Conformations and Cycloalkanes
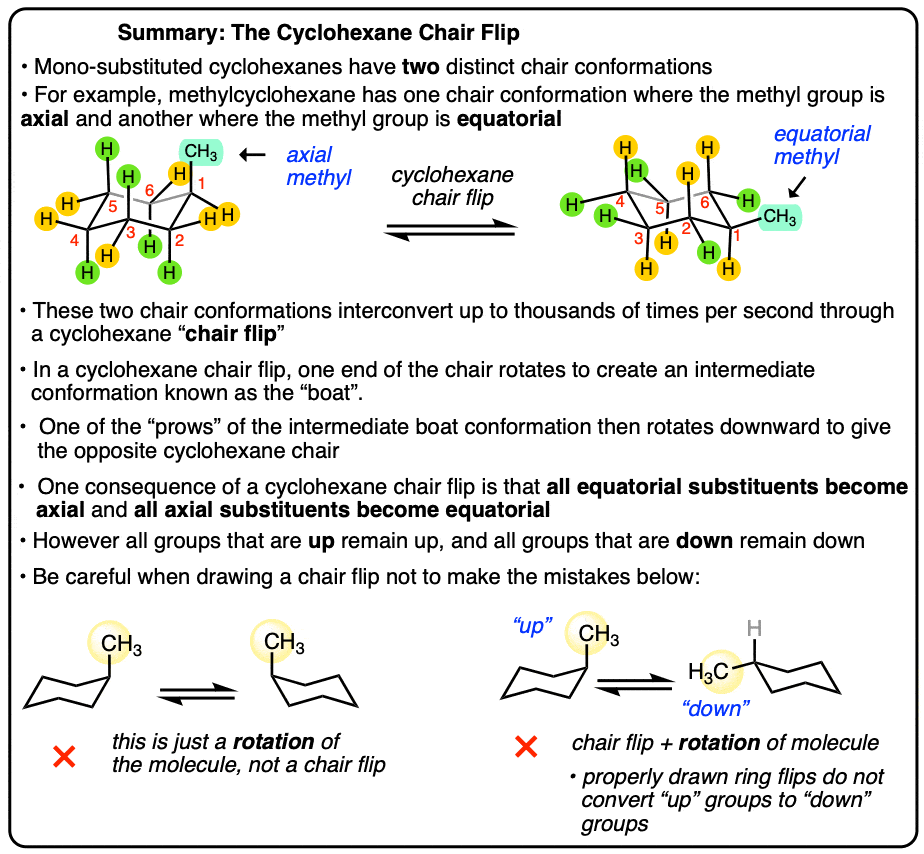
The Cyclohexane Chair Flip
Last updated: May 30th, 2026 |
The Cyclohexane Chair Flip
All about how to draw the product of a cyclohexane chair flip – and also, three ways not to!
- The Two Chair Conformations of 1-Methyl Cyclohexane
- How Do These Conformations Interconvert? Through A Cyclohexane “Chair Flip”
- Step One: Bring Up A “Footrest” Of The Chair To Make a Cyclohexane “Boat”
- Step Two: Pull Down The Opposite “Headrest” Of The “Boat” To Make a Cyclohexane Chair
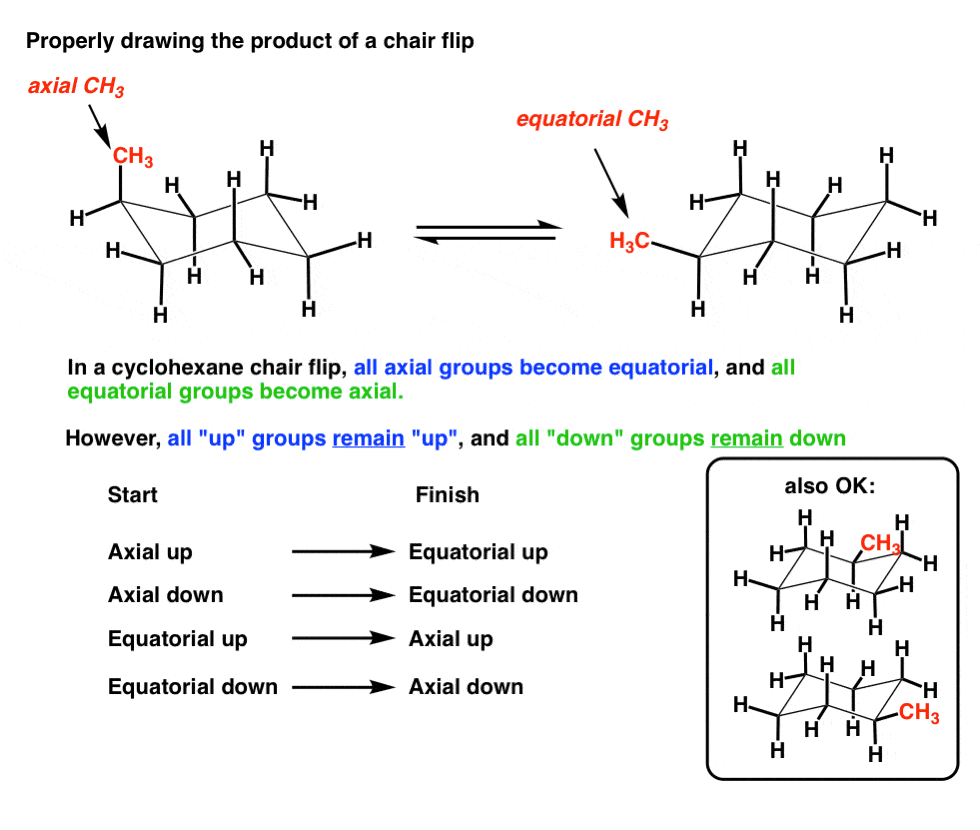
- The Chair Flip Converts All Axial Groups To Equatorial Groups And Vice Versa
