Amines
The Hofmann Elimination
Last updated: May 28th, 2026 |
The Hofmann Elimination Of Alkylammonium Salts: Examples and Mechanism
- The Hofmann Elimination is an elimination reaction of alkylammonium salts that forms C-C double bonds [pi bonds]. [Note 1] It proceeds through a concerted E2 mechanism.
- In contrast with most elimination reactions that make alkenes, which follow the Zaitsev (Saytzeff) rule, the Hofmann elimination tends to provide the less substituted alkene.
- In this post we go through the difference between Hofmann elimination and Zaitsev elimination and explain the key features in the Hofmann degradation mechanism that result in its preference for the “less substituted” alkene.
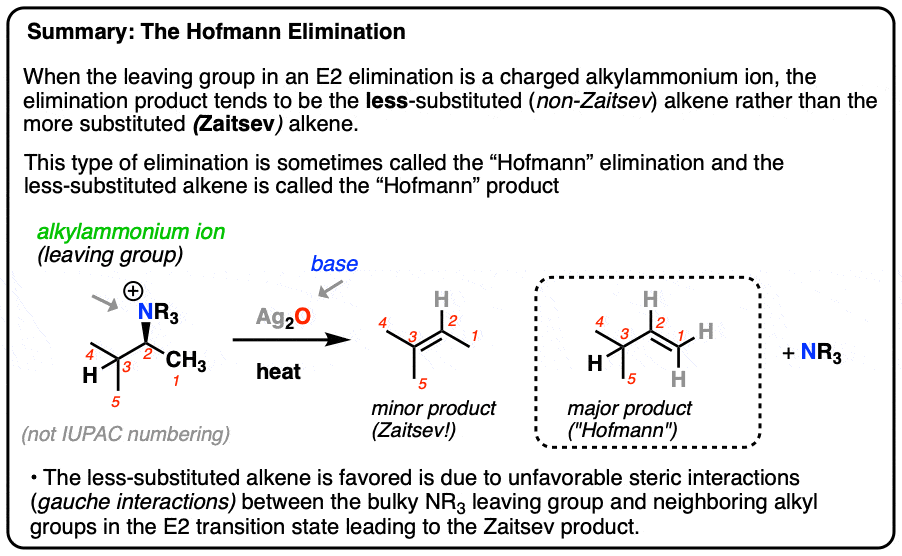
Table of Contents
- Quick Review: Zaitsev’s Rule
- “Non-Zaitsev” Products Can Dominate When Sufficient Steric Hindrance Is Present
- The “Hofmann Degradation”
- The Hofmann Elimination Has An Extremely Bulky Leaving Group, And This Leads To “Non-Zaitsev” Elimination Products
- Summary: The Hofmann Elimination
- Notes
- Quiz Yourself!
- (Advanced) References and Further Reading
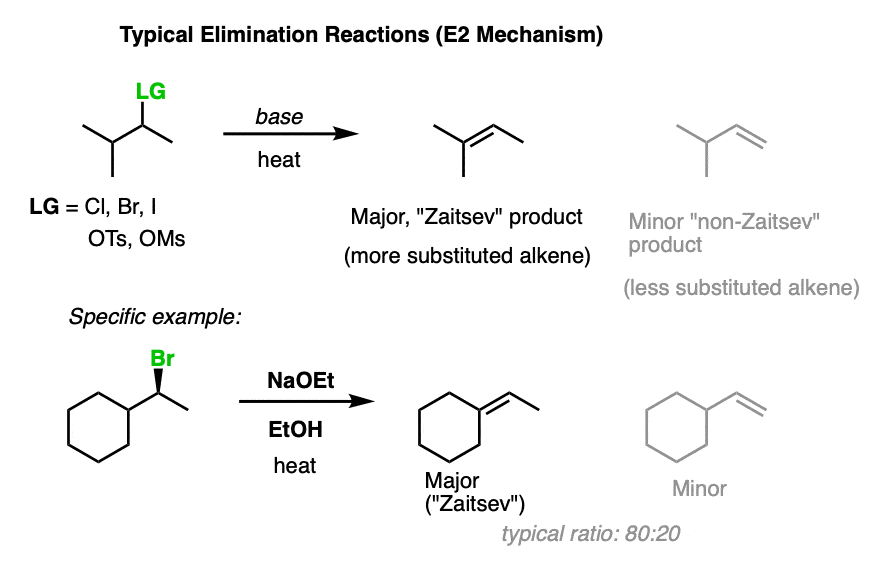
1. Quick Review: Zaitsev’s Rule
Conventional elimination reactions that occur via the E2 mechanism follow Zaitsev’s rule. The major product will be the more substituted alkene (that is, the alkene with the most carbons directly attached to the alkene).
