Conformations and Cycloalkanes
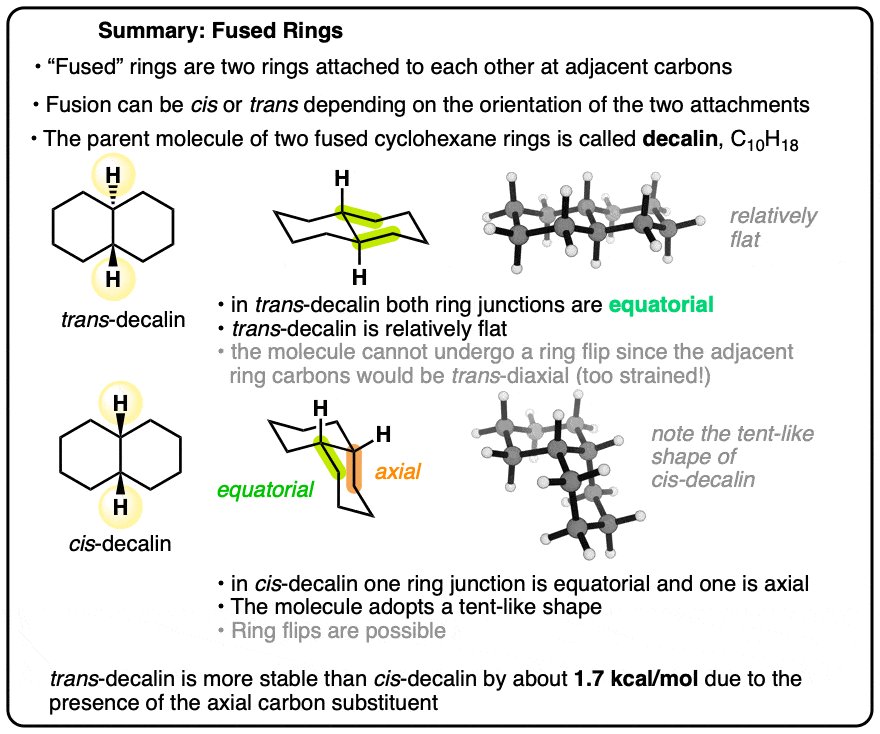
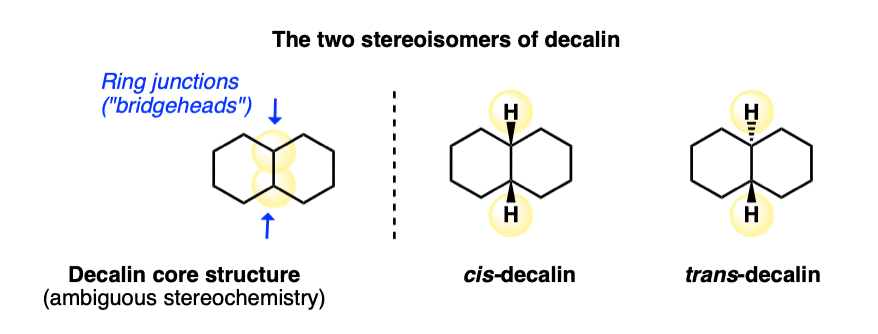
Fused Rings: Cis-Decalin and Trans-Decalin
Last updated: May 30th, 2026 |
Why Is trans-Decalin More Stable Than cis-Decalin?
At the beginning of this series I said that the fact that carbon can form rings leads to all kinds of interesting consequences. We’re going to see many examples of that in our post today!
So far, we’ve only talked about cyclic molecules containing one ring. But, of course, molecules with multiple rings are very common in nature. A prime example is the steroid structure, exemplified by the common oral contraceptive ethinyl estradiol.
Here, we’re just going to talk about two very simple cases of molecules containing two rings. We’re going to focus our efforts on cyclohexane, and examine in close detail the situation where two cyclohexane rings are bonded to each other in the most common way – with the two ring junctions (“bridgeheads“) on adjacent carbons, a situation we refer to as fused rings.
