Organometallics
The Heck, Suzuki, and Olefin Metathesis Reactions (And Why They Don’t Belong In Most Introductory Organic Chemistry Courses)
Last updated: July 3rd, 2025 |
The Heck, Suzuki, and Olefin Metathesis Reactions Are Really Cool. [But They Don’t Belong In (Most) Introductory Courses]
There has been a trend in recent years towards including transition metal catalyzed reactions in the introductory organic chemistry curriculum. The reactions most common covered are palladium catalyzed coupling reactions (Suzuki and Heck reactions in particular) and olefin metathesis.
I generally think this is a bad idea for most courses. In my opinion, covering transition metal catalysis introduces an excessive number of “open loops” – new reactions, reagents, mechanisms, and concepts – that simply cannot be closed in any satisfactory manner given the limited time available. More on that at the bottom of the post.
However, the fact remains that the material is often covered, and students have to deal with it. Rather than completely ignore this topic, I’ve chosen to provide a condensed overview of three important reactions involving “organometallics” – the Suzuki reaction, the Heck reaction, and olefin metathesis.
First, let’s look at things from the 50,000 foot perspective.
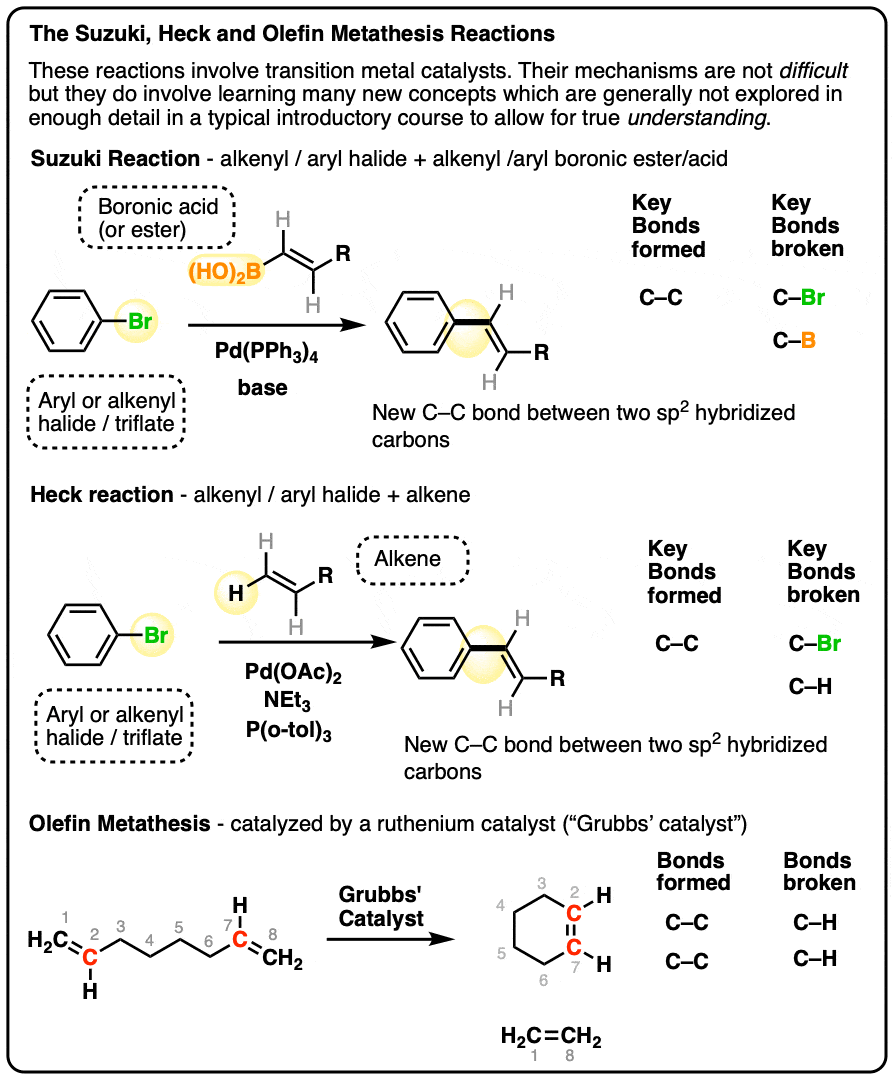
Table of ContentsRemove featured image
- Why Transition Metal Catalysis Is Important
- The Suzuki Reaction
- The Heck Reaction
- Olefin Metathesis
- Should These Reactions Be Covered In Introductory Courses? Pros and Cons
- Conclusion
- Notes
- Quiz Yourself!
- (Advanced) References and Further Reading
1. Transition Metal Catalysis Is Vital To Modern Drug Discovery and Fine Chemical Synthesis
Take a look at this excellent chart of the Top 200 Pharmaceutical Products by US Volume. Here are three examples.