For instance in the first example below, the “trisubstituted” alkene is favoured over the “mono-substituted” alkene.
Why? The thermodynamic stability of alkenes increases in the order
mono-substituted < disubstituted < trisubstituted <tetrasubstituted. (For more, see post on Alkene Stability)
The energy differences are quite small – about 2 kcal/mol, but that’s enough to deliver an 80:20 ratio of products! [How do we know this? It can be obtained by plugging 2 kcal/mol into the equation ln K = –ΔG/RT]
2. “Non-Zaitsev” Products Can Dominate When Sufficient Steric Hindrance Is Present
Sometimes “non-Zaitsev” products can be obtained through the use of a bulky base for the elimination reaction. A classic example is to use sodium or potassium t-butoxide (KOt-Bu); another is to use lithium di-isopropyl amide (LDA). The idea here is that the bulky base will react more quickly with the least sterically hindered proton on a beta-carbon, which results in formation of the least substituted alkene. [for more, see: Bulky Bases In Elimination Reactions].
You sometimes might see these “non-Zaitsev” products be referred to as “Hofmann products”. Why?
Back to amines.
3. The “Hofmann Degradation”
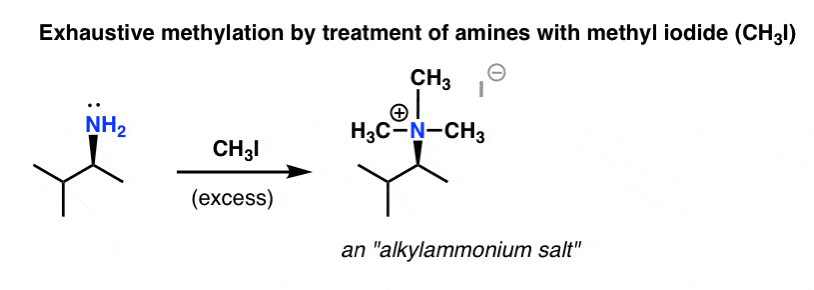
Back in 1851, not many techniques for analyzing complex molecules were available. One method for determining the structure of an unknown compound was to break it down into simpler pieces and look for clues in the fragments, a process called degradation. August Wilhelm von Hofmann developed a two-step degradation method for amines that was later to bear his name.
The first step is to treat an amine with a vast excess of methyl iodide [CH3I], which results in an ammonium salt [we saw this reaction, “exhaustive methylation”, in a previous post].
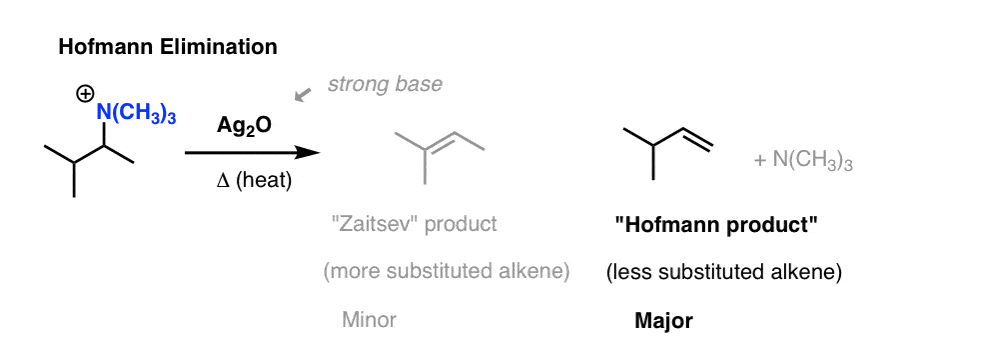
The second step is to distill the ammonium salt under low pressure in the presence of a strong base. Silver oxide (Ag2O) is often used.
It might not immediately look like it, but tertiary amines ( :NR3 ) are relatively weak bases (pKaH = 10) and therefore decent leaving groups. [recall that good leaving groups = weak bases]
Heating with strong base results in an elimination reaction: NR3 is lost and and a new alkene is formed.