Table of Contents
- cis-Decalin and trans-Decalin
- A Model of cis-Decalin Shows It Has A Tent-Like Shape
- A Model of trans-Decalin Shows It To Be Flat
- Why Is trans-Decalin More Stable Than cis-Decalin? All Carbons Are Equatorial
- cis-Decalin Has Two Gauche Interactions
- Doing A “Ring-Flip” On cis-Decalin
- trans-Decalin Cannot Undergo Ring Flips. It Is Conformationally “Locked”
- Summary: cis-Decalin versus trans-Decalin
- Notes
- Quiz Yourself!
- (Advanced) References and Further Reading
1. cis-Decalin and trans-Decalin
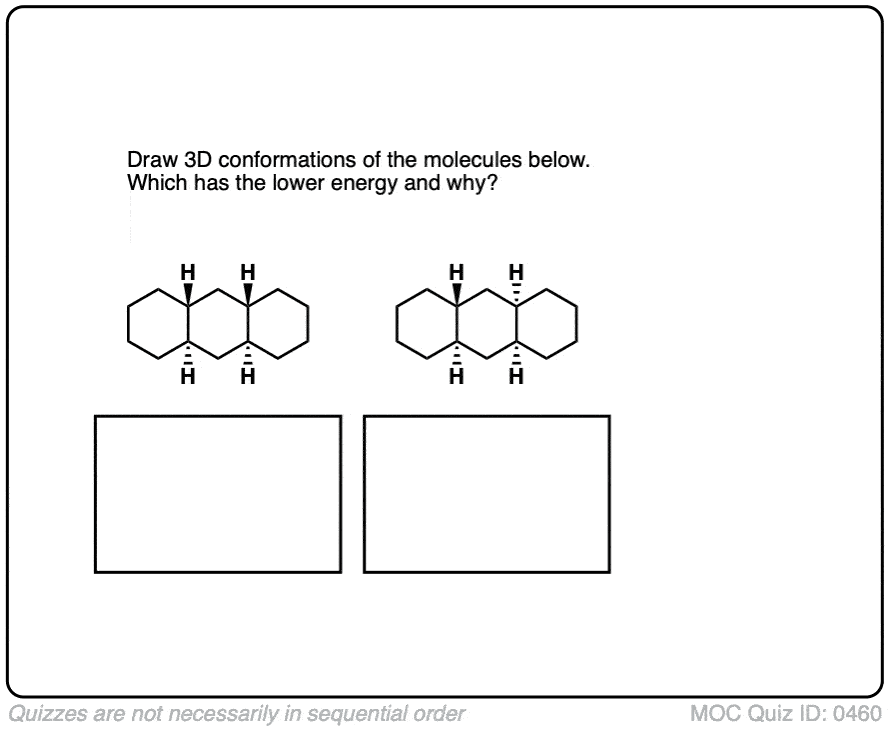
The parent molecule is called “decalin” since there are ten carbons in total. However, remember that any time we have two substituents on a cyclohexane ring (as we do here), it is essential to draw in the stereochemistry in order to avoid ambiguity! Two stereoisomers are possible here: one where the hydrogens at both ring junctions are “cis”, and the other where they are “trans”.
These molecules look so simple when we draw them on paper! What could be simpler than two hexagons joined together?
Well, just like with cyclohexane itself, the complexity comes when we examine their most stable three-dimensional structures. Each six membered ring will adopt a chair conformation. Believe it or not, the cis and trans stereoisomers of decalin have remarkably different shapes.
There’s no better way to see this than by making a model.
2. A Model Of cis-Decalin Shows It Has A Tent-Like Shape
Here’s cis-decalin. The hydrogens on the “bridgeheads” are highlighted in red and white. Notice how both cyclohexanes are in chair conformations, but the molecule adopts almost a tent-like shape.
3. A Model of trans-Decalin Shows It to Be Flat
And here’s trans-decalin. As we look at the molecule from an angle, it’s obvious that both cyclohexane rings are also in the chair conformation, but the molecule is much flatter overall.