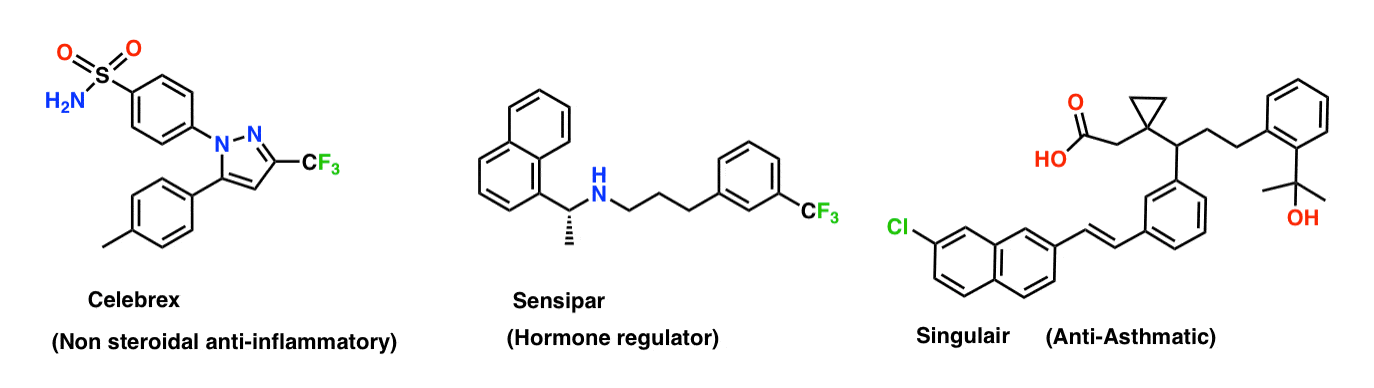
Each of these drugs represents the end result of incredibly expensive multi-year projects by major pharmaceutical companies and have generated untold billions of dollars in revenue. These are the “needles” found after searching through extremely large haystacks (10,000 molecules or more) in a process we call drug discovery.
This isn’t the place to cover drug discovery in detail (although if you’re curious, Derek Lowe’s In The Pipeline does that job admirably). I merely want to make the point here that organic chemists involved in this endeavour:
- need access to a large number of molecules (sometimes called “libraries”) that they can screen for biological activity (e.g. killing a certain cancer cell line, inhibiting a specific enzyme, etc.)
- often need to make small adjustments to “tune” or modify a molecule’s pharmacological properties (such as its fat-solubility, bioavailability, metabolic profile, and several other factors).
If you look at the molecules above, you’ll note that each of them contain aromatic rings and other sp2 hybridized carbons. This is a very common structural feature of drugs and drug-like molecules.
All this lead-up is merely to say that certain transition-metal catalyzed reactions are extremely useful for rapidly, predictably, and cheaply forming new bonds between sp2– hybridized carbons (e.g. aromatic rings) and thus have become an indispensable part of the modern organic chemists’ toolkit. They can be used to rapidly generate analogues of molecules for the purpose of improving their pharmacological properties.
[Just in case you need some perspective on the importance of small differences in structure, I should note that the mere presence or absence of a simple methyl group (CH3) has been known to sometimes make a 100-fold difference in the potency of a drug! ]
Let’s walk through the first and arguably most important transition metal catalyzed reaction we’ll cover: the Suzuki reaction.
2. The Suzuki Reaction
The Suzuki reaction is a palladium-catalyzed reaction for forming a new C-C bond between two coupling partners:
• an alkenyl or aryl halide (Br, Cl, or I) [or sometimes triflate (OTf)]
• an alkenyl or aryl boronic acid R-B(OH)2 [or sometimes boronic ester, R–B(OR)2]
The reaction is catalyzed by a palladium compound, which comes in many varieties (see below) but for simplicity, we’ll use Pd(PPh3)4. Base (such as NaOH or KOH) is also essential to the reaction.
The product of the reaction contains a new C–C bond between the carbon attached to the halogen and the carbon attached to the boronic acid. The halogen and the boronic acid are removed.
You can think of palladium as acting as a “matchmaker” in this reaction – it brings the two partners together, but is itself not incorporated in the reaction product.