The interesting observation here is that the alkene product from this process tends to be the least substituted alkene (“Hofmann product”) not the Zaitsev product.
What’s going on?
4. The Hofmann Elimination Has An Extremely Bulky Leaving Group, And This Leads To “Non-Zaitsev” Elimination Products
It’s not that there’s something about the product alkene that makes it more stable than the Zaitsev product (it isn’t).
The answer lies in the relative energies of the transition states leading to the two products.
It might help to look at the mechanism for the reaction again. Recall that the E2 mechanism demands an antiperiplanar (180°) arrangement of the C-H and C-LG bonds.
It really helps to visualize this by drawing out Newman projections. When we do that, what do you notice?
For most elimination reactions, the steric hindrance of the leaving group isn’t a factor we need to consider. Even though leaving groups like I and Br have a large Van Der Waals radius, their bonds to carbon are long, and being single atoms they don’t interfere with adjacent groups.
Contrast that to the NR3 group, which is like a big-ass ceiling fan spinning around its three alkyl groups – and each of the alkyl groups themselves is like a mini-ceiling fan spinning around three hydrogen atoms. It takes up a lot of space!
The conformation that leads to the “Zaitsev” product has a lot more steric hindrance (two gauche interactions!) than the conformation that leads to the “Hofmann” product, because of the extremely bulky N(CH3)3 leaving group!
These extra steric interactions are enough to disfavour the Zaitsev transition state relative to the Hofmann transition state, and lead to the Hofmann product as the major product.
5. Summary: The Hofmann Elimination
The Hofmann elimination is just another example of how tweaking a single variable in a chemical reaction can flip the outcome. We saw earlier how increasing the steric hindrance of the base can lead to the non-Zaitsev product. Here, we’re increasing the steric hindrance of the leaving group.
