Home / The Wolff-Kishner, Clemmensen, And Other Carbonyl Reductions
Reactions of Aromatic Molecules
The Wolff-Kishner, Clemmensen, And Other Carbonyl Reductions
Last updated: March 21st, 2025 |
The Wolff-Kishner, Clemmensen, and Other Fun Reductions
In this post we go through 4 ways of reducing C=O to CH2 including:
- The Wolff-Kishner reaction
- The Clemmensen reduction
- Catalytic hydrogenation
- Thioacetal formation and reduction
But first: why might you want to do this? Well, it’s a key component in a common little synthesis problem I like to call “The Great Friedel-Crafts Workaround”.
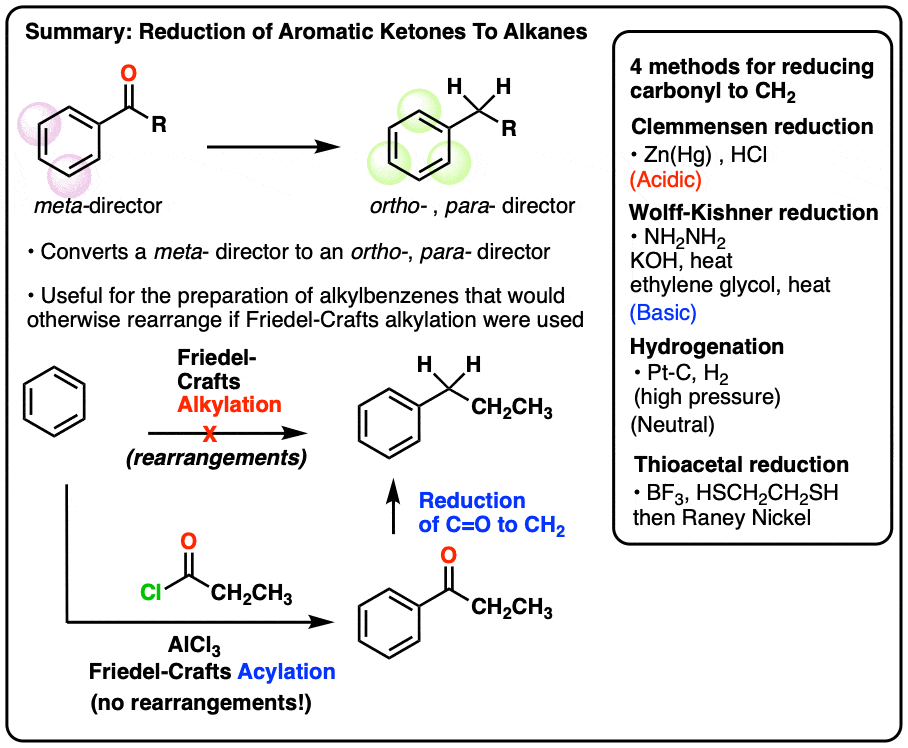
Table of Contents
- The Great Friedel-Crafts Workaround
- The Wolff-Kishner Reduction Of Ketones
- Mechanism Of The Wolff-Kishner Reaction
- The Clemmensen Reduction of Ketones
- Why Would You Prefer The Wolff-Kishner Over the Clemmensen, Or Vice Versa?
- A Third Method For Carbonyl Reduction: Catalytic Hydrogenation
- Reduction of Thioacetals
- A Workaround Example
- A Final Note: Reversing Polarity
- Notes
- Quiz Yourself!
- (Advanced) References and Further Reading
1. The Great Friedel-Crafts Workaround
A recent post covered oxidations on the “benzylic” carbon (i.e. on the carbon adjacent to the aromatic ring). We showed that benzylic C-H bonds are unusually weak, and can be converted relatively easily (and selectively!) to C–Br or C–O bonds.
Today we’re going to go in the reverse direction and address reduction of the benzylic carbon, notably reduction of ketones (C=O) to alkyl (CH2).
This is particularly important because of the Great Friedel-Crafts Workaround.
What’s that, you ask?