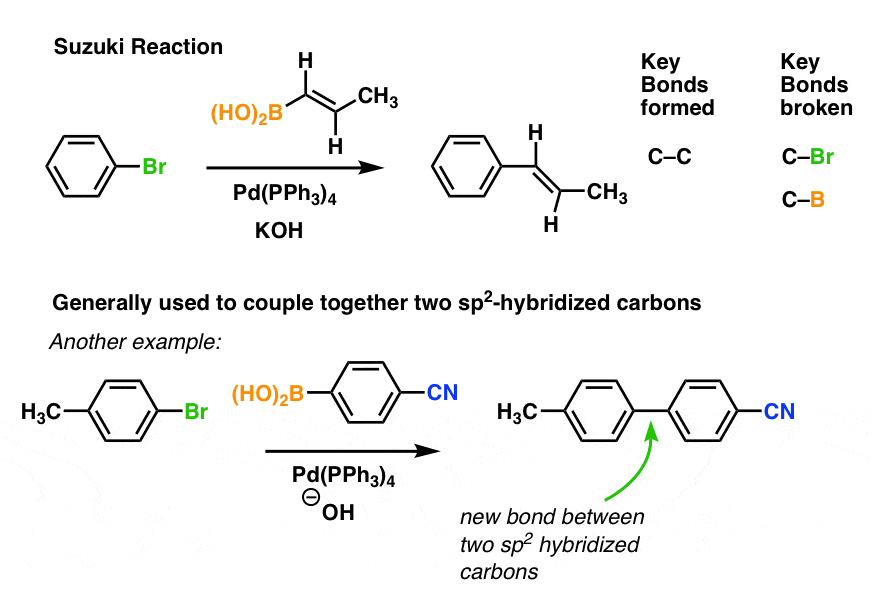
We can control where the bonds form by employing the appropriate precursors. For instance, in the second example above, note that our aryl bromide [1-bromo-4-methylbenzene] has a bromine “para” (opposite) the CH3 group on the aromatic ring. Had we used the “meta” isomer [1-bromo-3-methylbenzene] the new C-C bond would likewise have formed “meta” to the methyl group.
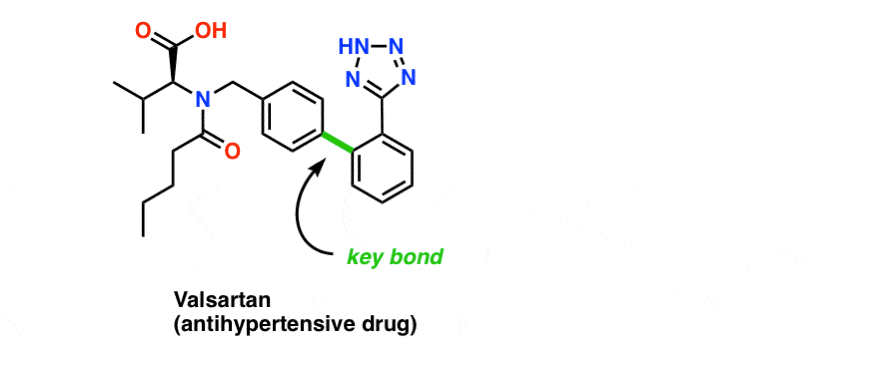
Pd-catalyzed cross coupling allows one to snap together complimentary pieces together like Lego blocks. For bonus points, you might imagine how you could use the Suzuki to build the crucial sp2-sp2 bond in Valsartan, for example.
For further reading and background on the Suzuki, I recommend the resources of Myers and White, and of course Mike Evans’ Organometallic Reader provides a general background for the key concepts of organometallic chemistry.
3. The Heck Reaction
A second important palladium-catalyzed C–C bond forming reaction is actually the great-granddaddy of them all, known as the Heck reaction.
Just as in the Suzuki, one of the coupling partners is an alkenyl or aryl halide (Br, Cl, I) [or sometimes OTf]
Unlike the Suzuki, there is no boronic acid employed. [The names of cross-coupling reactions tend to get assigned based on the identity of the “organometallic” coupling partner]. In the Heck, the coupling partner is a mere alkene.
Here’s an example. Pay close attention to the bonds that form and break here:
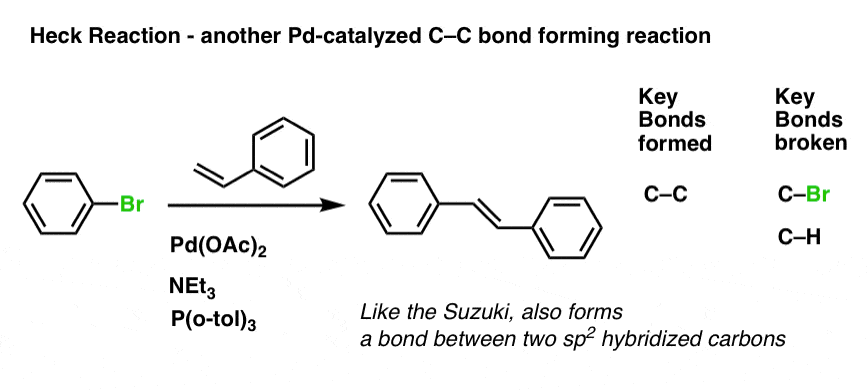
The Heck reaction ends up forming a new C–C bond between the aryl halide and the alkene, and we break both a C-Br and C–H bond (that C-H bond breaking can be tricky to see!).
No doubt by looking at this one example you might find yourself coming up with questions, such as, “what’s up with all those reagents under the arrow such as Pd(OAc)2 , NEt3 and P(o-tol)3 ?”, and “why did the bond form specifically on THAT part of the alkene when there could be two other C-H bonds to break?”. I understand. More on that below.
4. Olefin Metathesis
A third important transition metal catalyzed reaction often covered in introductory organic chemistry is olefin metathesis or sometimes ring-closing metathesis. The transition metal catalyst generally employed here incorporates ruthenium (Ru) as the active metal rather than palladium.
