Home / The Marriage May Be Bad, But the Divorce Still Costs Money
Organic Chemistry Tips and Tricks
The Marriage May Be Bad, But the Divorce Still Costs Money
Last updated: January 23rd, 2024 |
Common misconception: energy is released through the breaking of bonds.
Not correct.
Energy is released through the FORMATION of chemical bonds. Formation of a new bond between two atoms results in a net lowering of energy of the atoms involved, corresponding with a release of energy (heat) to the environment. If there wasn’t some kind of attraction to begin with, the chemical bond would never have formed in the first place.
It costs energy to break a bond. Even if the bond is weak.
For instance, the oxygen-oxygen bond of hydrogen peroxide is one of the weaker bonds you’ll encounter, with a bond dissociation energy of 142 kJ/mol.
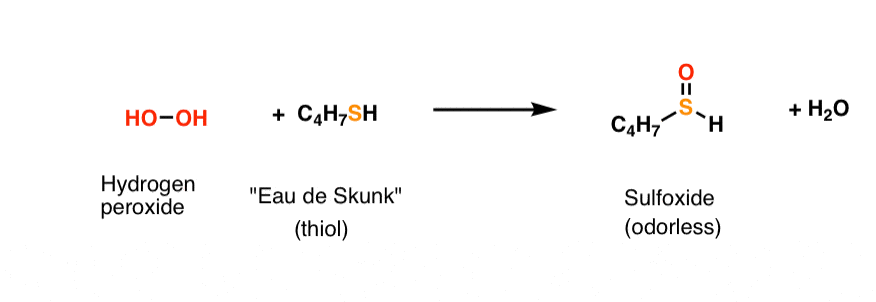
As you might be aware, hydrogen peroxide is useful for the de-skunkification of dogs: the hydrogen peroxide oxidizes the stinky thiols to odorless sulfoxides. [Note – despite what you might have heard, the tomato juice method is pure B.S.]. The peroxide-thiol reaction is extremely favorable, resulting in the formation of a strong S-O bond (bond strength 522 kJ/mol).
Here’s the reaction:
Notice that even though we’re breaking a weak bond like the oxygen-oxygen bond of HO-OH to form a strong bond like S-O, it still costs energy to break the O-O bond.
“Expenses”: breakage of O–O bond. 142 kJ/mol
“Income”: formation of S-O bond 522 kJ/mol
Energy profit/loss = expenses – income -380 kJ/mol (negative denotes exothermic)
So energy is released by the overall process, but breaking the weak bond in the first place still consumes energy.
The marriage of two oxygen atoms in a peroxide molecule may be a relatively unhappy one, but they still have to pay expenses to get a divorce.
00 General Chemistry Review
01 Bonding, Structure, and Resonance
02 Acid Base Reactions
03 Alkanes and Nomenclature
04 Conformations and Cycloalkanes
05 A Primer On Organic Reactions
06 Free Radical Reactions
07 Stereochemistry and Chirality
08 Substitution Reactions
09 Elimination Reactions
10 Rearrangements
11 SN1/SN2/E1/E2 Decision
12 Alkene Reactions
13 Alkyne Reactions
14 Alcohols, Epoxides and Ethers
15 Organometallics
16 Spectroscopy
17 Dienes and MO Theory
18 Aromaticity
19 Reactions of Aromatic Molecules
20 Aldehydes and Ketones
21 Carboxylic Acid Derivatives
22 Enols and Enolates
23 Amines
24 Carbohydrates
25 Fun and Miscellaneous
26 Organic Chemistry Tips and Tricks
- Common Mistakes: Formal Charges Can Mislead
- Partial Charges Give Clues About Electron Flow
- Draw The Ugly Version First
- Organic Chemistry Study Tips: Learn the Trends
- The 8 Types of Arrows In Organic Chemistry, Explained
- Top 10 Skills To Master Before An Organic Chemistry 2 Final
- Common Mistakes with Carbonyls: Carboxylic Acids... Are Acids!
- Planning Organic Synthesis With "Reaction Maps"
- Alkene Addition Pattern #1: The "Carbocation Pathway"
- Alkene Addition Pattern #2: The "Three-Membered Ring" Pathway
- Alkene Addition Pattern #3: The "Concerted" Pathway
- Number Your Carbons!
- The 4 Major Classes of Reactions in Org 1
- How (and why) electrons flow
- Grossman's Rule
- Three Exam Tips
- A 3-Step Method For Thinking Through Synthesis Problems
- Putting It Together
- Putting Diels-Alder Products in Perspective
- The Ups and Downs of Cyclohexanes
- The Most Annoying Exceptions in Org 1 (Part 1)
- The Most Annoying Exceptions in Org 1 (Part 2)
- The Marriage May Be Bad, But the Divorce Still Costs Money
- 9 Nomenclature Conventions To Know
- Nucleophile attacks Electrophile
Oh , Thanks a lot ..I love your posts.
super good explanation!! thank you I was honestly getting more and more confused by other things i was reading online but this cleared it up
I love this blog! I didn’t actually think that chem could sound interesting but it does in the way that you write it. Thank you so much!
Thanks!
I love your posts. I might not leave comments often enough but I read every single one of yours !!! Keep going, the chemical world turns with a better spin since you started posting !!!!