Fun and Miscellaneous
What Holds The Nucleus Together?
Last updated: May 24th, 2021 |
Here’s what I would call the One Sentence Summary Of Chemistry. If you learn just one thing about chemistry, learn this.
“Opposite charges attract, like charges repel”.
We’ll go into a lot more detail over the next few posts, but let’s get started by thinking of some examples.
- positive charges attract negative charges – Two examples here. Think of the negatively charged electron that orbits the positively charged nucleus. Or sodium chloride, which is composed of positively charged sodium ions held together with negatively charged chlorine ions. Positive attracts negative. Check.
- Negative charges repel negative charges. You may recall that the shape of a molecule depends on the number of bonds/electron pairs around an atom. Why is water bent, for instance? That’s because the oxygen lone pairs each take up space, and the electron pairs repel until they are the maximum distance apart. That’s also why a molecule like methane is tetrahedral and not square planar.
- Positive charges repel positive charges. It’s harder to think of examples here, but here’s one. It’s relatively easy to add a proton to water to give [H3O]+, but very difficult to protonate that further to give [H4O]2+ due to repulsion.
It’s this last one which seems weird. If it’s so hard to make [H4O]2+, for instance, how is it that the nucleus holds together, since it’s essentially made of positively charged protons with a few neutrons thrown in.? It’s especially weird when you consider that these positive charges are distributed over such a short distance.
Let’s take a step back to look at the Really Big Picture to find out.
The Really Big Picture
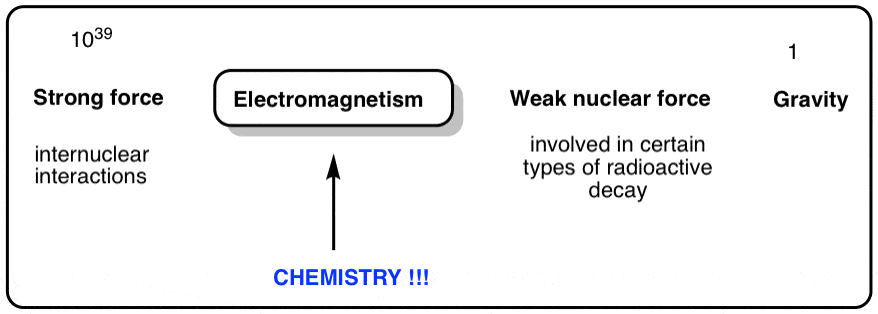
Physicists have identified four fundamental forces in nature that are responsible for all the phenomena we observe. They are
- gravity, which acts at long range, and is attractive (but weak)
- electromagnetism – responsible for the behavior of electric and magnetic fields.
- the weak nuclear force, responsible for certain types of nuclear decay (when a neutron breaks down into a proton and an electron, that’s the weak nuclear force at work).
- and then we come to the strong nuclear force, which acts at very short range and is attractive.
The forces vary in strength over many, many orders of magnitude (getting the extremely weak gravitational force to fit into a Theory of Everthing, in particular, has been driving physicists nuts for decades).
Two observations:
Chemistry is predominantly about electromagnetism . The study of chemistry is largely the study of electrons and how they flow between atoms. In chemistry, we’re essentially just looking at electrons and nuclei as point charges, and treating the chemical bonds that form between them as electrostatic phenomena. Furthermore, the electromagnetic fields accompanied by these charges can interact with radiation with wavelengths all across the electromagnetic spectrum, giving rise to the huge variety of spectroscopic techniques we have developed to help give us clues as to the structure of atoms and molecules.
What about the other forces in chemistry? Gravity is irrelevant – compared to the electromagnetic force, it’s a rounding error at the 30th decimal place, essentially. The weak nuclear force doesn’t much come into play either, especially when it comes to the meat and potatoes organic chemistry we’re going to be discussing. What about the strong nuclear force? It has one – and only one – important role in the chemistry we’re going to discuss, and that’s it.
What holds the nucleus together? The strong nuclear force. At extremely short range, it is stronger than electrostatic repulsion, and allows protons to stick together in a nucleus even though their charges repel each other. Remember that the size of the nucleus is really small compared to the size of an atom. Since it operates only over distances comparable with the diameter of an atom, it isn’t felt at longer distances, and for the purposes of chemistry, we ignore it.
When it comes to the strong force, there is a break-even point. Physicists have observed that the nucleus becomes increasingly more stable upon the addition of nucleons up to iron-56. Then, as successive protons are added, the nucleus gets more and more unstable, due to the electrostatic repulsion outweighing the attractive nuclear force. Once you get up to really high atomic numbers, the nucleus becomes unstable and can fragment into smaller, more stable components. This is the basis of nuclear fission, or why uranium (atomic number 92) and plutonium (atomic number 94) are used for atomic energy (and nuclear weapons). So over a large enough distance, there’s an limit to how much positive charge you can put in the nucleus without making it highly unstable. This is why I wouldn’t be expecting to read much about the chemistry of the g-block elements anytime soon.
Religion doesnt explain anything Science. Biblical inferences is for the lazy mind, who resorts to an easy way out of scienctific challenges.
Leave the bible in the church. Bible doesnt explain where God came from either. Fair and square.
This really very helpful regarding the basic point of holding the nucleus….but there is no idea that what produce the strong nuclear holding force
Well, the strong nuclear force is one of the four fundamental forces of nature, and answering what produces it is therefore a very deep and mysterious question.
I was right :D ”the neutrons are a source of more strong force for the nucleus since they participate in the meson exchange. These factors, coupled with the tight packing of protons in the nucleus so that they can exchange mesons creates enough strong force to overcome their mutual repulsion and force the nucleons to stay bound together.” Thank you for the confirmation :)
thank you for the reply :)
What about the effects of the neutrons since they are neutral, don’t they have an effect to ‘block’ or ‘absorb’ some of the repulsion of the protons?
I am not a nuclear physicist, so this article is better than anything I could have written on the subject: http://aether.lbl.gov/elements/stellar/strong/strong.html#:~:targetText=Since%20they%20have%20no%20charge,participate%20in%20the%20meson%20exchange.
I wouldn’t use the phrase ‘How absurd!’ about this phenomenon. For a chemist, as James explained, they really don’t even need to know how, let alone what, keeps a nucleus together (that’s what nuclear physicists are for), only that it does. But there are a number of important facts to be learned here, e.g., that the ‘strong force’ has its limits: The size of a nucleus cannot exceed a certain diameter before it becomes unstable. But it’s also true that this force will not keep pulling protons and neutrons together any denser than certain limits. It’s also been proven that protons and neutrons are not basic particles, like an electron, but are each comprised of _quarks_ and other theoretical particles which protons and neutrons exchange with each other that bind them together.
Now, lest anyone think I’m an atheist, I’m not. I believe the same words that Bill Perry quoted, but I’m all for attempting to describe a phenomenon in as much detail as we possibly can. Another fascinating fact: A proton’s mass is slightly less than a neutron’s, and if it was the same as a neutron, then Hydrogen could not exist; the proton itself would be unstable! And, we owe that fact to nuclear physicists. Although there will be those who do such research only in an attempt to prove God does not exist, I believe the more we find out about the Universe and how it functions, the more there is to lead us to belief in its Creator.
Thank you for your insights and biblical viewpoint.
How absurd! You call this holding of atoms together the “strong force”. Boy, I’ll say!!! Well, that is the NAME you give it – but WHAT IS IT? No physicist knows. But I do! Read Col.:1-15 through 17.
That’s a very interesting theory.
Thank you.