Unlike coupling reactions, which involve alkenyl or aryl halides along with “organometal” coupling partners, the key functional group involved in olefin metathesis are very simple: alkenes.
Simply put, olefin metathesis stitches two alkenes together. It can be done between two individual alkenes (“intermolecular” or “cross-metathesis”) but is more commonly used to form rings from a molecule containing two alkene functional groups.
In the example below, some labelling will help to keep track of what bonds are being formed and broken. Note that we break the double bond between C-1 and C-2 as well as the double bond between C-7 and C-8, while forming a new double bond between C-2 and C-7 (labelled in red). Since there are 4 carbons between C-2 and C-7 we will end up forming a 6-membered ring. [Note: Ring closing metathesis works well for 5, 6, and 7 membered rings (as well as larger ones) but fails if we try to make strained 3- and 4-membered rings]
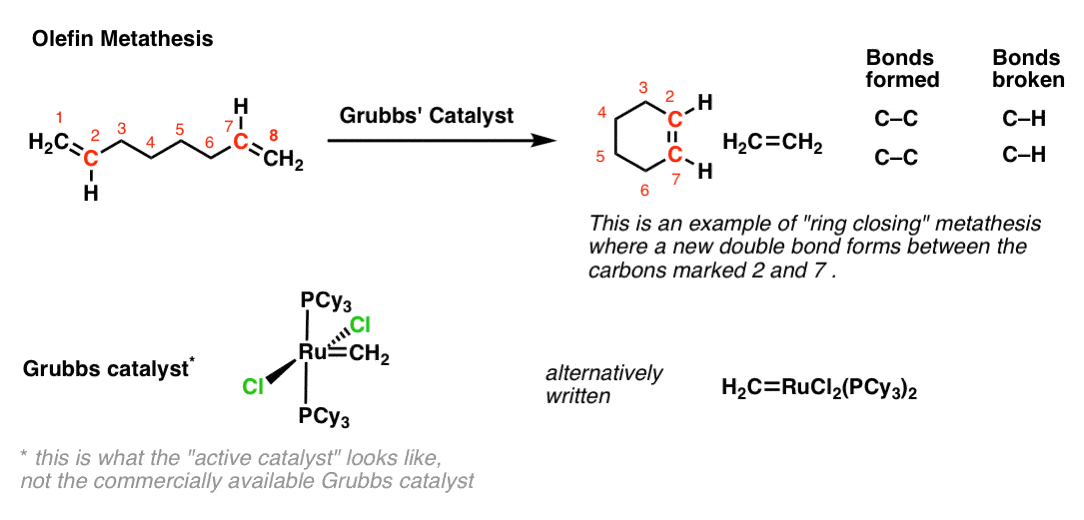
The reaction is catalyzed by a ruthenium compound which has come to be known as “Grubbs’ Catalyst”, after its discoverer, olefin metathesis pioneer (and Nobel Prize winner) Robert H. Grubbs. [I’ve taken a liberty with the structure of this Grubbs catalyst, showing the form of the catalyst when it’s active in the catalytic cycle here and not the commercially available “precatalyst”.]
There are also cases where the reverse reaction (ring-opening metathesis) can be employed, but it generally requires a cyclic alkene with some ring strain present (norbornene is a perfect example). [For the curious, you can read about ring-opening metathesis polymerization (ROMP) here.
One application of olefin metathesis has been to utilize unsaturated alkenes from plant sources (linseed oil, for example) and convert them into terminal alkenes via cross-metathesis with ethene, CH2=CH2. These terminal alkenes are commercially important: Elevance recently built a $40 million production facility to process plant oils via cross metathesis. The advantage with using a feedstock like linseed oil is that it is a renewable resource; until now, petrochemicals have typically been the hydrocarbon source for these products.
If the above seems like an overly brief coverage of three key transition metal catalyzed reactions, I agree. I actually don’t think these reactions belong in most courses, and here’s why.
5. Should Transition Metal Catalyzed Reactions Be Covered In Introductory Organic Courses? Pros and Cons
First, by “cover”, I really mean, “should this be tested”. It’s one thing to tell the class to put the pens down and just have a look at these reactions which have revolutionized organic chemistry. It’s quite another to expect students to study, learn, and most importantly , understand these reactions such that they can reasonably answer questions about them on an exam.
Let’s cover the reasons “Pro” first, and then address the “Con”.
Pro: It’s reflective of modern organic chemistry.
As I described earlier, transition metal catalyzed coupling reactions (the Suzuki and Heck, among others) and Ru-catalyzed olefin metathesis are powerful reactions that get a tremendous amount of use in modern synthetic organic chemistry. By including these reactions in an introductory course, we can convey to students some of the incredible inventiveness and creativity of cutting edge organic chemists and also show them reactions that are more relevant than, say, the Cannizarro reaction or the Sandmeyer reaction, which rarely get any use these days.
Pro: Nobel Prizes were awarded for this work.