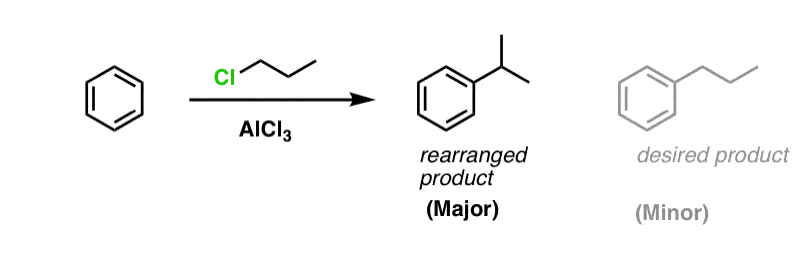
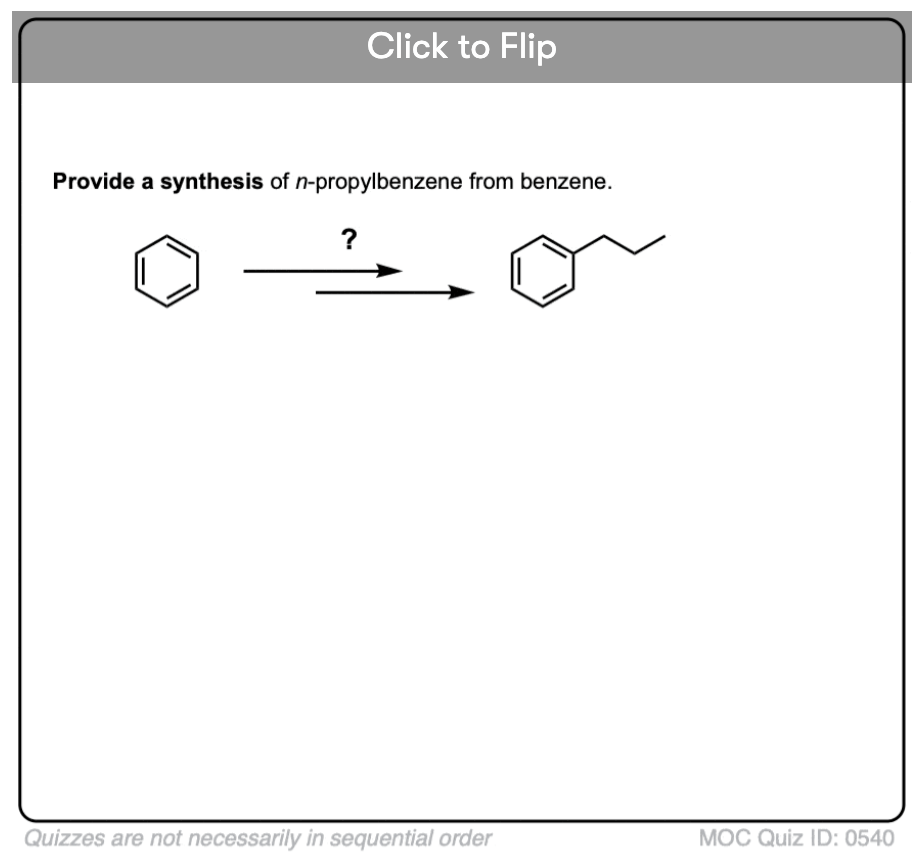
You may recall that Friedel-Crafts alkylation of aromatic rings with primary alkyl halides can result in carbocation rearrangements. For example, attempting a Friedel-Crafts alkylation of benzene with propyl chloride results in isopropylbenzene, not propyl benzene.
The Great Friedel-Crafts Workaround solves this issue. We begin with a Friedel-Crafts acylation, which proceeds without rearrangement, and follow by reducing the ketone down to CH2.