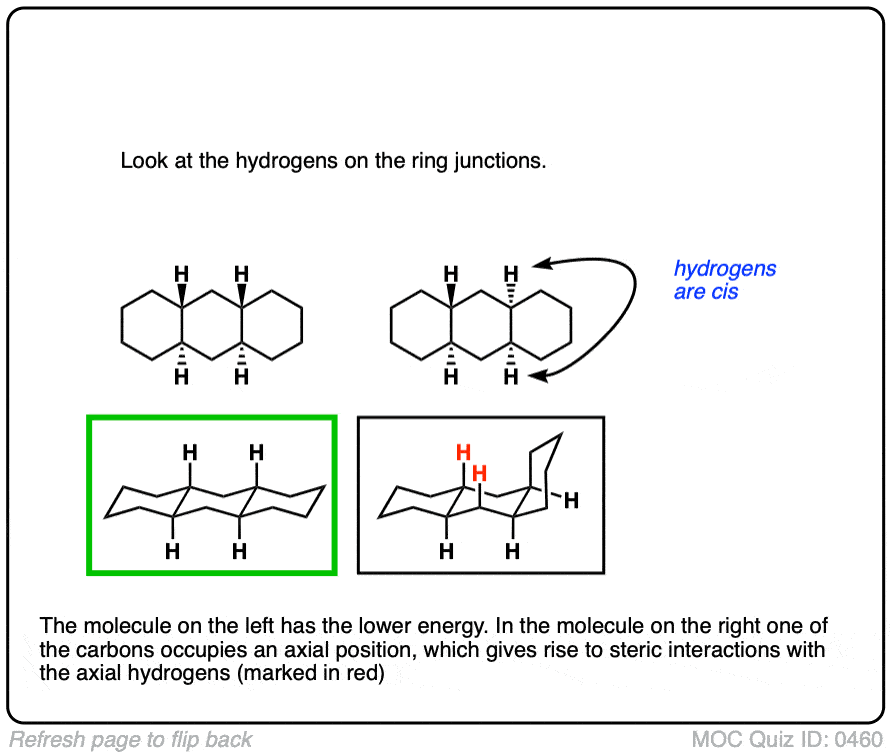
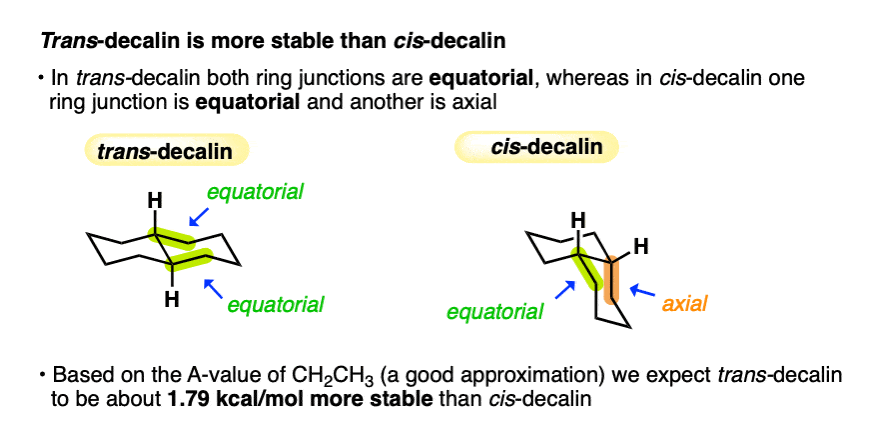
4. Why Is trans-Decalin More Stable than cis-Decalin? All Carbons Are Equatorial
As these movies show, changing the stereochemistry of the bridgehead carbon from a “wedged” H to a “dashed” H can make a huge difference in the overall shape of the molecule!
Let’s look at these two molecules in a bit more detail. First of all, which one should be more stable, cis-decalin or trans-decalin?
First of all, let’s look at line drawings of each of these molecules. When we do so, we notice that in trans-decalin, all carbons are equatorial, whereas in cis-decalin, one carbon is equatorial and the other is axial.
Based on the (safe) assumption that the A-value of the axial group should be about 1.75 kcal/mol, we can therefore make a judgement about the relative stability of these two molecules.