- The Chair Flip of 1-Methylcyclohexane, Mapped out
- Three Ways NOT To Do A Cyclohexane Chair Flip
- Notes
- Quiz Yourself!
- (Advanced) References and Further Reading
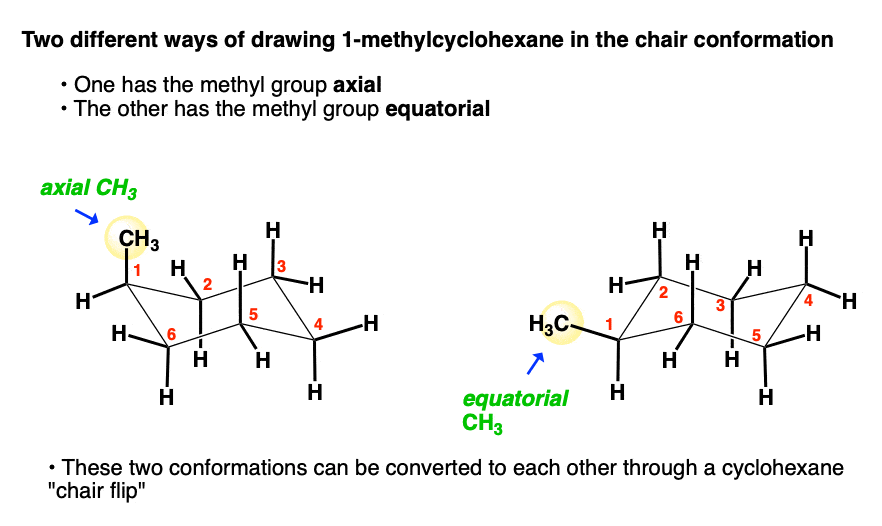
1. The Two Chair Conformations Of 1-Methyl Cyclohexane
In a recent post, an aerial tour of the cyclohexane chair, we showed that there are two different positions a substituent can occupy on a cyclohexane chair – axial (straight up and down, relative to the ring) and equatorial (off to the side of the ring).
This brings up an interesting thought experiment. Let’s take a really simple substituted version of cyclohexane – 1-methylcyclohexane, for example.
