Blog
Why organic chemistry is hard (2)
Last updated: October 6th, 2022 |
Today’s post is about how a simple reaction can lead to all kinds of complications.
I’m talking about the Grignard reaction here, although the principles can be applied to other reactions as well.
The Grignard reaction is simple enough. It involves a Grignard reagent – that’s some kind of carbon, attached to a magnesium salt – adding to a carbonyl compound such as an aldehyde, ketone, or ester.
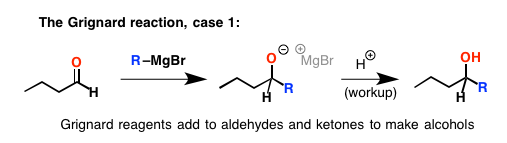
The reaction with ketones and aldehydes is pretty straightforward: Grignard reagents are nucleophiles, and they add to the electrophilic carbonyl carbon, breaking the C=O bond and forming an alcohol after acid is added.
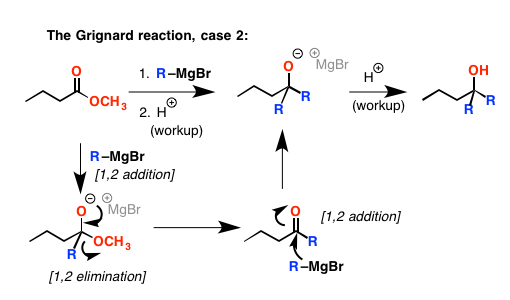
Things get a little bit more complicated with esters. If you add an excess of Grignard reagent, it adds to the carbonyl carbon too. Except in this case, it doesn’t stop there. One of the lone pairs on the newly negative oxygen atom can re-form a π bond with the neighboring carbon. This results in breakage of the bond between the carbon and the OR, which leads to its ejection and the overall formation of a ketone. But it doesn’t stop there. Now we have a ketone, which reacts really well with Grignard reagents – and we add a second equivalent, forming a new alcohol (after the acid workup).
What about carboxylic acids? You might expect they’d behave the same as esters. But no. Grignard reagents are strong bases, see, and when combined with a carboxylic acid, they’re protonated. The resulting negatively charged carboxylate salt (that’s the conjugate base of a carboxylic acid) is then pretty much impregnable to nucleophilic attack due to the strongly donating O(-) group. So it just sits there in solution until acid is added, giving us back our starting material.
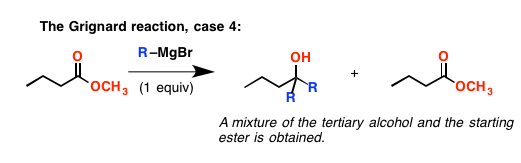
Let’s go back to esters for a minute. You might think that if you added only one equivalent of Grignard reagent, you could just get it to stop at the ketone stage. Well……no. What happens is that the rate of the elimination reaction is fast – faster than addition of Grignard to the ester – and the ketone outcompetes the ester for reagent. So at the end of the day you end up with a product where two equivalents of Grignard have added to the starting ester, and about half an equivalent of leftover ester.
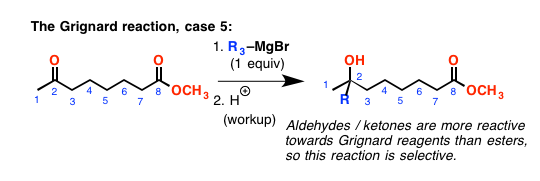
Which brings up the next point: ketones and aldehydes are more reactive than esters. [The order of reactivity, by the way, goes aldehyde > ketone > > ester]. So if you have a molecule with a ketone and an ester on it, and you add one equivalent of Grignard reagent, it will add to the ketone selectively.
