Acid Base Reactions
A Handy Rule of Thumb for Acid-Base Reactions
Last updated: September 30th, 2025 |
Reversible And Irreversible Acid-Base Reactions
Last time we learned about pKa and how it’s the closest thing we have to a universal measurement of the strengths of all kinds of different acids and bases.
I also referred to a post on how to use a pKa table (key lesson: stronger acid plus stronger base gives weaker acid and weaker base).
We can use pKa to estimate the equilibrium constants for acid-base reactions.
A handy rule of thumb for an acid-base reaction is that if the acid and the conjugate acid are separated by more than 10 pKa units, the reaction is essentially irreversible.

Table of Contents
- An Irreversible Acid-Base Reaction: Strong Acid (HCl) Plus Strong Base (NaOH) Giving Water
- An Easily Reversible Acid-Base Reaction: Methanol (pKa 15.2) With Water (pKa 14)
- At What Point Does An Acid-Base Reaction Become Irreversible For Practical Purposes?
- Application: In The Claisen Condensation, The Starting Ester Is Less Acidic Than CH3OH By About 10 pKa Units
- Notes
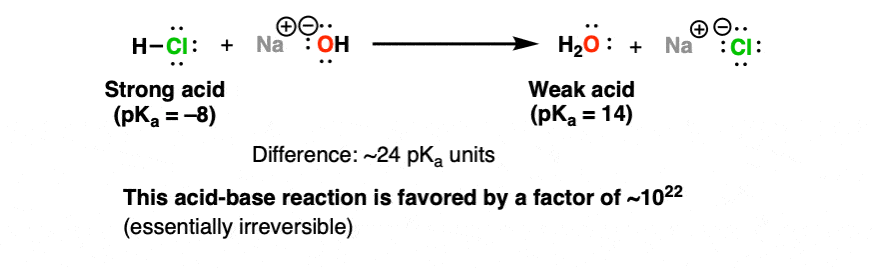
1. An Irreversible Acid-Base Reaction: Strong Acid (HCl) Plus Strong Base (NaOH) Giving Water
On one extreme, we have one mole of a really strong acid – let’s say hydrochloric aid (HCl), pKa –8. And to it, we add (slowly!) a solution of water containing one mole of sodium hydroxide (the conjugate base of water, pKa 14).
HCl and NaOH react to give water and NaCl . How favorable is this reaction? We can make a rough estimate. The pKa of HCl is -8. Sodium hydroxide is the conjugate base of H2O (pKa 14).
That’s a difference of about 22 pKa units – and since each pKa unit represents one order of magnitude, this reaction is favorable with an equilibrium constant of about 10 to the power of 22.
For all intents and purposes, a reaction with an equilibrium constant this huge is irreversible.
That is to say that HCl and NaOH are completely consumed when they react together, giving only H2O and NaCl.
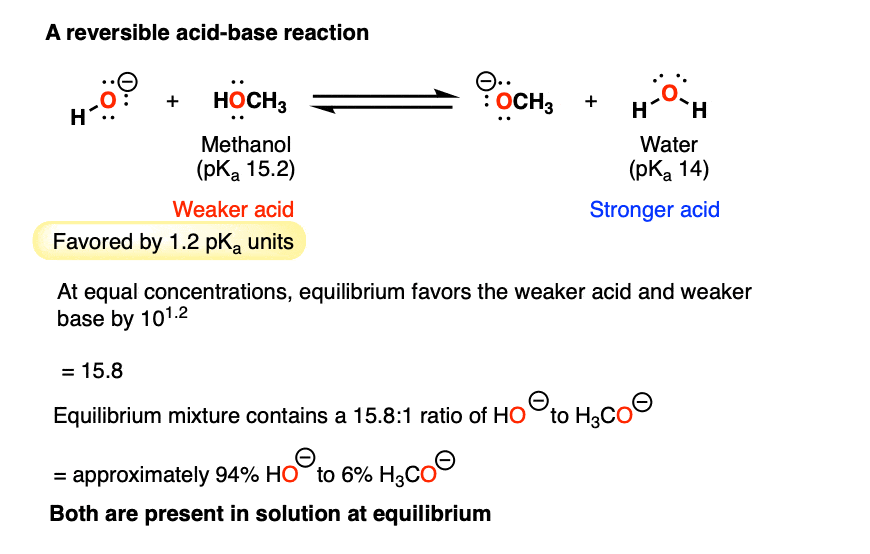
2. An Easily Reversible Acid-Base Reaction: Methanol (pKa 15.2) With Water (pKa 14)
What about the other extreme: the reaction of methanol (pKa of 15.2) with sodium hydroxide (the conjugate base of water, pKa 14)?