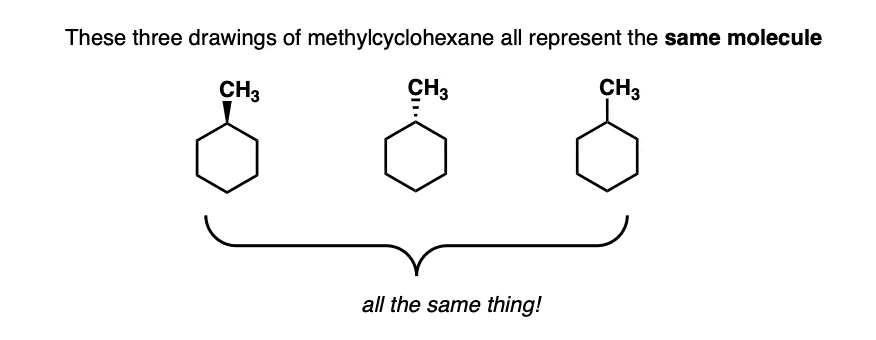
From the “dash wedge” diagram, there is only one way to depict 1-methylcyclohexane. Like this.
No matter how you rotate it, or flip it – it’s all superimposable versions of the same thing.
Now let’s draw the cyclohexane chair version. We draw a chair, and then…. hold on!
Can you see how this is a different situation? We could put the methyl group on an axial carbon or an equatorial carbon… and these are not superimposable on each other, unlike the “flat” drawing above.
2. How Do These Conformations Interconvert? Through A Cyclohexane “Chair Flip”
Same connectivity, different shape – this is a definition of “conformational isomers” if ever there was one.
However, unlike, say, conformations in linear alkanes, which involve rotations about single bonds, it might not be immediately obvious how the chair on the left can be converted to the chair on the right.
The point of this post is to describe how these two conformations can be converted into each other, through a series of bond rotations we call a “chair flip”.
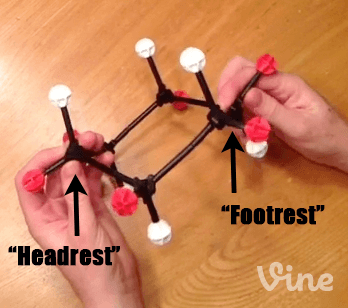
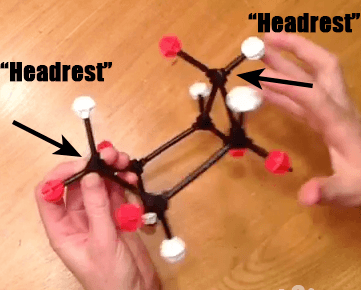
When we first introduced the cyclohexane chair, we mentioned that it was the lowest energy conformation of cyclohexane – but not the only conformation. Here’s a molecular model of cyclohexane, showing all the axial hydrogens (white) and equatorial hydrogens (red). Looked at from one perspective, we see the chair with its “head rest” on the left and “foot rest” on the right.
Step 1: Bring Up A “Footrest” of The Chair To Make A”Boat” Hammock
Now, if we take the “foot rest” of cyclohexane, and rotate several of the bonds, we are able to obtain a structure which looks like it has two “head rests”. This is called the “boat” [To be technical, it will actually be a “twist boat”, where there is a slight offset of the groups on the “prow”]. I am still partial to “hammock”. IUPAC, are you listening?
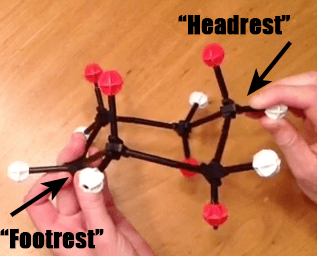
Step 2: Pull Down The Opposite Head Rest To Make A New Foot Rest
Now, if we take the “original” head rest (on the left) and pull it down, so that it makes a new “foot rest”, we have the following.
Your first thought might be – that’s pretty useless! Who cares if we just start with a cyclohexane chair and convert it into another cyclohexane chair. That’s like converting a dollar bill into four quarters and then exchanging them back into a dollar bill. It’s almost like nothing happened.
5. The Chair Flip Converts All Axial Groups To Equatorial Ones, And Vice Versa
Ahh! You see – it’s not QUITE the same. In this video, watch how this model of a cyclohexane chair is first converted into a boat, and then into a new cyclohexane chair.
Here’s the molecular ballet in motion. Pay close attention to the [white] axial groups and the [red] equatorial groups. What do you see?