Along the same lines, the fact that these reactions have recently won Nobel Prizes for their developers (Suzuki, Heck, Negishi in 2010 for palladium-catalyzed cross-coupling; Grubbs, Schrock, and Chauvin in 2005 for olefin metathesis) is noteworthy and drives home their relevance.
Pro: If it replaces a reaction that gets little use today, like the Canizarro reaction, what’s the loss?
From a teaching perspective, spending a lecture or two on these reactions necessitates dropping some other course material. If it means skipping the Canizarro and Sandmeyer (as previously mentioned), or the malonic ester synthesis, Robinson annulation, the Ruff degradation, Hofmann elimination, and so on – is that really such a huge loss?
Also: It’s more fun to teach advanced stuff
Cross couplings and olefin metathesis represent one of the few additions to the core organic chemistry curriculum in the past 20 years. As unfair as it might sound, adding this material thus provides an opportunity to alleviate some instructor boredom, as well as to challenge students abilities to think through new material. Along the same lines, research-oriented professors often relish the opportunity to teach material that is closer to the heart of modern organic chemistry rather than spend time covering reactions advanced students are unlikely to ever encounter in the lab.
Looking at the other side:
Con – The mechanisms are completely disconnected from the rest of the course
As instructors, we value and teach the importance of understanding reactions and electron flow, not merely memorizing what is going on.
One of the fun things about teaching organic chemistry is showing students how concepts and skills that they learn in the first few weeks of class are still relevant at the very end of the course.
In every reaction from the simplest acid-base reaction to the end of the course in peptide synthesis, students can identify curved arrows and point out nucleophiles and electrophiles. The tools that are useful in chapter 4 on acid-base chemistry are still useful in chapter 24 on peptide synthesis.
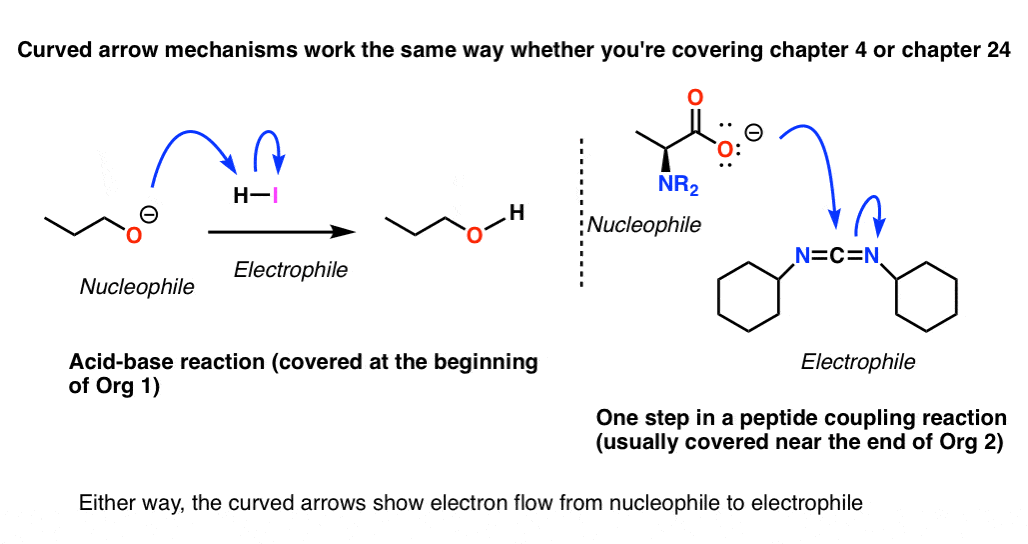
Where do transition metal mechanisms fit into any of this? Yes, of course they can be understood, given enough time, and of course they do follow the core tenets of chemistry in a broad sense.
However the mechanisms themselves are so foreign to a new observer that they require a tremendous amount of work filling in the background if they are to be understood properly.
Try identifying the nucleophile and electrophile in the reaction below. How do the electrons flow?
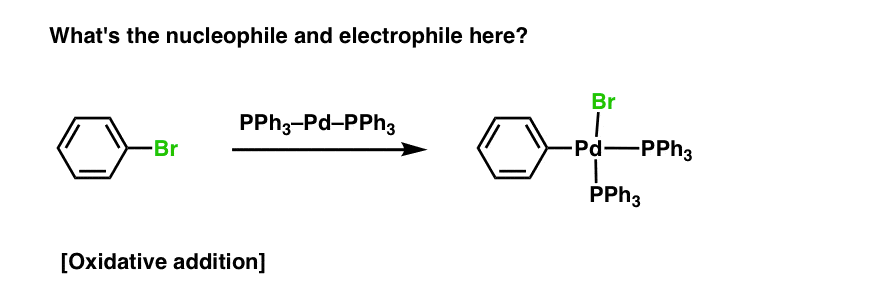
There actually is an electrophile and a nucleophile here, but not in any sense that students are accustomed to seeing.