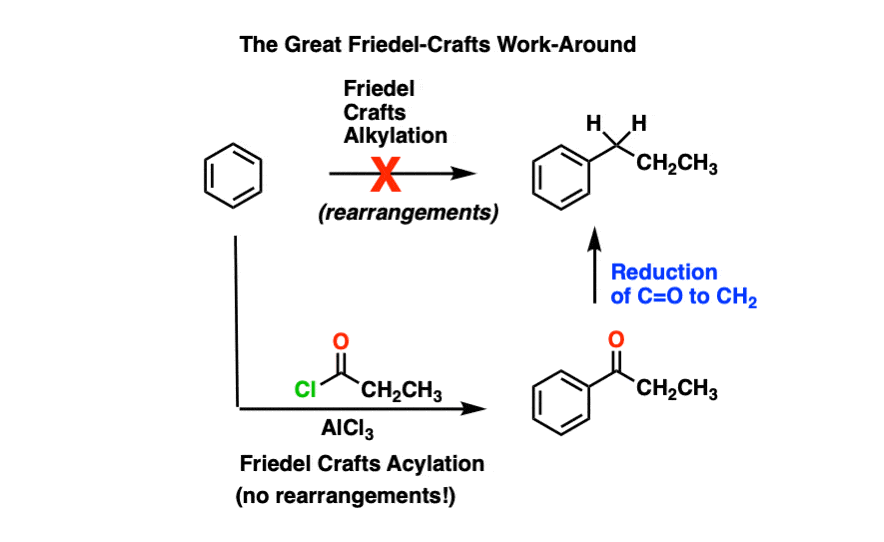
So how can we reduce the ketone down to an alkane? Four ways.
2. The Wolff-Kishner Reduction Of Ketones
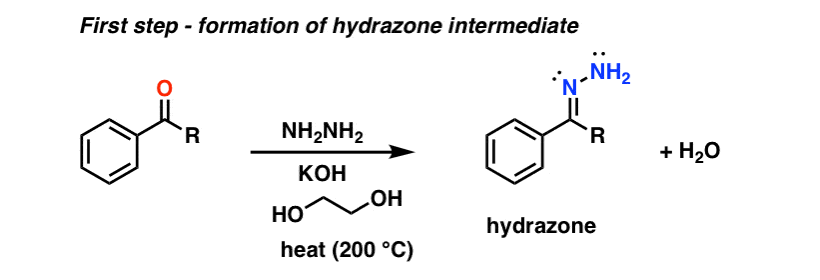
The Wolff Kishner reduction of ketones utilizes hydrazine (NH2NH2) as the reducing agent in the presence of strong base (KOH) in a high-boiling protic solvent (ethylene glycol, HO-CH2CH2-OH, boiling point 197 °C).
The driving force for the reaction is the conversion of hydrazine to nitrogen gas.
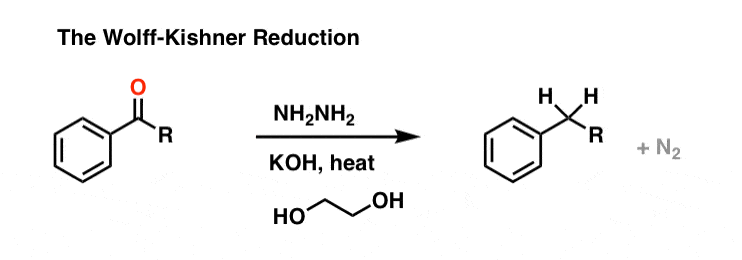
This is not exactly a gentle process; heating to almost 200 °C is required to make the reaction occur at a reasonable rate. [Note 1]
The first step is formation of a hydrazone from the ketone (hydrazones are a cousin of imines, which we cover later in the course). Hydrazine (NH2NH2) adds to the carbonyl, and following a series of proton transfer steps, water is expelled. Click here to see an image of the mechanism for hydrazone formation. (link to image)
Once the hydrazone is formed, the real action in the Wolff-Kishner begins!
3. Mechanism Of The Wolff-Kishner Reaction
The NH2 of the hydrazone is reasonably acidic (pKa about 21) and can be deprotonated by strong base at a high enough temperature (the base is likely the conjugate base of ethylene glycol, not KOH). This deprotonation appears to be the rate-limiting step.