A few kcal/mol difference in a transition state might not sound like a lot, but it’s more than enough to change the identity of the major product. This is what makes organic chemistry so frustrating to beginners… yet also so deeply interesting!
Notes
Note 1. Not strictly limited to ammonium salts. There are also examples of “Hofmann-type” eliminations with phosphonium (PR3+) leaving groups as well.
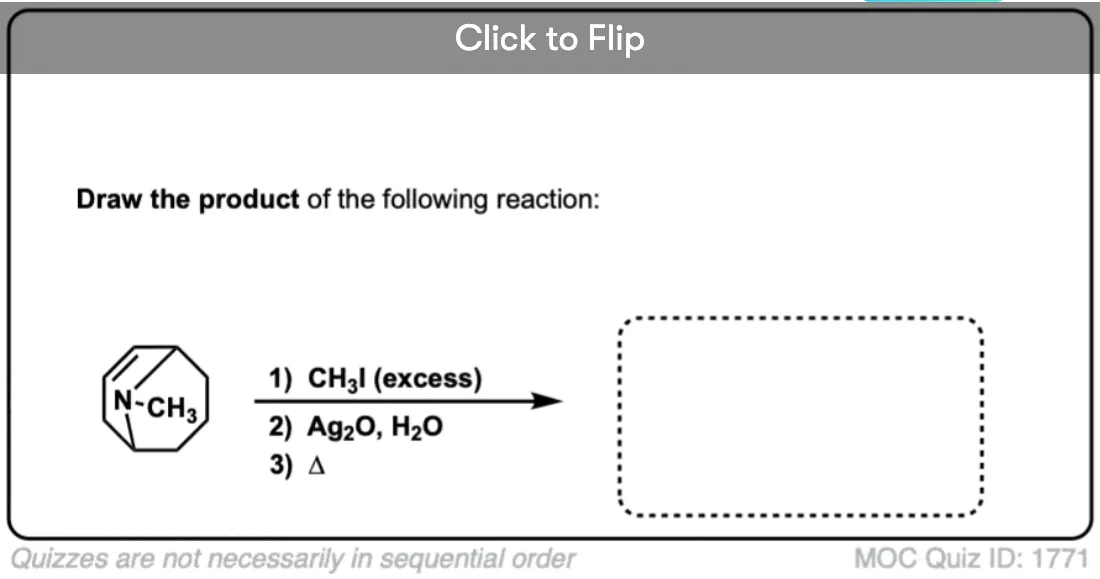
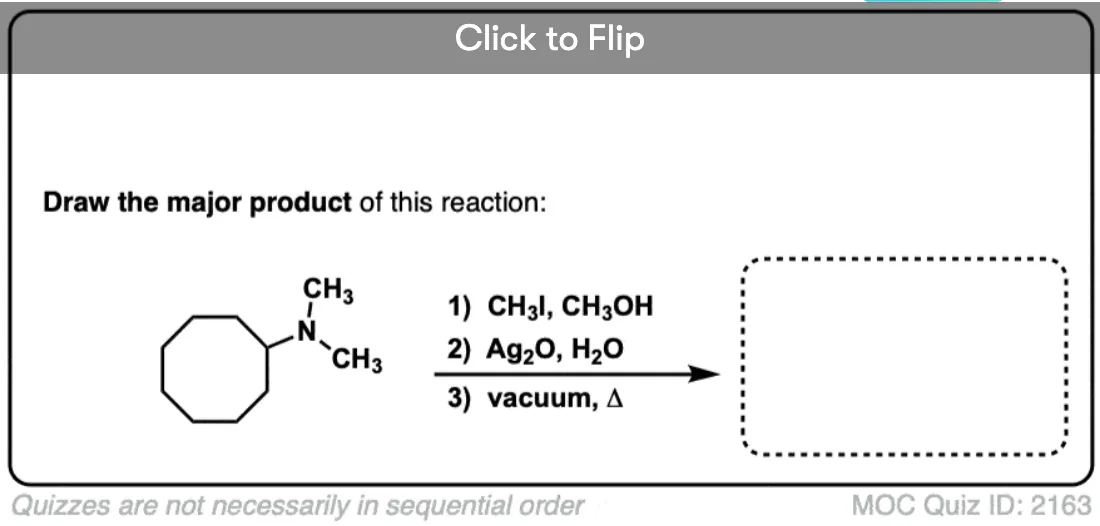
Quiz Yourself!
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
(Advanced) References And Further Reading
- Einwirkung der Wärme auf die Ammoniumbasen
W. Hofmann
Chem. Ber. 1881, 14 (1), 659-669
DOI: 10.1002/cber.188101401148
The original paper by W. Hofmann on a new method for olefin synthesis. He was a very productive organic chemist in the 19th century and his name has been attached to a variety of transformations, including amide degradation, isonitrile synthesis, and a few others.
- Olefins from Amines: The Hofmann Elimination Reaction and Amine Oxide Pyrolysis
Cope, Arthur C.; Trumbull, Elmer R.
Org. React. 1960, 11, 317-493
DOI: 10.1002/0471264180.or011.05
Organic Reactions, published and maintained by the ACS division of Organic Chemistry, is a source of comprehensive reviews on various transformations in organic chemistry. This particular review is written by Prof. Cope (MIT, of the Cope rearrangement). Detailed experimental procedures are provided towards the end. - Hofmann-Type Elimination in the Efficient N-Alkylation of Azoles: Imidazole and Benzimidazole
András Horváth
Synthesis 1994; 1994 (1): 102-106
DOI: 1055/s-1994-25414
Alkylation of cyanoethyl-substituted azoles followed by heating with a strong base yields acrylonitrile via a Hofmann elimination. - Cyclization in the Course of Clarke—Eschweiler Methylation
Arthur C. Cope and W. Dickinson Burrows
The Journal of Organic Chemistry 1965 30 (7), 2163-2165
DOI: 1021/jo01018a011
Two Hofmann eliminations are indicated in this paper, with compounds 5 and 7.