And this is just oxidative addition, to say nothing of other key organometallic mechanisms – reductive elimination, transmetallation, beta-hydride abstraction and so on. Covering this material is a huge diversion from the main goal of an introductory organic chemistry course: understanding electron flow.
Con: There isn’t enough time to cover the key concepts
Say you want to give students an understanding of how these reactions work. Where do you start?
In the case of the Suzuki, it’s one thing to say that it starts with oxidative addition of Pd into the C-X bond, followed by “transmetallation”, and then followed by reductive elimination. It’s another thing entirely to try to teach it in a course where students have had zero previous exposure to transition metal reaction mechanisms.
And no, putting up a slide which describes the catalytic cycle of the Suzuki is not the same thing as teaching it.
Here’s a partial list of some other concepts that will be unfamiliar to someone trying to learn cross coupling reactions in the context of an introductory organic chemistry course:
- What are “ligands” ?
- What makes ligands “electron-rich” or “electron poor” ?
- how do the structure of ligands affect the rates of oxidative addition and reductive elimination?
- why are phosphorus ligands so important in transition metal chemistry?
- why are “chelating” ligands so common?
- how does one do “electron counting” on a metal complex – and what’s the 18-electron rule?
- How does association and dissociation of ligands on transition metal complexes work?
- what’s an “open” coordination site?
- What factors affect transition metal geometries? Why is palladium square planar and not tetrahedral?
- Why might someone choose Pd(OAc)2 versus Pd(PPh3)4 or other Pd precursors?
- What is beta-hydride abstraction (in the Heck reaction) and when will it occur (or not occur?)
- How and why does transmetallation (e.g. in the Suzuki) happen?
- What’s the initiation step in the Heck reaction – why is it that we often start with Pd(OAc)2 but the first step involves oxidative addition of a Pd(0) species? How did it get to Pd(0) ?
Understanding these concepts is crucial to understanding how these reactions work – and I didn’t even get into such concepts as the different types of d-orbitals in transition metals or the key concept of pi backbonding.
Even in a graduate course devoted specifically to organometallics, these introductory concepts get at least a few lectures’ worth of time. But in a typical organic chemistry course, where there are only about 36 hours of lecture to go over the material, how much detail can possibly be covered?
Given the inevitable time constraints, sacrifices must be made. Often, the specific detail of the reagents involved is skipped.
How do you make the tradeoffs?
In some cases, extra details are skipped altogether. Here is an actual example of an exam question involving the Heck reaction.
What’s really being taught/learned here? Is this really so much better than teaching the ol’ Sandmeyer?
Con: It contradicts the dictum, “don’t memorize, learn the concepts”
The only way to get through Org 2 is to understand the key concepts/patterns, and not try to memorize every new reaction. But given the time constraints, how much “understanding” of organometallic reactions can possibly be achieved?
Preaching the importance of understanding concepts on one hand, and teaching the Suzuki and Heck reactions (or some watered down versions thereof) in a single day on the other, completely goes against this philosophy.
There’s better ways to include more advanced material without introducing all the new concepts that come with transition metal chemistry.
Some examples that come to mind are introducing concepts such as hyperconjugation, orbital symmetry, steric and stereoelectronic effects. These can augment and inform students’ understanding of material they are already learning without burdening them with a overwhelming number of “open loops” that transition metal chemistry introduces.
6. Conclusion: The Heck, Suzuki and Olefin Metathesis Reactions
Next post: Reaction Map – Reactions of Organometallics
Notes
Quiz Yourself

Become a MOC member to see the clickable quiz with answers on the back.

Become a MOC member to see the clickable quiz with answers on the back.

Become a MOC member to see the clickable quiz with answers on the back.