Neither side of the acid-base reaction is strongly favored. Here we’re dealing with a very small difference in pKa – only 1.2 pKa units.
So the equilibrium constant here would only be about 10 to the power of 1.2 —> 15.8 toward giving the weaker acid (CH3OH) and the weaker base HO(–).
At equilibrium we’d expect to have a mixture of about 94% HO(-) [the weaker base] and 6% H3CO(-) [the stronger base].
In other words, both species are present in solution.
3. At What Point Does An Acid-Base Reaction Become Irreversible For Practical Purposes?
So how far can we stretch this? In between these two extremes, at what point does a reaction become irreversible for practical purposes?
There’s no hard and fast rule on this. But for practical purposes, a good rule of thumb is about 10 pKa units.
That is to say, if the difference in pKa‘s between an acid and a base (actually, the conjugate acid of the base) is about 10 pKa units or less, it is useful to consider their acid-base reaction to be in equilibrium.
Think about what that means – a ratio of one molecule in 10 billion can make the difference in a reaction!
One in 10 billion might not sound like a lot. But when you consider that a mole contains 10 to the power of 23 molecules, and each of them are colliding millions of times per second, the odds aren’t really as bad as they look.
f your only chance of buying a private jet rested on you winning a Powerball lottery – but you were able to fill out hundreds of thousands of entries per second, every second, you’d be at the G5 dealer by next Tuesday.
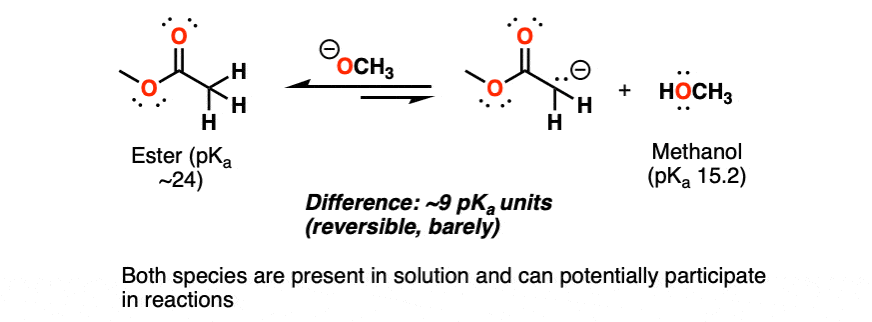
Here’s an example you’ll see in Org 2. The Claisen condensation begins with the deprotonation of an ester (pKa ~24) by an alkoxide ion (conjugate base of an alcohol, pKa ~15). (See article:The Claisen Condensation)
That’s disfavored by about 10 to the power of 9, since we’re going from a weaker base (alkoxide) to a stronger base (deprotonated ester, a.k.a ester enolate).
Even though there’s only a small amount of the deprotonated ester present at equilibrium, this can be enough to get the reaction to go! You can take my word for it – that this rule of thumb applies – and leave it there.
Or if you’d prefer to go through an actual application of this concept, I’ll finish up with that.
4. Application: In The Claisen Condensation, The Starting Ester Is Less Acidic Than CH3OH By About 10 pKa Units
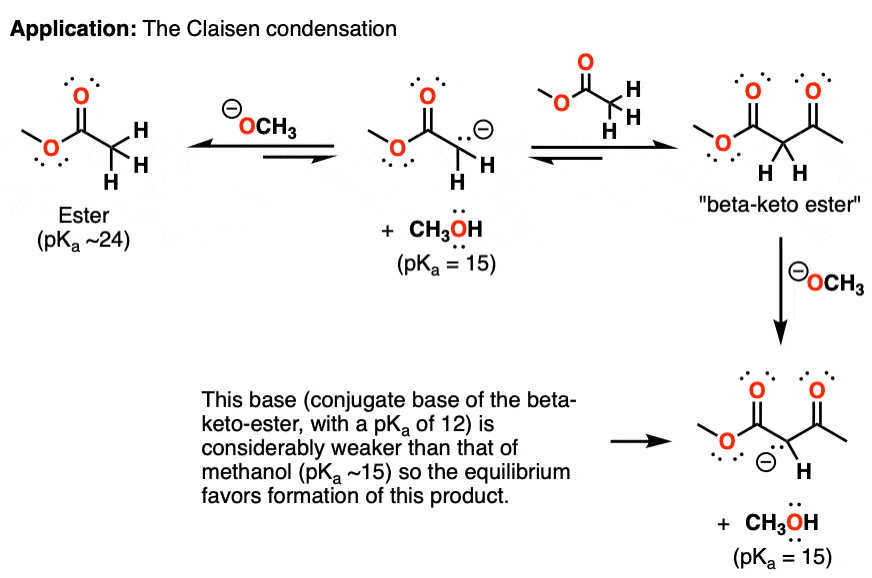
One application of this concept can be found in the Claisen condensation of esters. The Claisen condensation involves the addition of a deprotonated ester (an “enolate”) to another equivalent of an ester, through an addition-elimination reaction (see article: The Claisen Condensation)
The first step is deprotonation of the ester by an alkoxide ion [in this case CH3O(-) ] as mentioned above. This enolate can then attack a second equivalent of ester, which then eliminates an equivalent of alkoxide ion.