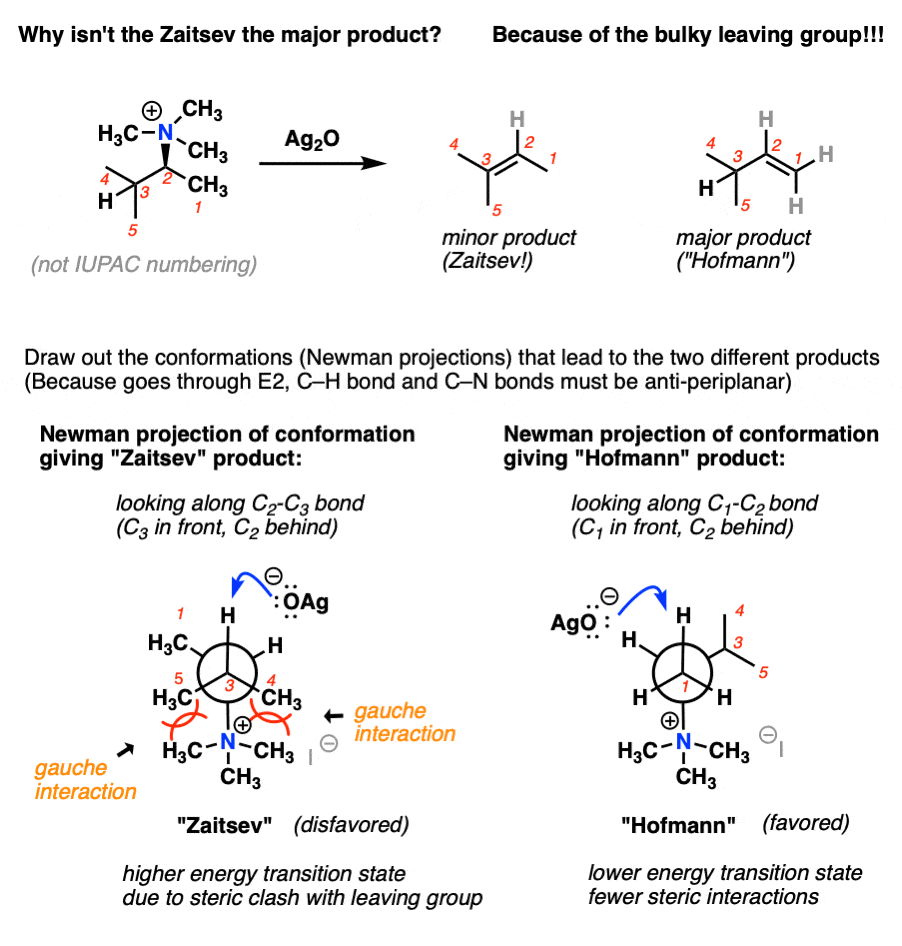
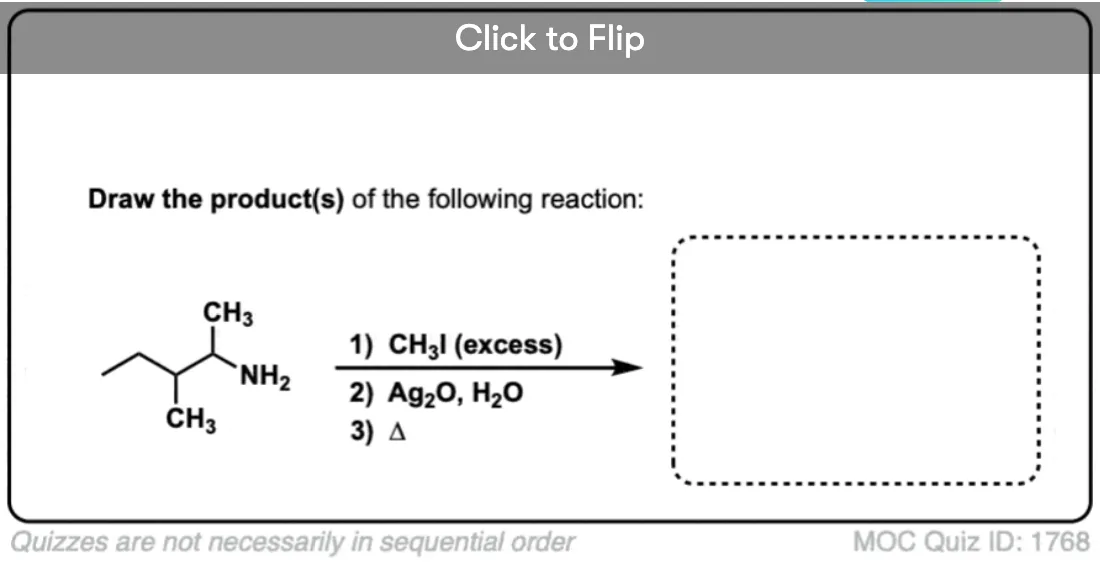
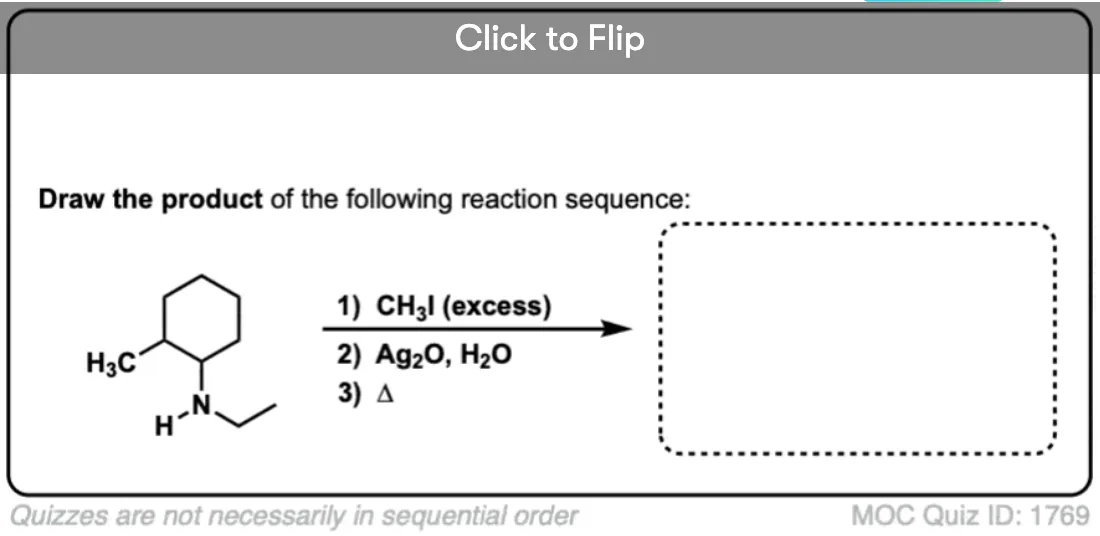
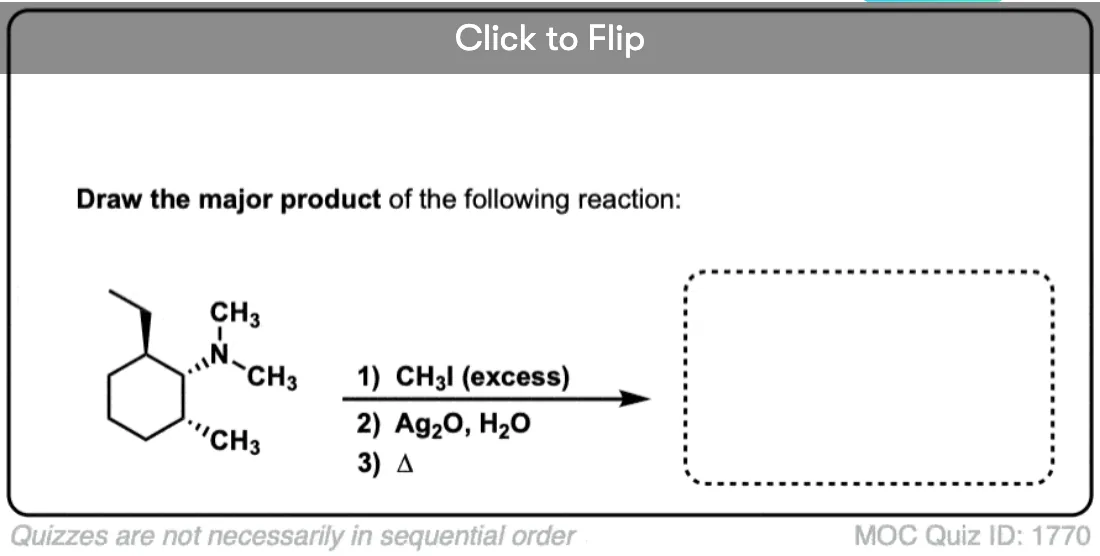
Why is silver oxide used in Hofmann Eliminations? Does the silver act as a catalyst somehow? Why are other cheaper bases not used instead?