During this chair flip, all the axial groups become equatorial… and all the equatorial groups become axial!
Here’s another way of showing how all equatorial substituents become axial (and vice versa) from this video courtesy of OChemNinja (YouTube)
This brings us back to 1-methylcyclohexane.
6. The Chair Flip Of 1-Methylcyclohexane, Revisited
These two conformations can be converted to each other using a chair flip. Note how on C-1, “axial up” becomes “equatorial up”. That’s the right way to do it.
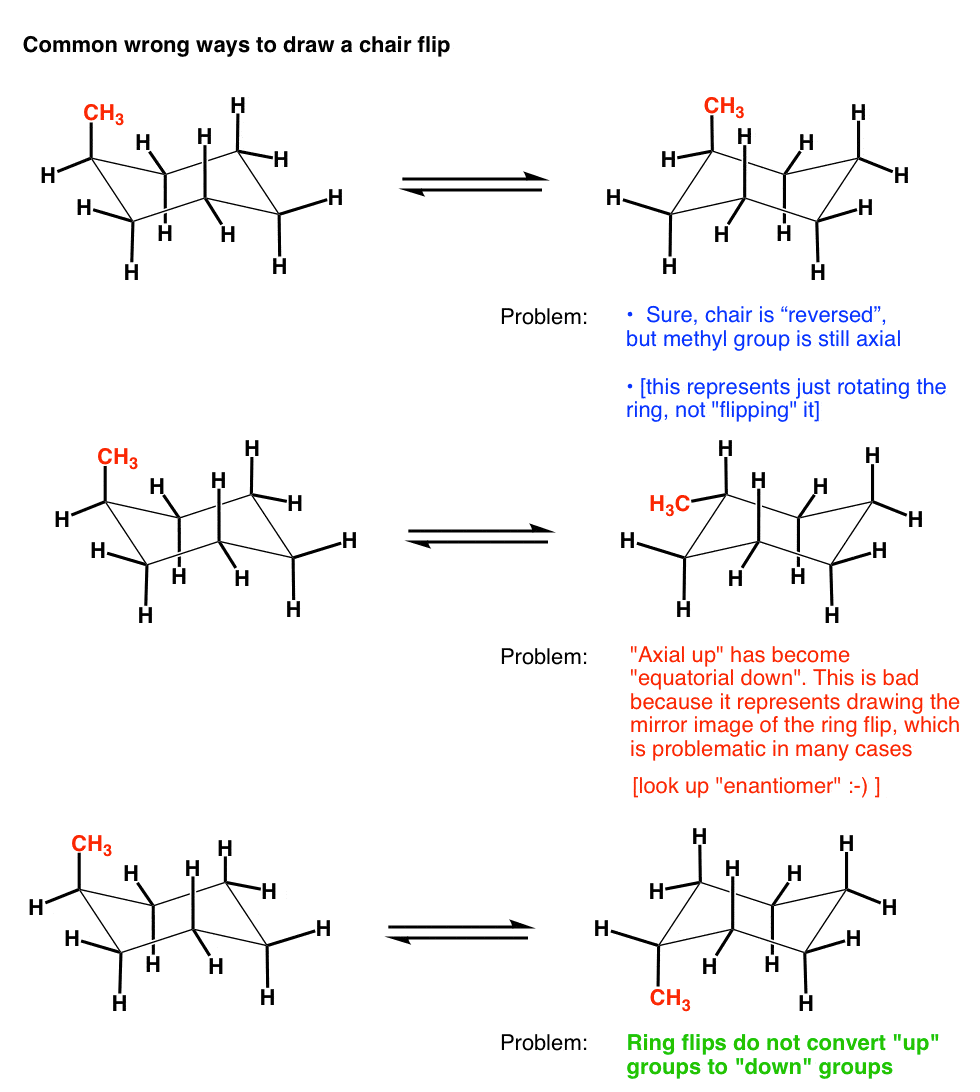
7. Three Ways NOT To Properly Do A Cyclohexane Chair Flip
This being organic chemistry, there’s plenty of ways to screw this up. Here’s a few wrong ways to draw a chair flip :
In the next post, we’ll look into this “ring flipping” process in a bit more detail. Stay tuned!
