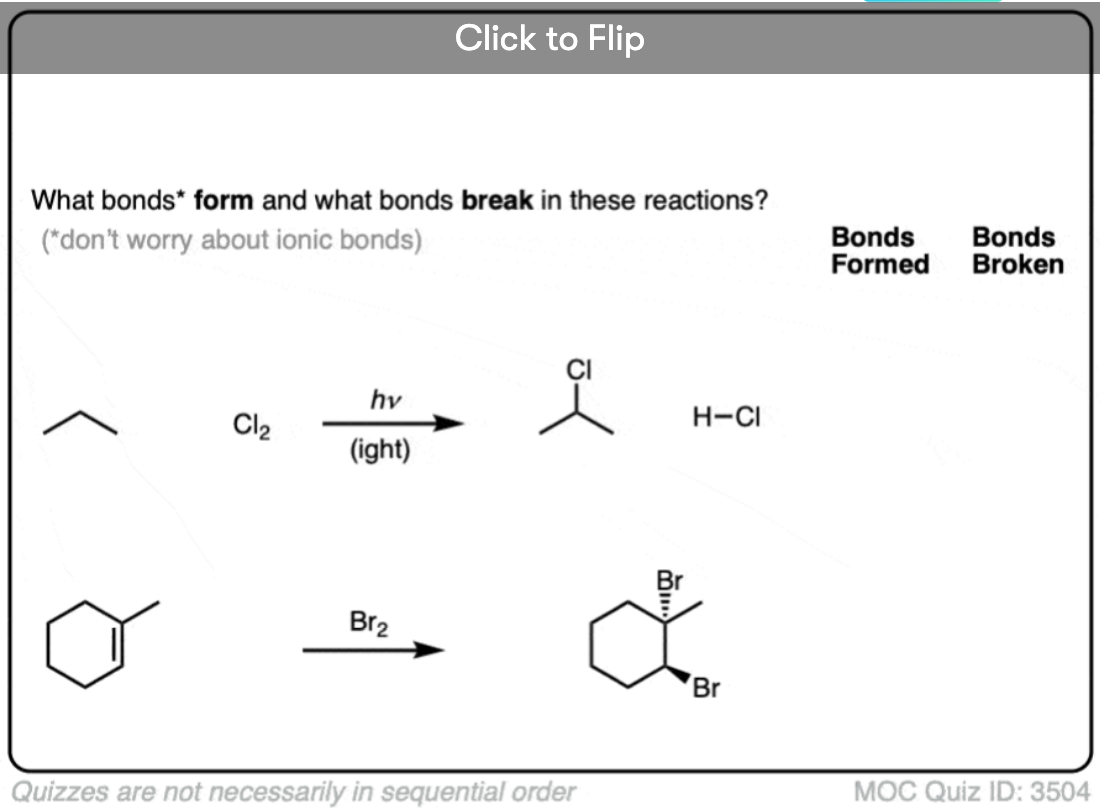
In the case of chlorination of alkanes, as we’ll see, the generation of an unstable chlorine radical is followed by subsequent removal of a hydrogen atom from an alkane followed by chlorination of the carbon free radical, in a chain reaction.
In the next post we’ll talk about the three stages of a free radical reaction – initiation, propagation, and termination.
For more specific information about what is actually happening when heat or light interacts with a molecule and how this leads to fragmentation, read on below the fold to Note 2.
Next Post: Initiation, Propagation, Termination
Notes
This is a direct quote from “Free Radicals in Solution” (1957) by Cheves Walling, p. 29.
Note 1. A common misconception that breaking bonds releases energy. This misconception probably arises because ATP is the body’s source of energy and energy is released when it hydrolyzes to ADP. However the fact that energy is released by this is because the P-O bonds in ATP are weaker than the P-O bonds being formed by hydrolysis. It’s this “trading” of a less stable P-O bond for a more stable P-O bond that releases energy.
Note 2. So what’s going on here?