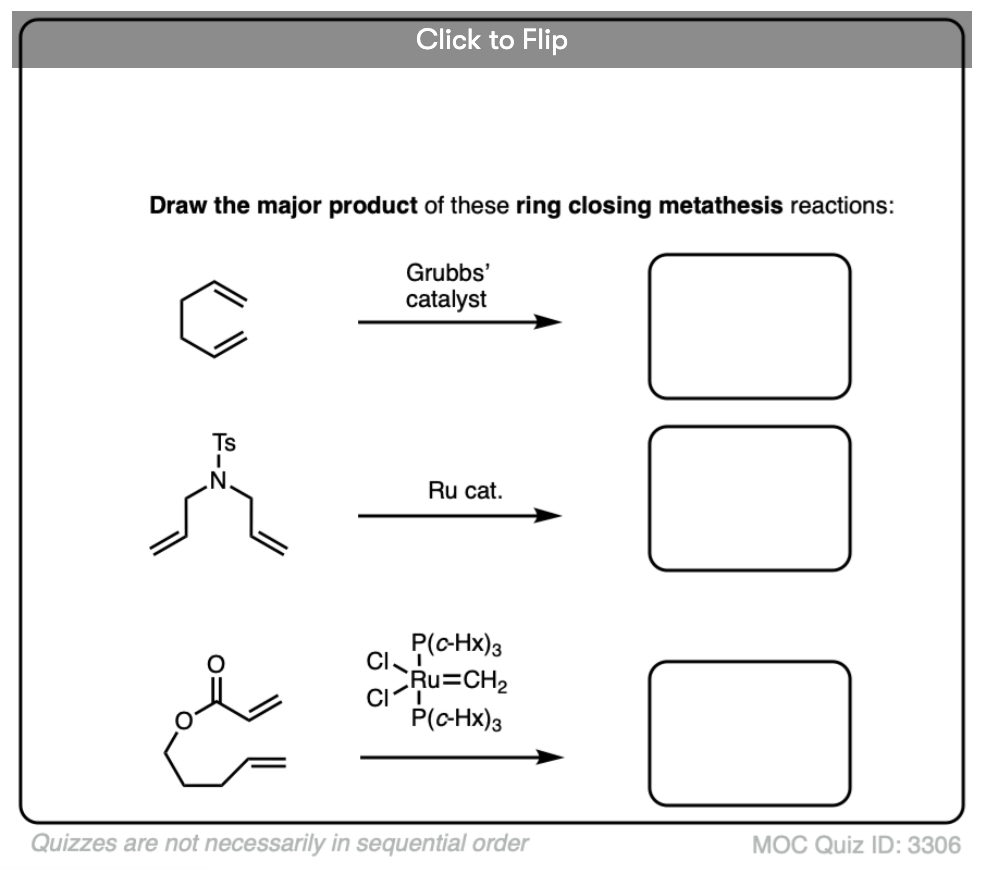
Become a MOC member to see the clickable quiz with answers on the back.
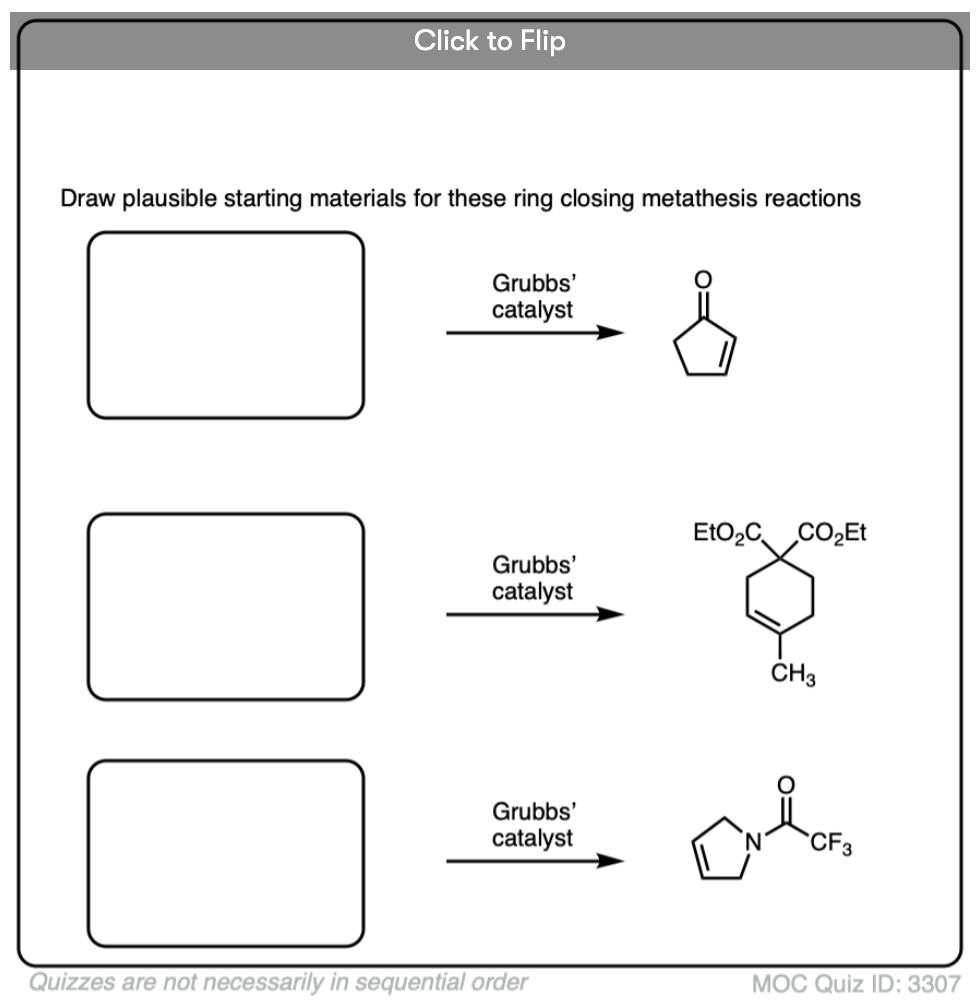
Become a MOC member to see the clickable quiz with answers on the back.
(Advanced) References and Further Reading
These reactions, which are based on fundamental organometallic and inorganic chemistry principles, are indeed challenging to teach in a traditional undergraduate introductory organic chemistry course. That being said, they are commonly included in most curricula nowadays and they are more or less here to stay.