Notes
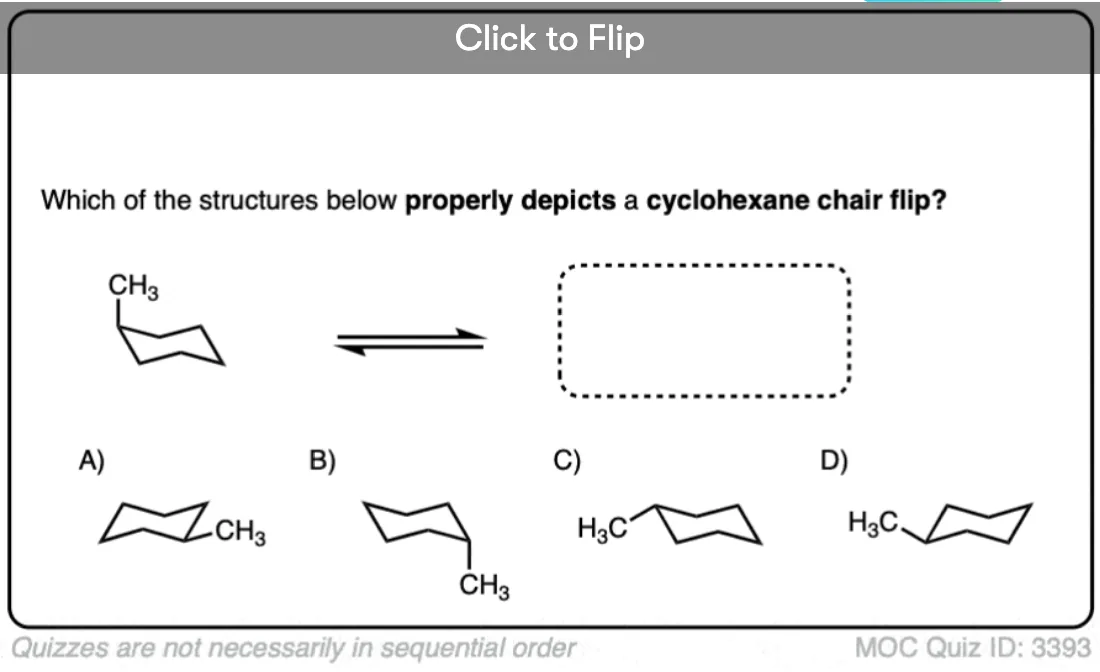
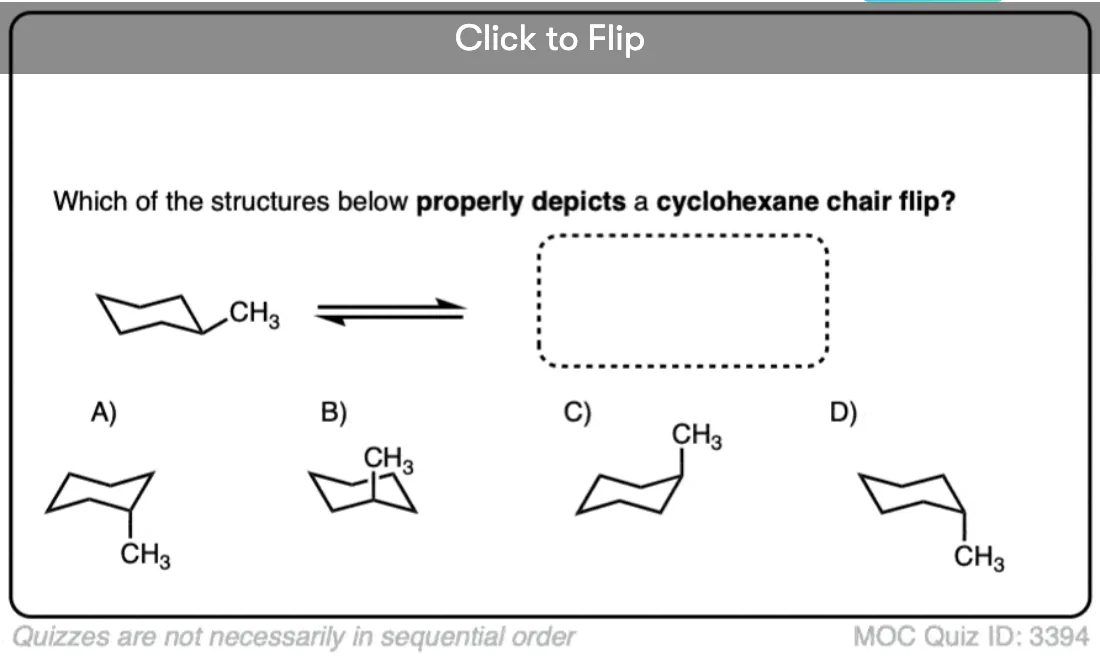
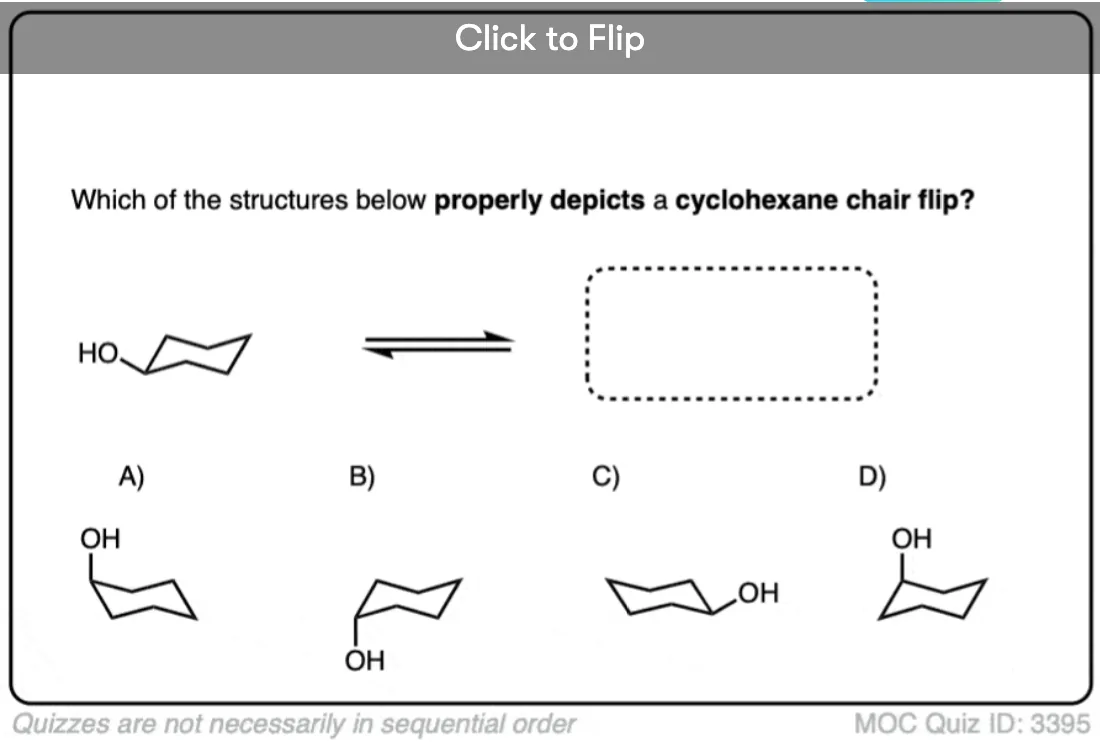
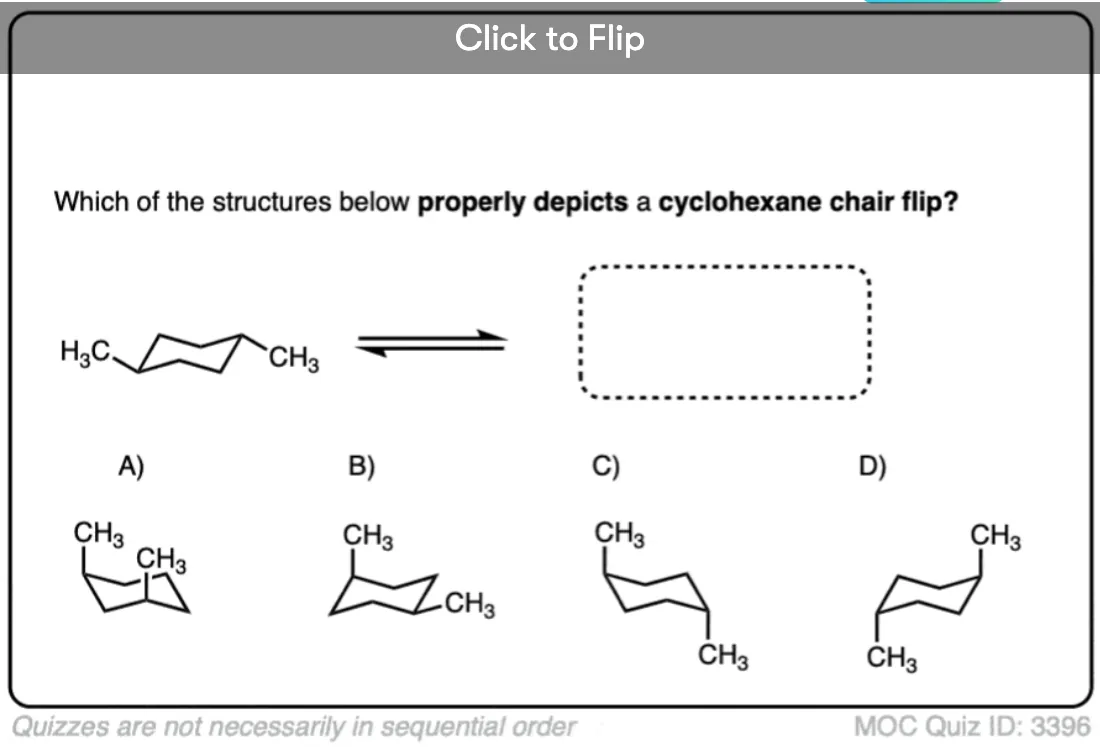
Quiz Yourself!
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
(Advanced) References and Further Reading
This is a topic commonly taught to undergraduates in Organic Chemistry. Cyclohexane’s ground state conformation is the chair, and it can undergo a ring ‘flip’, where axial substituents become equatorial substituents. This flip goes through some higher-energy intermediates (the boat, half-boat, and twist-chair).
- Ueber die geometrischen Isomerien der Hexamethylenderivate
H. Sachse
Chem. Ber. 1890, 23 (1), 1363-1370
DOI: 10.1002/cber.189002301216
The conformations of cyclohexane and related six-membered rings have been of active interest since at least 1890. - Die Baeyersche Spannungstheorie und die Struktur des Diamanten
Ernst Mohr
Journal für Praktische Chemie 1918, 98 (1), 315-353
DOI: 1002/prac.19180980123
A very early paper on the 3-D model of cyclohexane, showing that it is not flat, and providing models for the chair conformation. - The conformation of the steroid nucleus
H. R. Barton
Experientia 1950, 6, 316-320
DOI: 10.1007/BF02170915
This early paper by Nobel Laureate Sir Prof. D. H. R. Barton is on the conformational analysis of cyclohexanes and later applies this to the 3-D structure of steroids (which contain several fused 6-membered rings). He notes that cyclohexane confomers can interconvert, stating, “a small difference in free energy content (about one kilocal, at room temperature) between two possible conformations will ensure that the molecule appears by physical methods of examination and by thermodynamic considerations to be substantially in only one conformation.” - Nomenclature of cycloHexane Bonds
BARTON, D., HASSEL, O., PITZER, K., PRELOG, V.