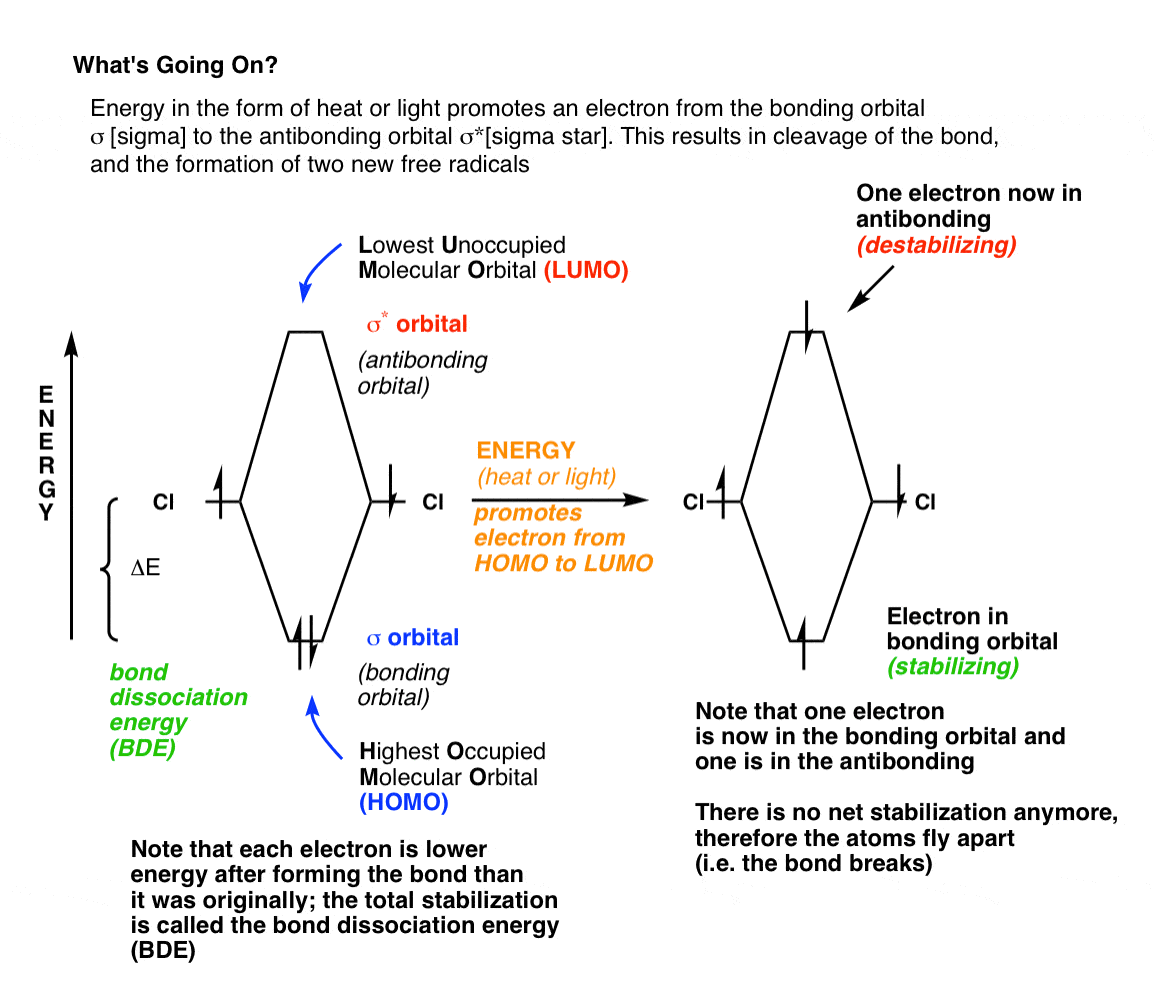
Fully understanding the importance of heat or light (i.e. energy put into the system) requires thinking about molecular orbitals. Recall that when two atoms come together to form a single (“sigma”) bond, there are two ways by which their orbitals can overlap. Constructive orbital overlap – where both orbitals have the same “sign” – results in an orbital in the space between the two atoms. Two electrons held between two positively charged nuclei results in an overall lowering of the energy of the system due to attraction between the opposite charges. This is referred to as “bonding“, and the overall energy of stabilization is referred to as the bond dissociation energy.
There is also an alternative means of orbital overlap, between two orbitals of opposite sign. This results in destructive interference, and therefore zero electron density in the space between the two atoms; the electrons are instead localized to the space away from the other atom. The result is that two positively charged nuclei are held tightly together in space without any negatively charged electrons to “glue” them together —> this is unstable, and referred to as “antibonding” (σ*). Even though putting electrons in the antibonding orbital results in instability, it’s the only orbital an electron can possibly be promoted to in our simple example. [Note 3]
Remember that energy levels in molecular orbitals work like staircases, not ramps. Imagine yourself on a staircase: the step you are standing on is the highest occupied step, and the next step up is the lowest unoccupied step. In molecules, we refer to the highest occupied molecular orbital [HOMO] which is the sigma orbital in the leftmost part of the diagram, and the lowest unoccupied molecular orbital [LUMO] which is the sigma* orbital.