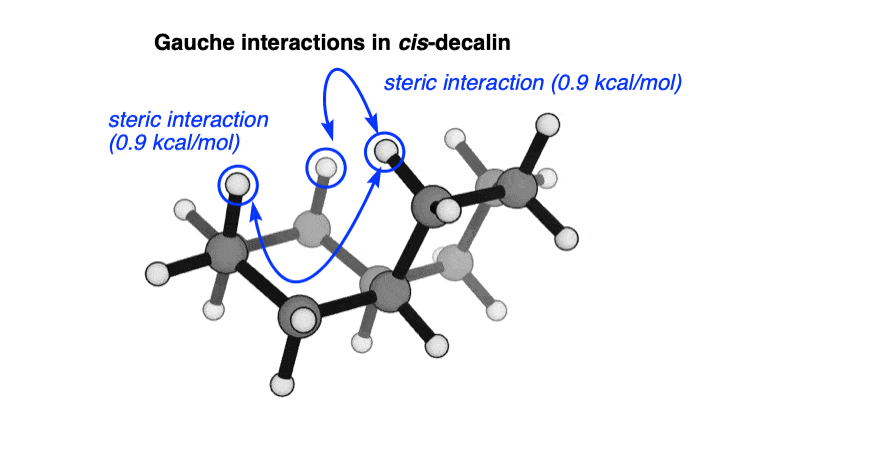
5. cis-Decalin Has Two Gauche Interactions
Just to nail the point home, when we look at cis-decalin from the side, we see the two gauche interactions (each worth about 0.9 kcal/mol) that are responsible for this increase in energy (this is a screenshot of the cis-decalin “flyover” video, above)
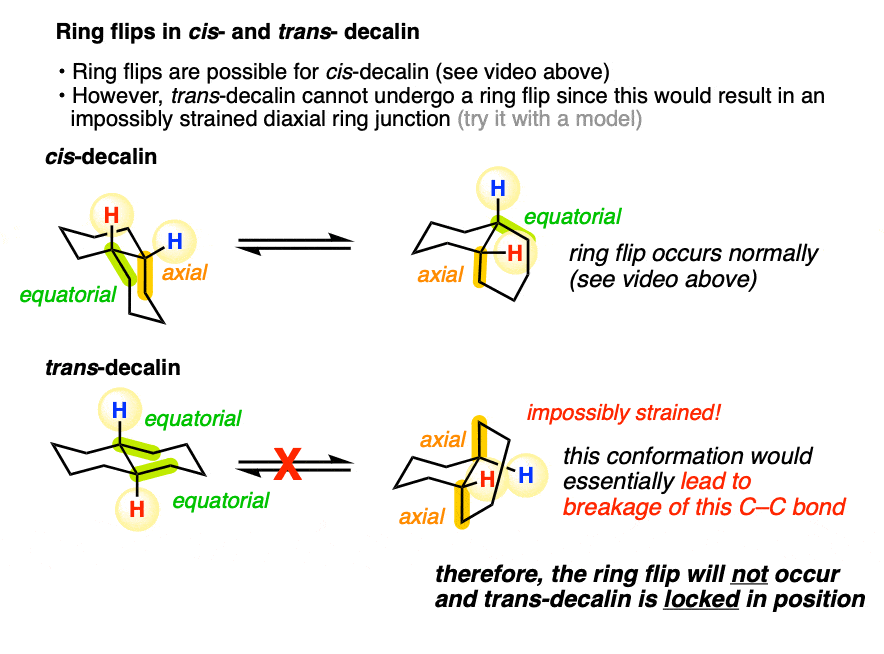
6. Doing A “Ring-Flip” On The cis– Isomer
OK. So we’ve established what cis and trans decalin look like, and which are more stable – but hold on, you might say. What about ring flipping? Does that change anything?
After all, when we do a ring flip on a cyclohexane, all equatorial groups become axial and all axial groups become equatorial. Will this affect any of the relative energies?
Let’s look at cis-decalin first. We have one group equatorial and one group axial. If you said that a ring flip on this molecule would lead to a conformer exactly equal in energy, you are correct!
Here’s a video of the “ring-flip” of cis-decalin in action. Looking at the left-hand cyclohexane, notice how the red (axial) hydrogen becomes equatorial, and the white (equatorial) hydrogen becomes axial. [BTW the cyclohexane on the right is meant to end up looking more like a chair – it turned out a bit flatter than I would have liked]
7. trans-Decalin Cannot Undergo Ring Flips: It Is Conformationally “Locked”
What about trans-decalin? Shouldn’t a ring flip convert both equatorial groups to axial groups, and therefore be higher in energy?
