Spectroscopy
Homotopic, Enantiotopic, Diastereotopic
Last updated: May 28th, 2026 |
Homotopic, Enantiotopic, and Diastereotopic Groups: What Does It Mean?
When you have two hydrogens attached to a single carbon, they can have three different types of relationships. We call them “homotopic”, “enantiotopic”, and “diastereotopic”.
To determine these relationships we imagine replacing each hydrogen in turn with a different atom or group, which we can call D.
- When replacement of each H with D results in the same product, we call these hydrogens homotopic.
- When replacement of each H with D results in enantiomers, we call these hydrogens enantiotopic.
- When replacement of each H with D results in diastereomers, we call these hydrogens diastereotopic.
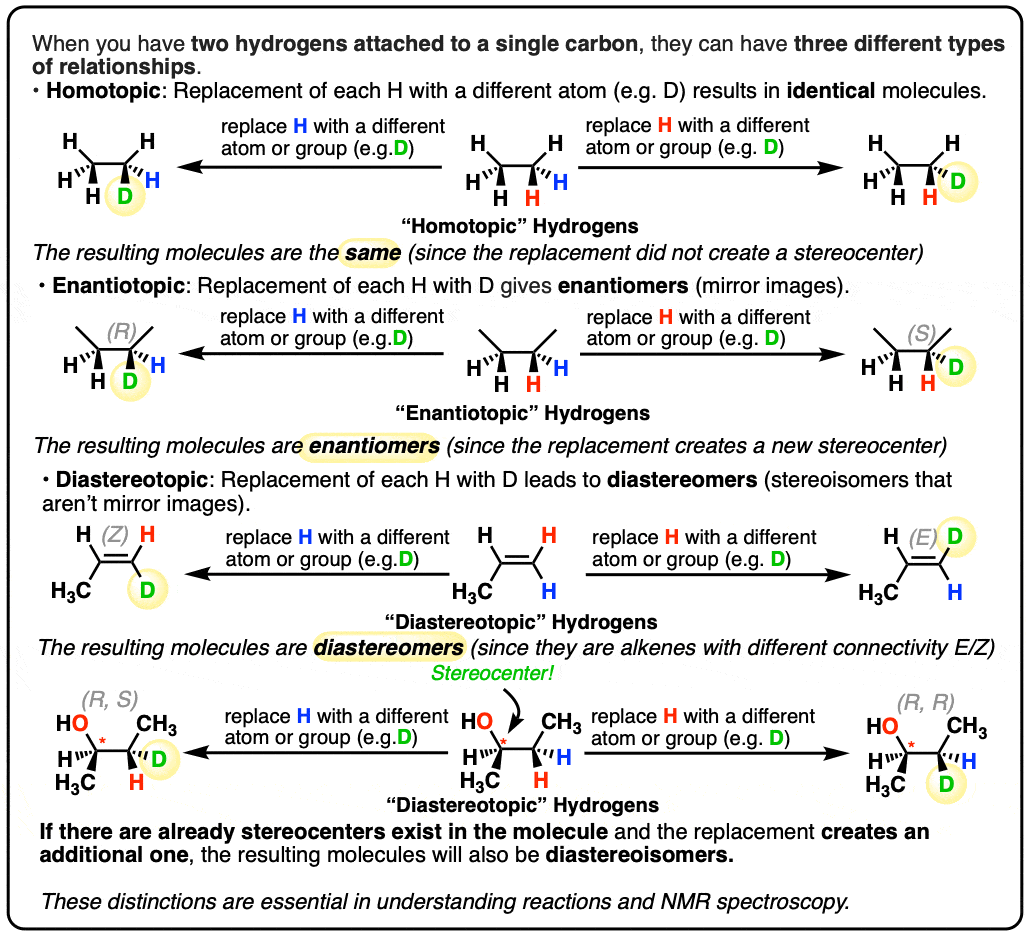
Table of Contents
- When Is “Homotopic, Enantiotopic, Diastereotopic” Important?
- Homotopic Atoms (or Groups)
- Enantiotopic Atoms
- Diastereotopic Atoms
- When Does It Matter?
- Notes
- Quiz Yourself!
1. When Is “Homotopic, Enantiotopic, Diastereotopic” Important?
- Certain reactions directly replace hydrogens with other atoms. For example, free radical chlorination replaces C-H bonds with C-Cl bonds. So understanding these principles help in understanding what potential types of products you could obtain from these reactions.