By no means does Ag2O *have* to be used. It was used by Hoffmann, perhaps because the elimination was performed simply by adding base after alkylation and heating until the elimination product distilled off. Perhaps Ag+ helps to further destabilize the ammonium ion by making insoluble AgI, which speeds elimination, but I don’t believe there is anything magical about Ag2O.
I have a doubt. I read in Paula Bruice that Hoffman Elimination is preferred in highly electronegative or bulky groups like F or -NR3+ . The main reason is because when the base abstracts the proton from adjacent Carbon, the poor leaving tendency of these groups causes a carbanion to form at the adjacent Carbon (This is unlike the normal E1 and E2 elimination where either carbocation is formed or both H and leaving group leaves simultaneously) . So the T.S. for a Hoffman Elimination is basically a carbanion. As 1°>2°>3° , therefore the double bond is formed at less substituted position.
Now, my question arises “What if we add a -M, -I functional group to the adjacent carbon. Will the carbonion be stabilised and form the Zaitsev product even when the leaving group is NR3 ?”
Hello James,
My professor told us that in the last example, the Hofmann product would be major because the trimethylamine is positively charged, and because the tertiary CH is less “acidic” than the secondary CH (which means that the sec. gives its H+ more easily), we would have the Hofmann product (because in thisn case the positive charge would make this difference stronger).
Would you say that it is also a valid explanation? (He didn’t mention the steric reason of the leaving group though.)
Thanks in advance!
Hi Johann – that is a perfectly good explanation, and possibly a more rigorous one. The positively charged nitrogen is a strong electron withdrawing group, which increases acidity of the beta hydrogens. However the basicity of the tertiary hydrogen will be less, since it is made more electron-rich by its adjacent alkyl groups. The secondary will be more acidic. Also, there is a statistical contribution since there are two secondary hydrogens and only one tertiary hydrogen.
Could you please explain why this reaction too yields a Hofmann product?
I couldn’t paste the image here so here’s a drive link:
https://drive.google.com/open?id=1v-yTJP15O7qASQfIIYIEEUSKZo8hGirB
It’s from a reliable source (chem.libretexts) but I can’t bring myself to accept the outcome. Any hints towards the probable mechanism please?
Thanks in advance.
POCl3 gives a phosphate leaving group. Like the trialkylammonium leaving group, It is not unknown that the phosphate leaving group can have unfavorable interactions with the rest of the carbon chain leading to “anti Zaitsev” products instead of the normal “Zaitsev” product. One other example where it has been observed is in eliminations adjacent to the steroid nucleus, as shown by Djerassi and co-workers. See Giner, J.-L. et. al. J Org Chem. 1989 54 369. DOI . https://pubs.acs.org/doi/pdf/10.1021/jo00263a020 .
“Dehydration of 22-
(S)-alcohol 7 gave the 20(22) E olefin 8. However, dehydration of 22(f?)-alcohol 9 did not produce the 20(22) Z
olefin, but rather stigmasterol or brassicasterol i-methyl
ether (6) together with a product thought, based on the
mass spectrum, to be the 22-chloride. This result can be
rationalized on the basis of unfavorable steric interactions
in the transition state leading to the 20*2® product (Figure
3)”.
Figure 3 shows a Newman projection rationalizing the product.
75 year old danishefsky/stork student. Lilly scientist, Butler un organic lecturer
discovered sunday that stork had died at 95 yrs old.
he completed Germine synthesis by himself and wife in columbia lab at 95 yrs old.
reviewing his synthesis and found your web site;
love your teaching format
fun to review old woodward synthesis from 1958
I will keep your web site for quick reviews
thank you and best of luck to you.
Gary – it is an honor to have you as a reader. Thank you for stopping by.
Hi. I have a problem related to Hofmann Elimination . Can you help me this exercises?
It’s here
When the (R,R) isomer of the amine shown is treated with an excess of methyl iodide, then silver oxide, then heated, the major product is the Hofmann product.
(a) Draw the structure of the major (Hofmann) product.
(b) Some Zaitsev product is also formed. It has the (E) configuration. When the same amine is treated with MCPBA and heated, the Zaitsev product has the (Z) configuration. Use stereochemical drawings of the transition states to explain these observations.
( I can’t upload the picture of this exercise here so I upload it in google driver .this is link :
https://drive.google.com/file/d/1POFMh0Diw9-Qs_8d31HFsjnGtyje6nN7/view?usp=drivesdk