In theory, it’s reasonable to think, “yes” – and in situation where we’re dealing with rings larger than 8 or so, that is what happens. However, this is a prime example of an unexpected consequences I was talking about earlier.
It turns out that a ring flip in trans-decalin isn’t possible because it would lead to too much ring strain. To use an analogy, the reason why trans-decalin can’t do a ring flip is a bit like why you can’t touch your nose to your spine – there just isn’t enough flexibility to do so without putting yourself in tremendous pain.
A different way of saying the same thing is that trans-decalin is locked in position.
So while cis-decalin can undergo ring flips, trans-decalin cannot.
8. Summary: cis-Decalin versus trans-Decalin
In the next post we’ll talk a little bit about some other situations that arise when we have molecules with multiple rings.
Next Post: Bridged Bicyclic Rings
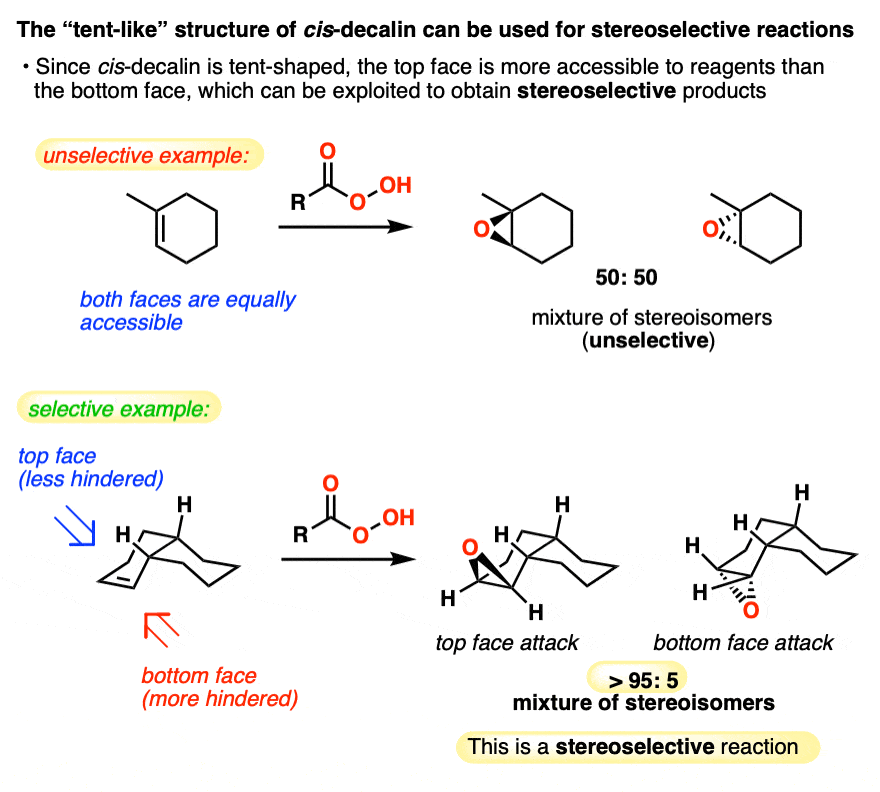
 Click to Flip
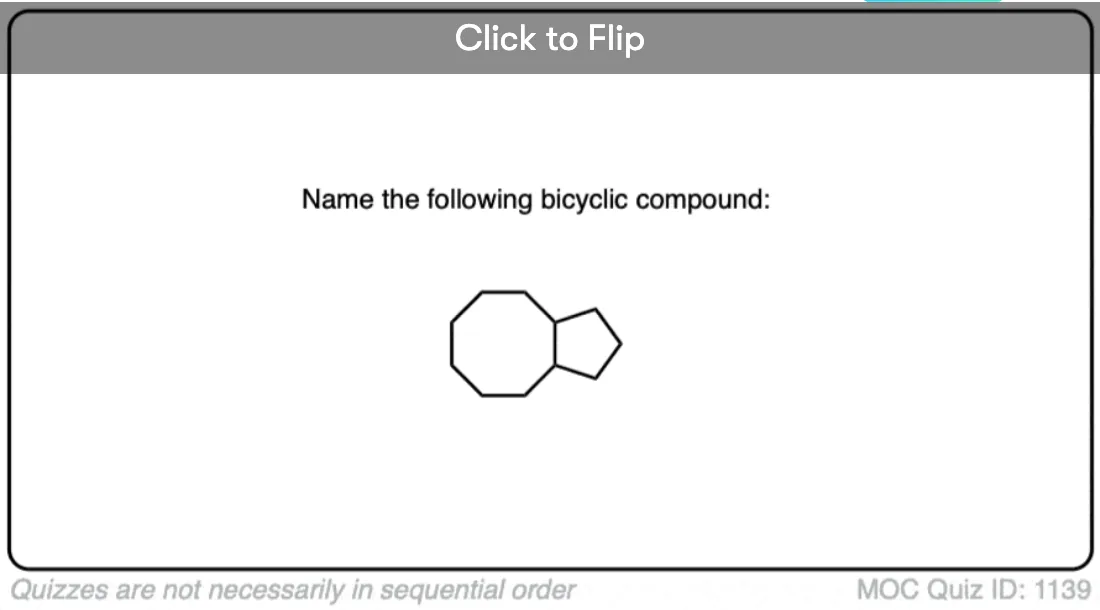
Click to Flip
Notes
Note 1. The ‘tent-like’ shape of cis decalin (see pic below) has a further important consequence that has been exploited by clever organic chemists for decades. The two faces of cis-decalin are not equivalent. The top face is more “exposed” to the surrounding environment than is the bottom face – in other words, a molecule encounters much less steric hindrance in approaching the top face than it does the bottom face (note how crowded the underside is).
An example will serve to illustrate.
Imagine that we have a decalin with an alkene. There are plenty of useful reactions of alkenes, as we talk about in this series.
To take one example, we can form an epoxide by treating an alkene with a peroxyacid such as m-chloroperoxybenzoic acid.
One example is its reaction with 1-methylcyclohexene, which gives rise to two stereoisomers – enantiomers, in this case [i.e. non-superimposable mirror images] in a 50:50 ratio because both faces of 1-methylcyclohexene are equally accessible. In other words, it is unselective. [I chose 1-methylcyclohexene and not cyclohexene because reaction at either face of cyclohexene gives rise to the same molecule (it has a plane of symmetry) and the issue of selectivity is moot]
If we have a decalin with an alkene, such as the molecule shown below, one face will be more reactive than the other because it is more accessible [in order to react at the bottom face, the peroxyacid would encounter significantly more steric hindrance].
So when we treat this alkene with our peroxyacid, we get a mixture of epoxides that will be selective for one stereoisomer over the other (“stereoselective”). In this case we are forming a mixture of diastereomers, so we say that this reaction is “diastereoselective” (a subclass of “stereoselective”)
For a classic example of how this can be used in target molecule synthesis, see Woodward’s synthesis of reserpine.