- In Nuclear Magnetic Resonance (NMR) these relationships determine whether or not these hydrogens are in the same “chemical environment”. In other words, whether or not they have the same or different signals.
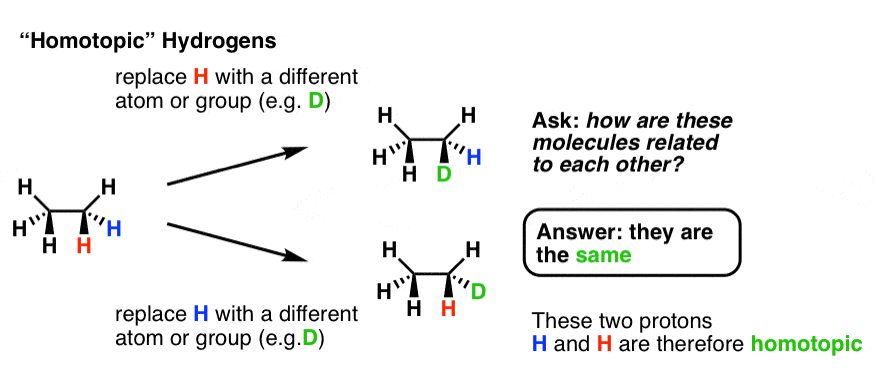
2. Homotopic Atoms
Take a molecule like ethane. Let’s label (with color) two different hydrogens, blue and red. Next, let’s replace each of these hydrogens in turn with a different atom. In this example it could be deuterium (D) but really this can be done with any atom or group (except hydrogen of course).
Replace the red H and the blue H in turn with D and compare the molecules that are formed. Ask: how are these molecules related?
In this case they are both deuterioethane. Since the two molecules are the same, the two hydrogens are said to be “homotopic”. Replacement of either gives rise to the same product.
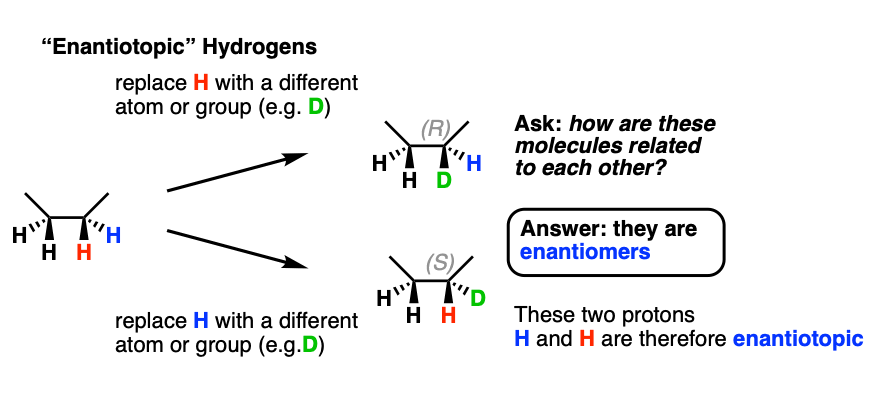
3. Enantiotopic Atoms
Let’s look at butane next; specifically, the second carbon of butane. Replacement of the red H with D leads to (R)-2-deuteriobutane, while replacement of the blue H with D leads to (S)-2-deuteriobutane. These hydrogens are therefore not homotopic. Since enantiomers are obtained here, these two protons are therefore enantiotopic.
Note that the CH3 protons of butane are homotopic; it’s only the C-2 (and C-3) hydrogens of butane that are enantiotopic.
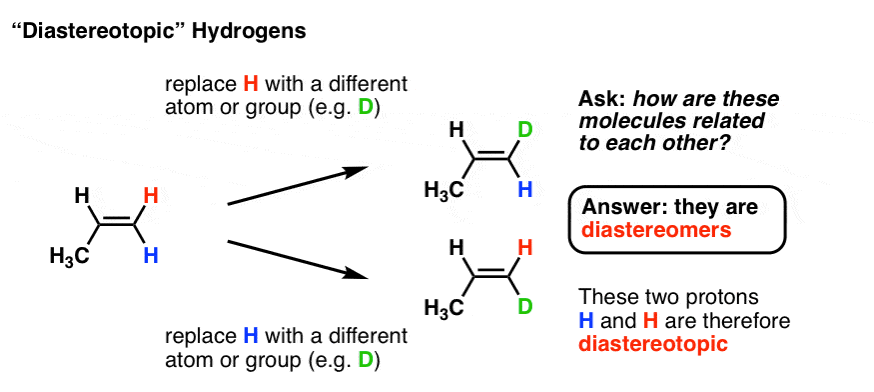
4. Diastereotopic Atoms
It’s also possible to have diastereotopic protons. Look at the alkene below. Replacement of the red H with D leads to the E-alkene, while replacement of the blue H with D leads to the Z-alkene. What’s the relationship between these two compounds? They’re diastereomers – stereoisomers, but not mirror images. So the two protons are said to be diastereotopic.