When thermal energy (“heat”) is imparted to a molecule in a quantity roughly equal to the energy gap between the HOMO and LUMO, an electron can be promoted from the HOMO to the LUMO, resulting in the situation shown on the right. Here, one electron is in the bonding orbital and the other is in an antibonding orbital. No longer do we have net stabilization from the two chlorine atoms being bonded relative to the two chlorine atoms being separate [in fact, it is even more unstable due to the repulsion of the two electrons] – therefore, the most favorable course of action is for the bond to break.
Likewise, light can also act in place of heat. The energy gap between HOMO and LUMO is some value ΔE. When the frequency of light that shines upon the molecule such that E = hγ, an electron will likewise be promoted from the HOMO to the LUMO, and bond cleavage can occur.
Note 3. Students often have a hard time understanding antibonding. I recall once being asked, “why does antibonding exist?”. I will offer a non-technical analogy as means of explanation.
Imagine two people that have never met and are unaware of each others existence. Now imagine those two people meeting, getting interested in each other, falling in love, and finally getting married and living in the same house. We’ve gone from indifference (zero energy, as reference) to love (a lowering of the overall energy of the system). The couple is attracted to each other.
Now, imagine one partner being unfaithful or partaking in some type of betrayal, and the other partner finds out. Now we have two people living in close quarters who have a strong animus to each other. This is hatred – a much more unstable situation than it was before they knew of each other’s existence. This leads to immediate separation (fragmentation) to the point where they are far apart again.
Bonding = love
Non-bonding = indifference
Antibonding = hatred
Quiz Yourself!
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
(Advanced) References and Further Reading
- The Kinetics of Decomposition of Benzoyl Peroxide in Solvents. I
Kenzie Nozaki and Paul D. Bartlett
Journal of the American Chemical Society 1946, 68 (9), 1686-1692
DOI: 1021/ja01213a002
Benzoyl peroxide (which is often used in face and body washes to treat acne) is a common initiator for free-radical reactions in organic chemistry. Upon heating, the O-O bond homolytically cleaves to give 2 benzoyloxy radicals. This paper shows that the half-life of this decomposition is around 4-5 hrs at 79.8 °C, which is quite conveniently near the boiling point of benzene or cyclohexane. Additionally, oxygen, which is also a triplet diradical in its ground state, acts as an inhibitor of free-radicals, and so must be rigorously excluded when carrying out a free-radical reaction. The typical protocol is to dry the solvent thoroughly (e.g. with sodium), freshly distill it, and reflux it under nitrogen/Ar for an hour or two prior to adding the reagents and substrates. Alternatively, ‘freeze-pump-thaw’ methods can be used for degassing the solvent. - The Photosensitized Decomposition of Peroxides
Cheves Walling and Morton J. Gibian
Journal of the American Chemical Society 1965, 87 (15), 3413-3417
DOI: 1021/ja01093a022
Light can also be used to initiate radical reactions by inducing homolytical cleavage of the O-O bond in benzoyl peroxide. However, a photosensitizer must be used for peroxides. Photosensitizers absorb and transfer the light energy to another molecule. In this case, aromatic ketones (e.g. benzophenone, which forms a ketyl radical readily) are used as photosensitizers. - Effects of solvent on the unimolecular decomposition of t-butyl peroxide
Earl S. Huyser and Richard M. VanScoy
The Journal of Organic Chemistry 1968, 33 (9), 3524-3527
DOI: 1021/jo01273a036
The activation enthalpy of homolysis of t-butyl peroxide is measured here and shown to be solvent dependent, varying from 31 kcal/mol in acetonitrile to 40 kcal/mol in cyclohexane (at 125 °C). This is consistent with a process which requires significant heating above room temperature for initiation.