Note 2. Some calculations on trans- and cis- decalin, courtesy of Rowan Labs.
Trans-Decalin:
Cis-Decalin
Quiz Yourself!
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
(Advanced) References and Further Reading
- Die Baeyersche Spannungstheorie und die Struktur des Diamanten
Ernst Mohr
Prakt. Chem. 1918, 98 (1), 315-353
DOI: 10.1002/prac.19180980123
Mohr predicted in 1918 that decalin, which has puckered six membered rings, should exist as both cis and trans isomers. - Zur Stereochemie bicyclischer Ringsysteme. I. Die Stereoisomerie des Dekahydronaphthalins und seiner Derivate
Walter Hückel
Lieb. Ann. Chem. 1925, 441 (1), 1-48
DOI: 10.1002/jlac.19254410102
Two planar six membered rings as pictured by Baeyer could only have a cis junction. The pivotal demonstration of the existence of two decalin stereoisomers helped to convince organic chemists that cyclohexane is not planar. The separation of cis and trans decalin by distillation (8 degrees difference) was also achieved, demonstrating that they are two separable, independent compounds. - Physical Chemical Properties of cis- and trans-Decahydronaphthalene
F. Seyer and R. D. Walker
Journal of the American Chemical Society 1938, 60 (9), 2125-2128
DOI: 10.1021/ja01276a028
An early paper rigorously recording and documenting the physical properties of cis and trans-decalin. - The molecular structure of cis- and (trans-bicyclo [4.4.0] decane in the gas phase, studied by electron diffraction and molecular mechanics
Van Den Enden, H. J. Geise, A. Spelbos
J. Mol. Struct. 1978, 44 (2), 177-185
DOI: 10.1016/0022-2860(78)87026-4
A combined theoretical and experimental study of the structures of cis and trans decalin, showing that both cyclohexane rings are in the chair form in both isomers. - Conformational inversion rates in the dimethylcyclohexanes and in some cis-decalins
David M. Grant, Don K. Dalling, and LeRoy F. Johnson
Journal of the American Chemical Society 1971, 93 (15), 3678-3682
DOI: 1021/ja00744a021
For those interested in the power of NMR spectroscopy in structural analysis, this is a very nice paper. Using VT (variable-temperature) 13C NMR, the authors were able to determine that the barrier to ring inversion for cis-decalin is 12.6 kcal/mol at RT. Interestingly, two of the authors are from Varian Associates, one of the pioneers in NMR manufacturing. This paper also came out at the time when NMR technology had just advanced to the point where taking 13C NMRs of unenriched samples was practical. Varian was acquired by Agilent in 2010, and in 2014, Agilent shut down the NMR division of Varian, bringing an end to a chapter in chemical history. - THE TOTAL SYNTHESIS OF RESERPINE
R. B. Woodward, F. E. Bader, H. Bickel, A. J. Frey, and R. W. Kierstead
Journal of the American Chemical Society 1956, 78 (9), 2023-2025