The next step is the trickiest: protonation on the carbon. With the caveat that resonance forms don’t really exist, it can be helpful to imagine forming the resonance form of this species that has a negative charge on the carbon, and then protonating it with solvent (ethylene glycol).
This gives a species with a nitrogen-nitrogen double bond , which , after deprotonation by base, decomposes irreversibly to give nitrogen gas and a carbanion (i.e. a negatively charged carbon).
Protonation of the carbon completes the process.
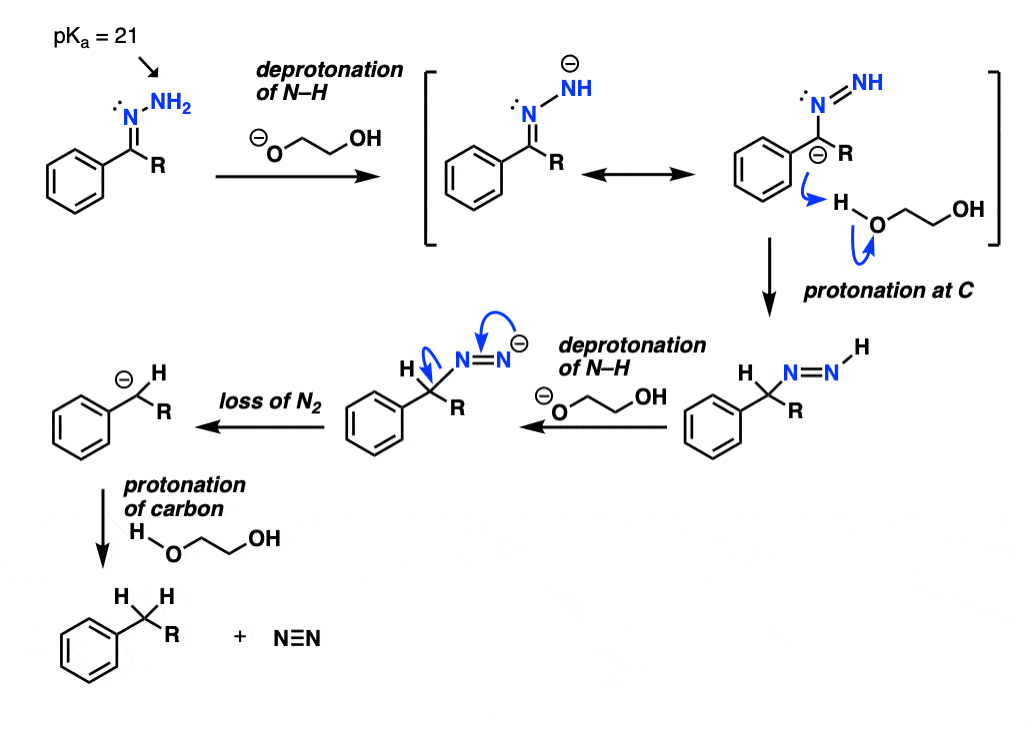
Hover for full arrow-pushing mechanism or click on this link.
4. The Clemmensen Reduction of Ketones
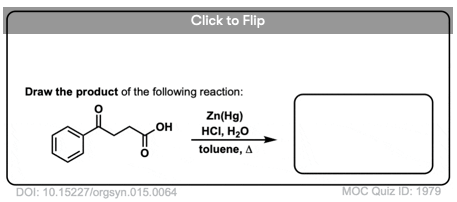
A second way to go about reducing the carbonyl of an aromatic ketone is to use a reaction known as the Clemmensen Reduction. The reductant here is “zinc amalgam” (Zn-Hg) which is used under acidic conditions; one method calls for the presence of aqueous HCl, for example:
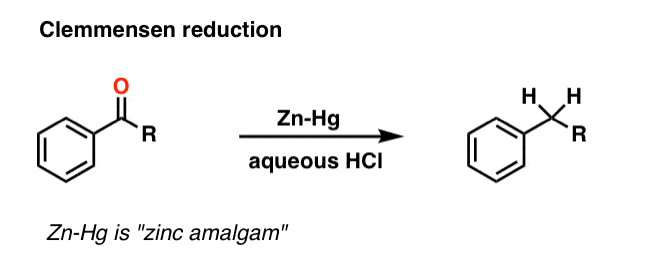
This process works best for aromatic ketones; non-aromatic ketones, not so much. The mechanism has not been thoroughly worked out; it’s thought to occur through a series of one-electron transfers from zinc amalgam.