Nature 1953, 172, 1096–1097
DOI: 1038/1721096b0 - Nomenclature of Cyclohexane Bonds
H. R. Barton, O. Hassel, K. S. Pitzer, V. Prelog
Science 1954, 119, 49
DOI: 10.1126/science.119.3079.49
These are the first instances of the terms ‘axial’ and ‘equatorial’ being used to denote the two positions substituents can take in cyclohexane. This was also back in the day when scientists could safely cross-publish to get better visibility – pretty much the same article is published in both Science and Nature, considered top journals. - Nuclear Magnetic Resonance Line-Shape and Double-Resonance Studies of Ring Inversion in Cyclohexane-d11
A. L. Anet and A. J. R. Bourn
Journal of the American Chemical Society 1967, 89 (4), 760-768
DOI: 10.1021/ja00980a006
This paper covers a classic experiment and is commonly mentioned in undergraduate and graduate organic chemistry or NMR courses. At room temperature, cyclohexane gives one signal because interconversion of chair forms occurs rapidly. At low temperatures, however, it gives a very complex 1H NMR spectrum. At low temperatures interconversions are slow; the chemical shifts of the axial and equatorial protons are resolved, and complex spin-spin couplings occur. At -100 °C, however, cyclohexane-d11 gives only 2 signals of equal intensity. These signals correspond to the axial and equatorial hydrogen atoms. Interconversions between these conformations occur slowly at this low temperature, but they happen slowly enough for the NMR spectrometer to detect the individual conformations (the nucleus of a deuteron has a much smaller magnetic moment than a proton, and signals from deuteron absorption do not occur in 1H NMR spectra). Prof. F. A. L. Anet is an Emeritus Professor at UCLA and was a pioneer in the use of NMR spectroscopy for conformational analysis. - Non‐Chair Conformations of Six‐Membered Rings
M. Kellie, F. G. Riddell
Topics in Stereochemistry 1974, 8
DOI: 10.1002/9780470147177.ch3
This reference contains useful information on the inversion barrier for cyclohexane, as well being the first paper to actually invoke the ‘twist-boat’ conformation during this process. - Conformational equilibrium trapping by high-vacuum cryogenic deposition
A. L. Anet and M. Squillacote
Journal of the American Chemical Society 1975, 97 (11), 3243-3244
DOI: 10.1021/ja00844a067
The chair-twist energy difference has been directly measured by low-termperature IR spectroscopy. The chair was determined to be 5.5 kcal/mol lower in enthalpy than the twist. - Conformational structure, energy, and inversion rates of cyclohexane and some related oxanes
Herbert L. Strauss and Herbert M. Pickett
Journal of the American Chemical Society 1970, 92 (25), 7281-7290
DOI: 1021/ja00728a009
This paper describes a theoretical method for setting up calculations for ring inversion. - Conformational analysis. 130. MM2. A hydrocarbon force field utilizing V1 and V2 torsional terms
Norman L. Allinger
Journal of the American Chemical Society 1977, 99 (25), 8127-8134
DOI: 1021/ja00467a001
The MM2 (Molecular Mechanics 2) method was developed by Prof. Allinger for conformational analysis of hydrocarbons and other small organic molecules. This paper documents results for calculations using this method, including the ring inversion of cyclohexane. MM methods are considered crude nowadays because they neglect quantum and relativistic effects, but they are nonetheless useful for doing initial geometry optimization of a structure before doing a higher-level calculation.
Is a ring flip just another way of saying chair flip?
Yes – ring flip, chair flip, same thing.
when we convert skeletal structures (with stereochemistry) to chair conformation, is there a rule that axial means out of paper (wedge) and equatorial means into paper (dash)? Or does it depend, like axial up & equatorial up means out of paper (wedge), and axial down & equatorial down means into paper (dash)? Thanks :-)
“Down” means dash and “Up” means wedge.
It’s possible for groups that are ‘up’ (wedged) to be axial *or* equatorial. Likewise, groups that are down can also be axial *or* equatorial.
https://www.masterorganicchemistry.com/2014/05/14/an-aerial-tour-of-the-cyclohexane-chair/#eight
Wow am happy this website has been a source of inspiration to me as it help me soft some difficult chemistry problem thanks
Great, thank you.
Thank you so much this website has helped me so much
Great, thank you Erika.
Superb
Wow! This is so helpful. I’m so grateful that someone created this resource for free- this is better than ANY textbook that I’ve ever seen….a truly exceptional set of explanations.
Hey James,
Just wanted to say thanks for putting all this up, I found it really helpful.
Josh
Hey james
Want to say thank you alot for your work .
Hey James, great post! In addition to the video, there’s also an interactive movie of a cyclohexane chair flip on the CWC website: http://web.chemdoodle.com/tutorial/3d-structure-canvases/moviecanvas3d
You can copy the code shown, and also link the CWC JS file from here: http://web.chemdoodle.com/installation/download
Best!