This reaction is also potentially reversible. However, the protons of the new product – a “beta-keto ester” – are considerably more acidic than those of the starting ester, and an acid-base reaction between it (pKa 12) and alkoxide (pKa 15) is quite favorable.
The equilibrium eventually favors the final product, because the conjugate base of the beta-keto ester (pKa 12) is considerably weaker than methanol (pKa ~15).
In other words, even though the first step is extremely disfavored, this is made up for by the fact that there is a very good “driving force” for the subsequent reaction.
Next Post: Acid-Base Reactions Are Fast
the “claisen condensation product” in summary table there should be Methyl acetoacetate is it? the enolate compound do substitution nucleophile to other Methyl acetate that produce Methyl acetoacetate instead Dimethyl malonate, like the explanation that show us
Oh, you are correct. Good catch. Thank you!
In the second point you have stated that H2O is the weaker acid and CH3O- is the weaker base of the reaction I believe which is not true as it can be observed that the pka of water(14) is less than the pka of methanol(15.2) so, water is definately stronger acid than methanol. Because of this I am not able to properly grasp what is given in the second point can you please explain.
This has been fixed. Thank you for bringing it to my attention!
In the Water and methanol reaction, isn’t methanol (pka – 15.2) a weaker acid than water (pka-14)? Shouldn’t the equilibrium favor methanol??
This was a mistake. Fixed. Thanks!
These posts are really helpful – thank you. In the graphic showing a Claisen condensation reaction, isn’t the final product CH3OH, rather than CH3O? Else, where has the H gone? Maybe I missed something..
Thanks
Corrected this typo. Thanks for letting me know!
The reaction of -OH and MeOH would favor the left side not vice versa. MeOH is a weaker acid than H2O and thus MeO- is a stronger base. The equilibrium would favor methanol and not water.
Fixed this mistake. Thank you.
BJ
I normally find no problem with any of the posts on this website (its beautifully laid out organic chemistry material.)
but I do agree that I find some irregularities in the information.
I think this might be a ploy to make us really think hard about the numbers and discern for ourselves whether the information presented is correct or not.
the irregularity in this post that bothered me was the reversible reaction between methanol and hydroxide to create h20 and methyloxide anion.
if we use the pka values of 14 for water and 15.2 for methanol, the reaction should proceed right to left, from stronger acid to weaker acid.
Therefore, the ratio of 15.8:1, if thats even the correct ratio, should be of hydroxide to methyloxide, not the way it has been written there.
Also, an interest note:
My organic chemistry textbook says the pKa of water is 15.7; this post says 14. Another irregularity.
So naturally, I googled, and the consensus seems to be that 15.7 is wrong, and 14 is correct.
So, my textbook is not infallible. Awesome.
I fixed the typo with respect to the favorability of the acid-base reaction. Thanks.
After doing more research I think I understand your post better but I still have questions.Please correct me on anything I write below that is incorrect.
1) all reactions are reversible- meaning all reactions have an equilibrium( technically even the reactions we say are irreversible and go to completion are reversible, but we treat them as only going one way( with the single arrow) because the Ka is so large.)
2.)Further, the forward and reverse reactions never stop occurring but at some point there is no longer an observable change in the concentration of reactants and products. When this has happened we say the reaction has reached a state of equilibrium.