DOI: 10.1021/ja01590a079 - A SIMPLIFIED ROUTE TO A KEY INTERMEDIATE IN THE TOTAL SYNTHESIS OF RESERPINE
R. B. Woodward, F. E. Bader, H. Bickel, A. J. Frey, and R. W. Kierstead
Journal of the American Chemical Society 1956, 78 (11), 2657-2657
DOI: 10.1021/ja01592a102 - The total synthesis of reserpine
R. B. Woodward, F. E. Bader, H. Bickel, A. J. Frey, R. W. Kierstead
Tetrahedron 1958, 2 (1-2), 1-57
DOI: 10.1016/0040-4020(58)88022-9
These three papers are on the total synthesis of reserpine, by the legendary Prof. R. B. Woodward, among the greatest organic chemists of all time. An early Diels-Alder reaction creates the fused ring system with a cis decalin, which is used to set the stereochemistry in the downstream steps.

For instability of cis-decalin, you have mentioned that there are two gauche interactions. How many gauche interactions are there in trans-decalin? When I drew the Newman projections, there are 3 gauche interactions in cis and 2 are in trans. But I may be wrong.
What shape would a trans decalin be that had a double bond opposite where the rings fuse? And would this have an impact on epoxide formation (using a peroxide) and subsequent reduction of the epoxide to an alcohol?
I’m not sure which trans decalin alkene you’re referring to, but to answer your second question, opening of epoxides happens in a very specific way – it occurs so that both substituents will end up axial so the half-chair can relax into a boat. It’s called the Furst-Plattner rule. https://en.wikipedia.org/wiki/F%C3%BCrst-Plattner_Rule
I have the same modeling kit.
How did you connect the middle carbons ( the carbons that create the bridge) in a cis confirmation?
Remember that when two adjacent substituents on a ring are “cis”, one is axial and the other is equatorial. Takes some time to get used to, but they are on the same side!
It’s completely explained topic in best way. Thanks
Glad to hear, thank you for stopping by, Komal.
Thank you! This is wonderfully helpful!
I was wondering what model kit you use. They look easy to manipulate bonds around.
Thanks!
They are Darling models. https://www.darlingmodels.com/index.php?dm=true
Bro the video doesn’t help.
In cis decalin having one methyl group substitution which conformer is more stable?
Where are you putting the methyl group?
If you drew a chair and a flipped chair connected together would that also be a trans decalin? Also would another reason that trans is more stable is that the methyl groups are going in different directions (one in and one out) so there is less interaction when compared to the cis decalin?
I’m assuming you’re talking about the isomer of dimethyl decalin where there are methyl groups on the ring junctions. Yes, with cis decalin those methyl groups would have a dihedral angle of 60 degrees to each other (staggered) whereas in trans decalin they would be staggered.