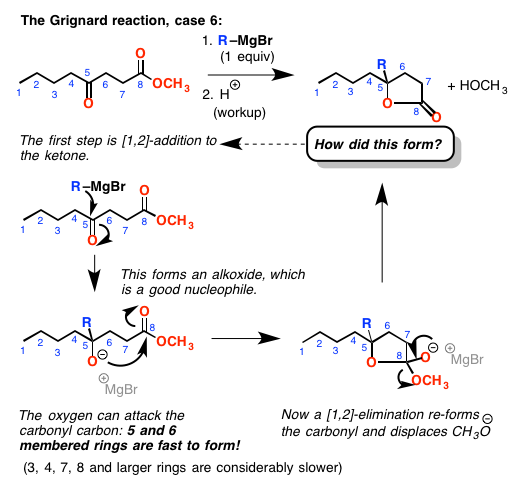
But watch out! When you add to a ketone or aldehyde, you form a new alkoxide (negatively charged oxygen). These are good nucleophiles! If the alkoxide oxygen is 5 or 6 bonds away from the ester – and if it can reach – it is possible for it to attack the ester carbonyl, doing a [1,2]-addition / [1,2]-elimination reaction to form a lactone (cyclic ester).
Why do 5 and 6- membered rings form quickly, whereas 3, 4, 7 (and higher) membered rings do not? Long story, but that’s just the way it is. It’s yet another thing you need to watch out for.
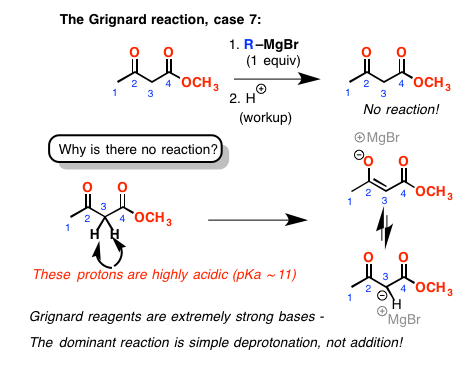
Finally, we come to this last example. We have a ketone and an ester. We can’t form a ring. You might think it would just add to the ketone here. Again, not the case. This time, it turns out that having those two carbonyl groups in close proximity has made the protons on the adjacent CH2 group very acidic. The Grignard, being a strong base, simply removes a proton and doesn’t add to the carbonyl carbon at all. [Yet another complicating factor: ketones like this one are largely present in their enol form, due to a stabilizing hydrogen bond between the O-H and the ester carbonyl oxygen.]. After acidic workup, we end up with starting material.
The lesson: Functional groups and reagents can interact in funky ways that can be rationalized in retrospect, but are damn hard for a newbie to predict. It’s part of what makes organic chemistry challenging and frustrating, to be sure, but also deep and rewarding. That’s just the way it is, folks.
Key points:
1) Some reagents can act both as nucleophiles and as bases. The trick is identifying when this is.
2) Aldehydes are more reactive towards nucleophiles than ketones, which are more reactive than esters.
3) Once a Grignard adds to an ester, there ain’t no stoppin’ it.
4) Five and six-membered ring formation is fast.
5) 1,3-dicarbonyls are unusually acidic, and this can complicate Grignard reactions.

Your explanation is way better than my organic lecturer! thank you so much for making everything so simple!
OK, I’m glad you find the site helpful Nagina!
In the sentence above which begins “So at the end of the day you end up with a product where two equivalents of Grignard have added…”, it might be clearer to say “So at the end of the day you end up with a product where two equivalents of Grignard have added to the starting ester…”
This is a really good website, keep up the good work.
Changed it. Thank you!
Your site is very clear and helpful!! I love that each step is clearly explained and the colors and numbers are so easy to follow. Thank you!!!!!
thanks a lot! great article. thanks for covering all possible details.
If the alpha hydrogen on a ketone or aldehyde has a pka of around 17-19, why wouldn’t the carbanion (a very strong base) abstract a alpha hydrogen.
Generally, additions to carbonyls are faster than removal of the alpha proton unless the carbonyl is sterically hindered. One reason it’s not faster is that the C-H is only acidic when it’s in a conformation where the bond is aligned with the p-orbitals of the carbonyl.
Great article! Thank you :-)
hi, if a grignard reagent in a grignard reaction to form a secondary alcohol reacted with CO2 and formed a metal carboxylate, why would this side reaction not be problematic in the reaction?
I think I need more context from you here. Why would you deliberately be adding CO2 to a reaction for the formation of a secondary alcohol?
Great site. Sorry about the professor thing not working out, but (as you know) your explanations and detail to the point of pointing out anomalies are extremely helpful to us organic chemistry students. Thank you.