For more information on each of these reactions, here are some links to the relevant entries in the Reaction Guide:
Here are some more pedagogical papers on the above topics:
- Nobel Chemistry in the Laboratory: Synthesis of a Ruthenium Catalyst for Ring-Closing Olefin Metathesis
George E. Greco
Journal of Chemical Education 2007, 84 (12), 1995
DOI: 1021/ed084p1995
This paper describes an experiment suitable for an undergraduate inorganic or organometallic laboratory course – synthesizing the Grubbs’ catalyst using Schlenk techniques and then applying that to an olefin metathesis reaction. - Synthesizing Stilbene by Olefin Metathesis Reaction Using Guided Inquiry To Compare and Contrast Wittig and Metathesis Methodologies
Timothy J. Bannin, Partha P. Datta, Elizabeth T. Kiesewetter, and Matthew K. Kiesewetter
Journal of Chemical Education 2019, 96 (1), 143-147
DOI: 1021/acs.jchemed.8b00313
This paper describes an experiment suitable for undergraduates demonstrating that the olefin metathesis reaction can be a superior alternative to the Wittig reaction for alkene synthesis. With olefin metathesis, the only side product is ethylene, as opposed to the Wittig reaction, which generates Ph3PO, and separating that from the desired product is the bane of organic chemists everywhere. - Suzuki Cross-Coupling Reactions: Synthesis of Unsymmetrical Biaryls in the Organic Laboratory
Christopher S. Callam and Todd L. Lowary
Journal of Chemical Education 2001, 78 (7), 947
DOI: 1021/ed078p947
This describes a straightforward Suzuki reaction for biaryl synthesis, which is actually one of the most common uses for the Suzuki reaction in practice. - Experimenting with a Suzuki–Miyaura Cross-Coupling Reaction That Demonstrates Tolerance toward Aldehyde Groups To Teach Undergraduate Students the Fundamentals of Transition-Metal-Catalyzed Reactions
Jie Dai, Dadong Lu, Tao Ye, Shouyun Yu, and Xu Cheng
Journal of Chemical Education 2019, 96 (11), 2672-2675
DOI: 1021/acs.jchemed.9b00191
This paper describes an experiment designed to highlight the major advantage of Pd-catalyzed cross-coupling reactions – chemoselectivity and functional group tolerance. This is what makes these reactions so practical and widely used. - The Heck Reaction: A Microscale Synthesis Using a Palladium Catalyst
William B. Martin and Laura J. Kateley
Journal of Chemical Education 2000, 77 (6), 757
DOI: 1021/ed077p757
A Heck reaction suitable for an undergraduate laboratory session. - Kinetic Study of the Heck Reaction: An Interdisciplinary Experiment
Christel Gozzi and Naoual Bouzidi
Journal of Chemical Education 2008, 85 (8), 1126
DOI: 1021/ed085p1126
This paper states, “We think that students should have a global view of chemistry. They need to realize that fields, such as organic chemistry, catalysis, kinetics, analytical chemistry, and chemical engineering, are interdependent and cannot always be studied separately”.
Why palladium is used as a catalyst in most of these reactions?
do you ever consider adding those organometallic concepts to your web page ?
it would be very helpfull in my organometalic course !
These are the most widely used organic reactions and should be covered during organic chem II rather than some pointless 30+ step total synthesis.
…rather than some pointless… Who said anything in favor of pointless 30+ step total syntheses? This is a straw-man argument, and doesn’t help the discussion.
Interesting. I think it is certainly worth a mention, but I agree that the full mechanism needs significantly more background. However, there are enough other reactions that are in the undergrad curriculum that are mechanistically complex, and we gloss over (or simplify) mechanistic details. The Sandmeyer reaction comes to mind again. Along with benzylic oxidation using KMnO4, oxidation with dichromate, .. I can go on. I really think a strongly mechanistic approach is a great way to teach organic; but the question is: – Is there a place for important reactions, even though we’re not going into the mechanism right now? If the answer to this is yes, then I think these reactions deserve more of a mention than some of the ‘classics’ that are currently covered.
Not sure what the answer is, but it is an interesting discussion.
I completely agree with your analysis, and your conclusion that these reactions shouldn’t be taught as a part of the core organic chemistry curriculum. But if we’re going to be graduating informed, practicing chemists, they do need to learn some of this material.
It speaks to the need for a new class covering organometallic chemistry. Where I went to undergraduate, neither the organic nor the inorganic profs wanted to tackle that course. But it’s really integral to the field these days.
It definitely has a place in an undergraduate chemistry curriculum – just not in the introductory organic course! Ideally it should be taught concurrently or after a standard introductory course in inorganic chemistry where students are exposed to transition metal reaction mechanisms, orbitals, bonding, geometry, and so on.
Having said that, the main route to Celecoxib does not involve TM-catalysed couplings, to my knowledge (but this was some time ago I last looked at this literature)
Great topic today, and a good read. I admit I am of the same opinion.