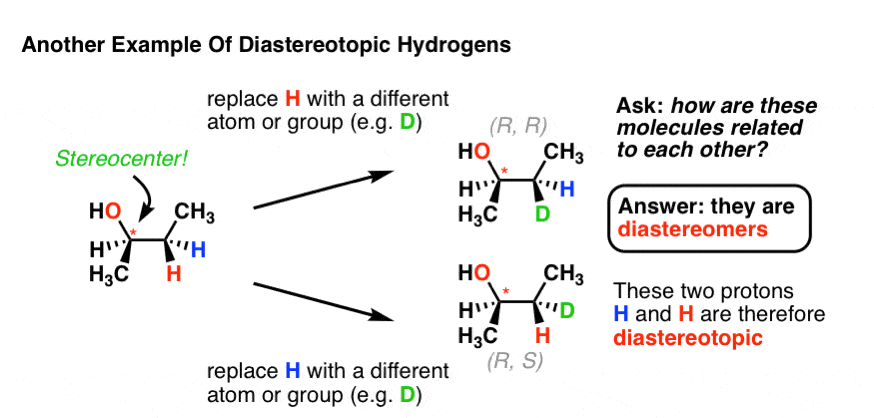
There’s another potential situation which can lead to diastereotopic protons. Look at the molecule below – (R)-butan-2-ol. Replacement of the red H leads to the (R, R) product. Replacement of the blue H leads to the (R, S) product. Therefore, these two products are diastereomers, and the two protons are diastereotopic.

5. When does it matter?
Two situations:
- In free radical chorination – say, of butane – on the second carbon (C-2), substitution of C-H with Cl will lead to a mixture of stereocenters. It’s important to recognize when this can happen.
- (Most common) – In NMR spectroscopy:
- homotopic protons have the exact same chemical shift
- enantiotopic protons have the same chemical shift in the vast majority of situations. However, if they are placed in a chiral environment (e.g. a chiral solvent) they will have different chemical shifts.
- diastereotopic protons have different chemical shifts in all situations
Notes
Quizzes
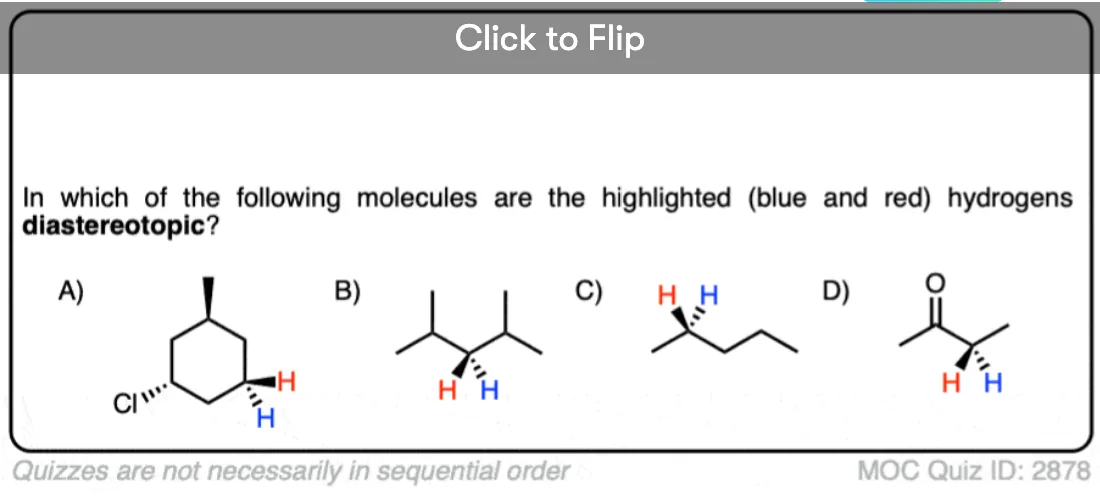
Become a MOC member to see the clickable quiz with answers on the back.