5. Why Would You Prefer The Wolff-Kishner Over the Clemmensen, Or Vice Versa?
It’s somewhat rare to encounter conditions in an introductory class where a Wolff Kishner would be called for over a Clemmensen, or vice versa, but here are some things to think about.
- The Wolff-Kishner is done under strongly basic conditions using high heat in a polar protic solvent.
- The Clemmensen is performed in strongly acidic conditions. If you have a protecting group somewhere which can be removed with acid, such as an acetal or silyl ether, consider an alternative.
Two other methods deserve mention, although you might not seen them covered until later in the course when ketone chemistry is addressed.
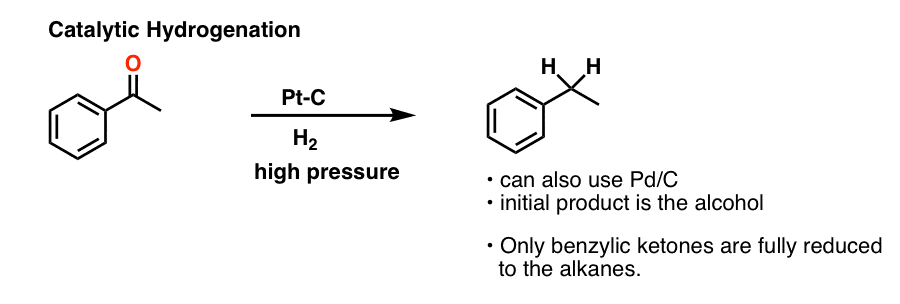
6. A Third Method For Carbonyl Reduction: Catalytic Hydrogenation
The first is catalytic hydrogenation, i.e. using a metal catalyst such as Pd/C or Pt/C with hydrogen gas (H2).
We’ve mostly seen catalytic hydrogenation used for reducing alkenes and alkynes, but it can also be used for ketones if you crank on it enough (i.e. higher temperatures, with higher pressure of H2).
One subtle tweak in conditions, sometimes not mentioned, is that platinum on carbon (Pt-C) or platinum oxide (PtO2) is often used instead of palladium (Pd).
Normally, reduction of ketones usually stops at the alcohol stage. However, in the case where the alcohol is on a benzylic position, (i.e. on a carbon adjacent to an aromatic ring) reduction with can occur further to the alkane (recall that bonds at benzylic positions tend to be easier to break, since the adjacent aromatic ring can donate electron density to them).
7. Reduction of Thioacetals
The second method that sees use is conversion of the ketone to a “thioacetal” with HS-CH2CH2-SH and a Lewis acid such as BF3 . [Note 2] This is followed by treating the thioacetal with a reducing agent known as Raney Nickel: it’s a form of finely divided nickel containing adsorbed hydrogen that cleaves C-S bonds to give C-H bonds, through a somewhat mysterious process also thought to involve free-radicals. [See: Reagents – Raney Nickel]
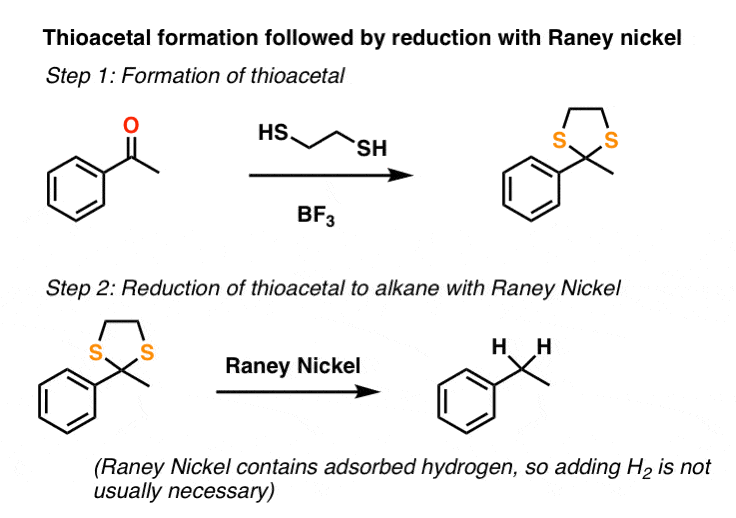
Unlike the Clemmensen and catalytic hydrogenation, thioacetal/Raney nickel method isn’t limited merely to benzylic ketones. It can be used to completely “disappear” an aldehyde or ketone, as was part of the strategy in Woodward’s synthesis of erythromycin.