In comparison to benzoyl peroxide, t-butyl peroxide undergoes homolysis at >100 °C. It was initially reported that the rate of t-butyl peroxide is the same in the gas phase as in solution, but certain solvents with OH groups can increase the rate of decomposition. - Reality of solvent effects in the decomposition of tert-butyl peroxide
Cheves Walling and Douglas Bristol
The Journal of Organic Chemistry 1971, 36 (5), 733-735
DOI: 1021/jo00804a030
These authors reexamine the results from Ref. #4, and find that the rate constants for decomposition of t-butyl peroxide in various solvents are also concentration dependent. They also obtain different values for the activation enthalpy of homolysis in acetonitrile (34.2 kcal/m3l) and cyclohexane (38.4 kcal/mol). - Uses of Isotopes in Addition Polymerization
G. Ayrey
Chemical Reviews 1963, 63 (6), 645-667
DOI: 10.1021/cr60226a005
Radical reactions are also widely used in polymer chemistry; some of the most well-known polymerization mechanisms are based on radicals. A common criticism of radical chemistry is that “transformations involving radical intermediates have long harbored the reputation of being difficult to control, suitable only for the synthesis of polymers and tars”. - Solvent Effects in the Decomposition of 1,1′-Diphenylazoethane and 2,2′-Azobis-(2-methylpropionitrile)
Raymond C. Petersen, J. Hodge Markgraf, and Sidney D. Ross
Journal of the American Chemical Society 1961, 83 (18), 3819-3823
DOI: 1021/ja01479a021
Azo compounds are also commonly used for initiation of radical reactions – these will thermally decompose to give N2 and the respective radicals. - Free Radical Addition to Olefins to Form Carbon-Carbon Bonds
Walling, Cheves; Huyser, Earl S.
Organic Reactions 1963, 13, 91-149
DOI: 10.1002/0471264180.or013.03
Organic Reactions, maintained by the ACS Division of Organic Chemistry, is a useful source of comprehensive reviews in Organic chemistry topics. This review has a section on free-radical reaction initiation. AIBN (azobis(isobutyronitrile)) is a widely used initiator of free-radical reactions. It is a crystalline substance which decomposes upon heating into isobutyronitryl radicals and nitrogen with a half-life of about an hour at 80 °C. As with most other unimolecular fragmentations, the half-life of AIBN diminishes rapidly with increasing temperature – it drops to only a few minutes at 110 °C, the temperature of refluxing toluene (see p. 113 for a graph of half-life vs. temperature of common free-radical initiators). - Triethylborane-Mediated Atom Transfer Radical Cyclization Reaction in Water
Hideki Yorimitsu, Tomoaki Nakamura, Hiroshi Shinokubo, and Koichiro Oshima
The Journal of Organic Chemistry 1998, 63 (23), 8604-8605
DOI: 1021/jo981774p
Another common initiator for free-radical reactions is triethylborane (Et3B). Upon exposure to oxygen, this releases ethyl radicals, which can initiate a chain reaction. The advantage of using Et3B as an initiator is that high temperatures can be avoided – room temperature (or lower) becomes possible. The authors in this paper demonstrate a convenient method for initiating a reaction with Et3B – the reaction flask can be equipped with a toy balloon filled with Ar, and O2/air can slowly diffuse into it and hence the reaction medium. This paper also shows that water can be used as a solvent for radical reactions, in contrast to reactions involving strong electrophiles!





For the plot of note 2, should we double the length of arrow of △E so that it start from HOMO and ends in LUMO?
thanks so much, I really very like your article because this article is very informative for me and is also provide very deep and correct information about light and light energy.
v minor – but you seemed to have used ‘gamma’ instead of ‘nu’ for representing energy/light.
Yes, thank you. Fixed
Just wanna say thanks :)
Glad you found the article useful, Taha.
I bumped into your explanation while searching an answer to the question “why homolytic (why not heterolytic)?” And I can’t find the kind of answer I’m seeking.
Let me have another crack at it. Homolytic cleavage usually happens when you’re breaking a bond between two identical atoms which have a weak bond, such as Br-Br or RO-OR. They have equal electronegativity and therefore the bond isn’t polarized toward one of the atoms. The result is that the bond breaks such that the electrons are distributed symmetrically.
If heat can excite an electron from HOMO to LUMO for radical reactions, why can’t heat excite an electron from HOMO to LUMO in a diene?
The role of heat in these cases is to fragment a weak bond such as O-O or Br-Br .
Heat, by itself, may excite an electron from HOMO to LUMO in a diene, but this method can lead to a lot more side reactions than if one were just to use light.
Hello,
What if there is presence of alkene with halogen and no light?
Probably a different reaction then.
what an analogy!!! i will never forget this. thank you
Oh you big tease! You get so close, ever so close to free-radical polymerization, and then you stop short and withdraw.
You even tempted me further by the thoughts of recombination, something that AIBN is exceedingly well known for as a polymerization initiator. But do you let me carry out my polymerization reactions to my satisfaction?
I can’t take this anymore. We either have to go all the way next time or I’m outta here.
Excellent analogy! Worth to share with the students next time we cover MOs. Thank you!