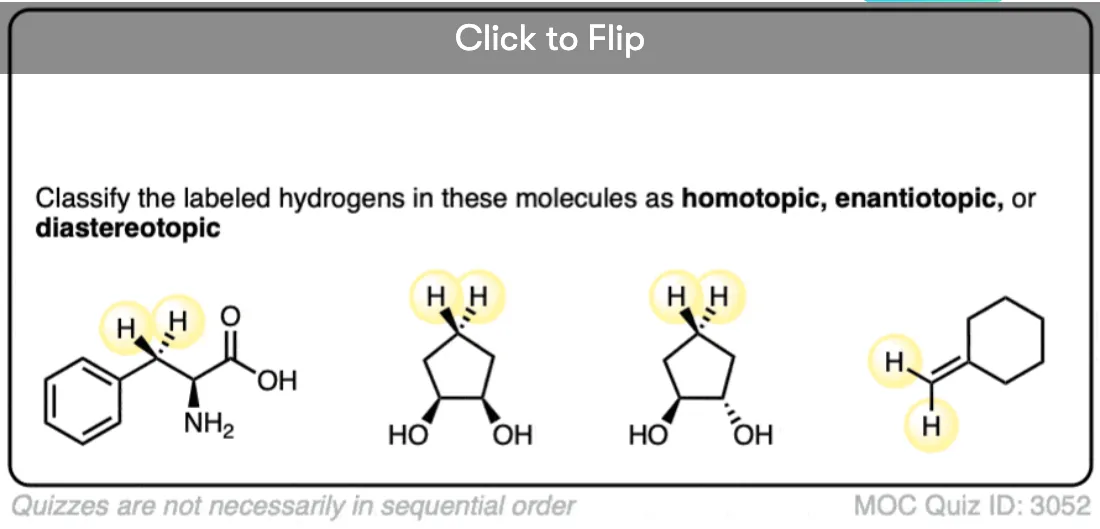
Become a MOC member to see the clickable quiz with answers on the back.
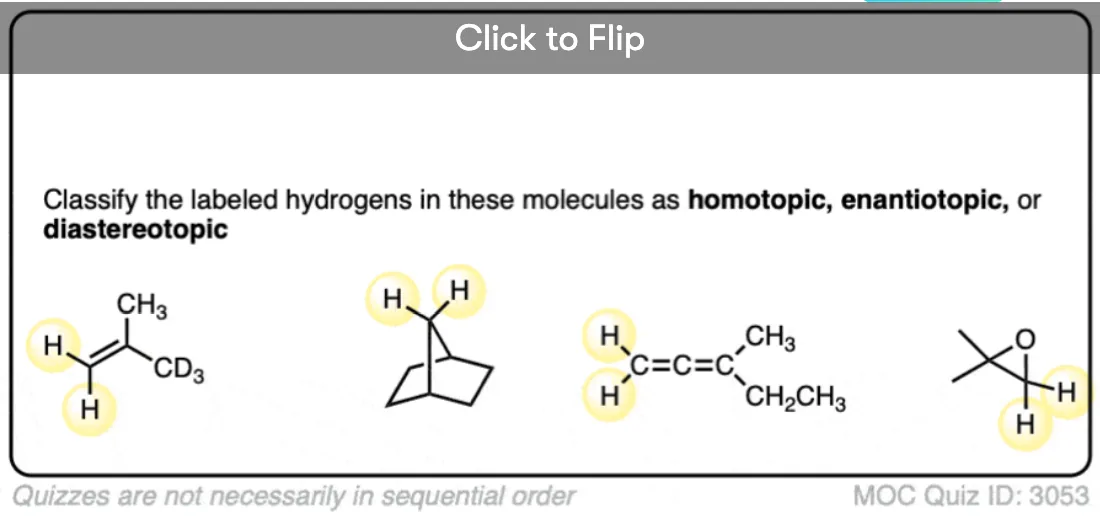
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.


Whenever i tap into this specific page it kicks my account out for some odd reason making me unable to check the quiz.
Thank you. Looking into this.
Enantiotopic and disteriotopic atom and face
This was so brief, simple, and understanding
the article is straight forward, thanks a lot I’ve benefited
For meso compounds, can they still have diastereotopic protons?
Certainly. To take a simple example, (R,S)-1,2-cyclohexane diol. Every methylene (i.e. CH2) carbon in that molecule bears a pair of diastereotopic protons.
It’s really helpful, clearly described..I want some examples on diastereotpic faces of alkenes with symmetry and addition criterion
Yes – thank you, and you are right, it does refer to an earlier version!
Very clear descriptions but your Enantiotopic figure is described as butane but is still ethane so the figure itself is Homotopic. Also your descriptions often are of a chlorine replacement but the figures are still of deuterium (an earlier version?).