3.)The extent to which starting material is converted to product is governed by thermodynamics (changes in energy). It is favorable for a system to go from high energy to low energy. Therefore, the side that is lower in energy is favored at equilibrium.
4.) We use Keq to describe the reaction at equilibrium, so
Keq( Ka for acid)=[products]/[reactants]
If Keq>1 then [products] > [reactants].Therefore, since this is the case it means the products are energetically favored( meaning they are lower in energy and thus more stable). Therefore, the equilibrium would have a larger arrow pointing towards the right and a smaller arrow pointing towards the left in a reaction.
If Keq<1 then [products]< [reactants].In this case, because there are more reactants this indicates they are favored energetically and are thus more stable.Therefore, the equilibrium will favor the reactants over products.This leads to a bigger arrow pointing towards the reactant and a smaller pointing towards the products.
If Keq=1 then the [products]=[reactants].Neither is more favored than the other ,so the arrows would be equal in length and point in opposite directions.
Now to your post info.
1.)The first part- I assume when you use your rule of thumb you imply that any difference in the pKa's of an acid and conjugate acid greater than 10 pKa units is an irreversible reaction? I see the method you used in the practice problems to get 10^22.But couldn't you use 10^deltapKa(meaning 10 ^pKa(conjacid)-pKa(acid).)
2.)For the second part I keep getting -1.2 pKa difference when using your method.So, what gives? Is the pKa difference considered as an absolute value? Also, where are you getting all those other numerical values in this part like the 15.8 to 1 ratio and such?
3.)Third part- Based on your handy rule, can we assume every acid /base rxn that has about 10 or less pKa difference between the acid and conj acid will always be considered or treated as an equilibrium reaction?
Lastly,In the third part I get -9pKa difference with the final reaction. Why do I get a negative value when I figure out the difference but you get a positive value? Finally, you say the reaction is "disfavored by about 10 to the power of 9, since we’re going from a weaker base (alkoxide) to a stronger base ." Does this mean the reaction is disfavored if we look at it going in the forward direction?
I may be wrong in my analysis of this section,so please correct me if I am wrong.
1.) highly favorable acid /base reactions will have differences of pKa greater than 10.So, if you see a difference of pKa between an acid and the conjugate acid over 10 you know the reaction is highly favorable and will go to completion.
2.)Any acid/ base reaction with a pKa difference between the acid and conjugate acid of about 10 or less is a reaction considered to be in equilibrium.Because the reaction is in equilibrium both sides of the reaction occur even if the reaction is not very favorable.The trick then is to figure out which side of the equilibrium is more favored than the other.
I realize I may be wrong, so please correct me with any other analysis you see fit to use.
When you say, “difference in pKas between an acid and a base (actually, the conjugate acid of the base),”does this mean we are comparing the pKa’s of the acid on the left and the conjugate acid on the right?
This is the first article of the acid/base info that I have found to be confusing.I guess I am blind /ignorant, but I can’t seem to get all of the basic gist of what is being said here. The first part talks about an irreversible reaction. I can see there is a 10 ^22 difference in the magnitude between their pKa’s.(In the Acid/Base questions you use a technique of Keq=Ka(reactant)/Ka(product)=10^-pKa/10^-pKa. Where does this idea come from and can you use it with any acid/ base problem?)
Does the large magnitude of 10^22 imply that the reaction is so extremely favorable that it must go to completion?
For the second part I am totally lost.Why is this a reversible reaction at all? Why does it even proceed at all when we are going from weaker acid/weaker base to stronger acid/ stronger base?I thought reaction only proceeds when you have stronger acid /stronger base to weaker acid/ weaker base?Or does this idea only relate to how favorable the reaction is or not?How do we know equilibrium exists here?( I assume its based on the handy rule of thumb below?)And why is weaker acid/ base favored at equilibrium?
P.S.The technique you use on the Acid/base questions doesn’t seem to work out as I keep getting a negative number of 10^-1.2.
You say,” if the difference in pKas between an acid and a base (actually, the conjugate acid of the base) is about 10 pKa units or less, it is useful to consider their acid-base reaction to be in equilibrium.” So, I assume anything greater than a 10 pKa difference would be an irreversible reaction ,correct?Does this also mean that any acid/conj acid difference that less than a 10pKa will always proceed and be in equilibrium?
Any help would be appreciated.A short concise bottom line version of this page would help too.
Is methanol is stronger acid than water ?
In many books the pKa of water is listed as 15.7. However there seem to be several errors in how this number was arrived at. I have corrected earlier versions of this post which had that value and replaced them with a value of 14, so no, methanol is not a stronger acid than water.