I bought your spectroscopy pdf but haven’t had a chance to read it yet…. but your site has been so helpful figured I try it!
Fixed. Thank you George!
Thank you!! I liked it… short ,, sweet & clear!
Alright. Thanks for letting me know Malith!
can someone highlight me on si- face and re- face with some examples, am stack
Good topic for a future post.
Nice explanations, as a chemistry student am happy this topic was difficult but here I find it easy and educative
OK, thank you Devon.
How do you determine two proton chemical shift in diastereotopic mixture by 1H-NMR (threo-erythro)?
Do the protons have to be adjacent to a chiral center in order for them to be diastereotopic (like in example 2)? Or can they be further down the chain or anywhere else in the molecule.
Nope, in theory they could be 100 carbons down the chain or anywhere else in the molecule.
It’s really helpful… But it will be more if we get some easy and common examples
Thank you so much for all of your articles. They’re so clear and well-written, and as a science student at mcgill I can confirm that you’re a lot easier to understand than many of my org chem profs! We’re learning IR/NMR/MS identification stuff right now and I’m up to my neck in confusing slides and this article was the best thing that’s ever happened to me!
Now I understand this clearly.. Thank you
OK, great Ramesh!
if those examples are illustrated the it would be very nice
THANK YOU SO MUCH! You are awesome!!
Hi, thanks for the examples on homotopic, enantiotopic and diastereotopic but i don’t get the difference between the enantiotopic and second example of diastereotopic. I will really appreciate it if you could explain it to me. Thanks. :)
We look at the hydrogens in pairs. An enantiotopic pair of protons will produce different chiral configurations on substitution. Looking at the example above, substitution of one of the protons creates an R center; substitution of the other creates an S center. We either have an R center or an S center. These are both opposite chiral configurations of each other, which means enantiomers.
For the diasteretopic set, let’s start with the first example of a double bond. Chirality doesn’t apply to a single double bond, so going like above, upon substitution of either hydrogen we end up with two stereoisomers–the Z and E configurations. To confirm they are stereoisomers, try flipping either isomer around, and you will see you can’t get them to match. Simply establishing that stereoisomers are created on a substitution of a single double bond means we have diastereotopic protons.
Moving on to the second example of diastereotopic protons: we are examining the proton set next to the chirality center. Diastereomers are two molecules that have the same formula, but their chirality centers have different configurations. If their configurations are completely opposite, we have enantiomers. In the case above, we end up with either an R,S or R,R product of chlorobutanol. These are two molecules with the same formula, but their chirality configurations are different; hence, they are diastereomers. If they were enantiomers, they would need to be complete opposites, eg. an R,S product and an S,R product.
So to check for diastereotopic protons, the first thing you do is substitute each proton in turn and compare the resulting products. Remember your goal specifically is to identify a product’s chirality. If you have diastereomers (different chiral configurations), they are diastereotopic; if enantiomers (opposite chiral configurations), enantiotopic; if the same molecule (no change in chiral configuration), homotopic.
epic response. Thanks Blaise.
Hi! This definitely helped- I have a quiz on this stuff today, so this will hopefully help me out! One suggestion- maybe you could include heterotopic ones as well? I get a little confused with those, and we’re learning about those in tandem with the ones you have listed. Great Website!
Definitely put together otherwise confusing NMR terms well. I’ll be sure to direct my junior level chemists to this next time it comes up.
Good suggestion. thanks!
This is a good explanation of a topic that several PhD students in my group could definitely grasp a bit better…
Finally, we dip our toes into NMR!
I actually don’t recommend my students use chlorine in their substitution exercise… The molecule might already have a chlorine in it – confusing the situation. Do the substitution exercise on C2 of 1-chloroethane… then what happens?
I use some ridiculous transition metal (for personal reasons, my favorite choice is element 76)… or I have them do the substitution exercise with something nonsensical like a smiley face. We can still name the results and compare their relationship… It’s (R)-2-smileyfacebutane and (S)-2-smileyfacebutane.
:)
The foolproof method is to use something achiral that’s not even an element, like Q. Or a cat… :-P
But the letter Q is chiral… (was it you who tweeted the achiral letters a while ago?)
And as James has shown, cats can be chiral, too…
/troll
dipping a little end of my pinky toe into NMR. Still reluctant to talk about it yet.
thanks for the suggestion re: replacing Cl.
Deuterium works well for me (:
Yes, good point.
use Deuterium…