I don’t understand. In previous post you said that if you do an acid-base reaction and products are stronger base/acid than reactants reaction won’t happen. Methanol has a pka of 15.2, water 14, so water (product) is stronger acid than methanol. Therefore methoxide (product) is stronger base than OH-. Why would than reaction happen?
Fixed. Thank you.
What if the pka difference between an acid and base species is significantly greater than 10. I know the reaction would be irreversible but is it practical? (Say for instance, Na+ Cl- and CH4)
Generally not practical for pKa difference of >10.
Absolutely not practical for NaCl and CH4. We’re talking 55 orders of magnitude, at least. If you waited around from the beginning of the universe until today, you might see not even see it happen.
NaOH is not the conjugate base of H2O. NaOH is not a Bronsted species at all.
HO- is the conjugate base of H2O.
In practice we label NaOH as a strong base. In fact it is a strong electrolyte and contains the strong base HO-. Rigorously, HO- is not a strong base, it is at the border between weak and strong bases.
NaOH is not the conjugate base of H2O. NaOH is not a Bronsted species at all.
HO- is the conjugate base of H2O.
In practice we label NaOH as a strong base. In fact it is a strong electrolyte and contains the strong base HO-. Rigorously, HO- is not a strong base, it is at the border between weak and strong bases.
Let’s keep going down the pKa table, then. Terminal alkynes? Weak acids. Acetylide ions are strong bases. Secondary amines? Weak bronsted bases. Their conjugate bases are strong bases. Alkanes are extremely weak acids, alkyl anions are extremely weak bases. I can’t see how “the conjugate base of a weak acid is a weak base” holds up.
I had some confusion about the pKa’s of Methanol and Water… I expected the Methoxide ion to be more unstable than the Hydroxide ion.
But some google search suggests that pKa values are not constant across the solvents…
In water, Methanol is less acidic than water but in DMSO, its vice versa!
Definitely confusing.
Dear James, thanks for posting but I still have some confusion. If strong acid and strong base react to give weak acid and weak base, how do you know which direction the equilibrium lies?
In your first example, you have HCl (stronger acid) forming water (weaker acid). Couldn’t I just as easily say you have water (stronger base) and HCl (weaker base). That would imply that HCl formation is favored. Obviously, this is not true so what am I doing wrong here? Are there any general rules using pKa or pKb that help predict this? Thanks for any insights on this.
Hi – the difference is that in the reverse direction, the identities of the “acid”, “base”, “conjugate acid” and “conjugate base” would switch.
Acid: the species where the bond to H breaks
Base: the species where the bond to H forms
Conjugate acid: what becomes of the base once it’s added the bond to H
Conjugate base: what becomes of the acid once it’s lost the bond to H
If you were to draw the reaction between H2O and NaCl giving HCl and NaOH:
Acid: H2O
Base: Cl-
Conjugate acid: HCl
Conjugate base: NaOH
The conjugate acid (HCl, pKa -8) is clearly stronger than the acid (H2O, pKa 15). Therefore this reaction would not proceed.
It’s easier to make the judgement call with pKas since those measurements are readily available. We don’t generally deal with pKb’s.
Thanks for the response. If I understand correctly, HCl has lower pKa so its a strong acid meaning Cl- must be a weak conjugate base. H2O has higher pKa so its a weak acid meaning OH- must be a strong conjugate base. In your example, the strong acid and strong base are both on the reactant side of the equation while the weak acid and weak base are on the products side. And thermodynamics says the reaction equilibrium lies to the weak side?
I’m confused about the hydroxide-methanol reaction you gave. Since methanol is a weaker acid than water, shouldn’t methoxide be a stronger base than hydroxide? I seem to remember that alkoxides are stronger bases than hydroxides from my organic chemistry class. If you could please explain this, that would be great.
Thanks,
Jordan
Nevermind, I misread the pKas, sorry for the confusion.
Thank you for this post it was very informative.
One thing that I found lacking however was what happens, for example, if you did a 2:1 (not equal) methoxide/methanol to water solution? What ratio would you get then?
Thank you!
I don’t think you could make a solution that concentrated. If you calculate the molarity of the “solution” formed by, say, starting with 1 L of methanol (791 g, or 24 moles) you would have to add you would have to add 48 moles of NaOMe (2.6 kg). I don’t think it can be done.
Does that answer your question?