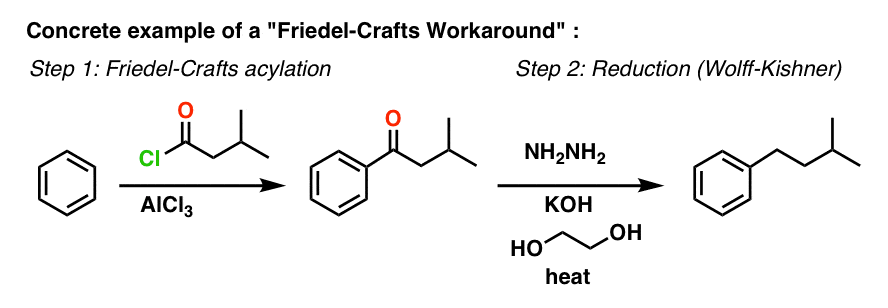
8. A Workaround Example
With these methods in our toolbox, we can now fill in the vague description “reduction” over the arrow with something a lot more specific.
Here’s a concrete example of a Friedel-Crafts Workaround:
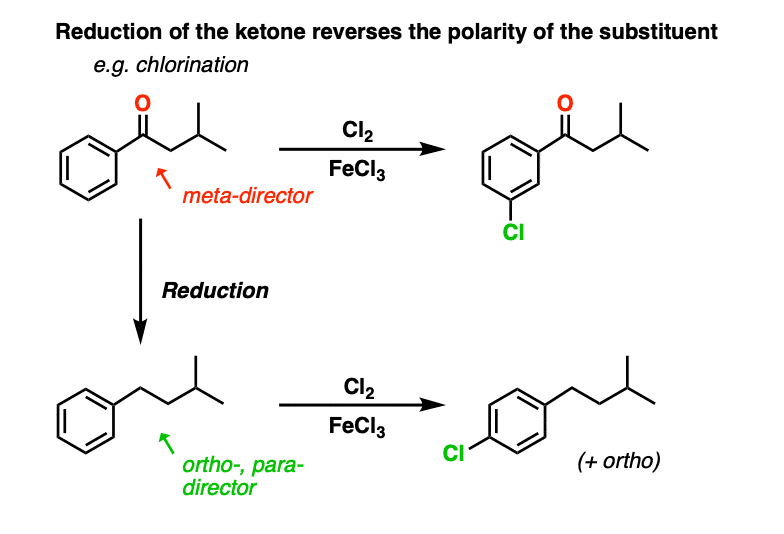
9. A Final Note: Reversing Polarity
A final thing to note here is that reduction of a benzylic ketone to the alkane reverses the polarity of the substituent. It converts an electron-withdrawing meta-director (an acyl group) into an electron-donating ortho-, para- director. We’ll have more to say about this when we address synthesis in aromatic compounds, but just take a gander at these two examples….
Two more examples of reversing the polarity of a substituent is reduction of the nitro group and Baeyer-Villiger oxidation of a ketone to an ester. We’ll cover those next!
Notes
Related Articles
- More Reactions on the Aromatic Sidechain: Reduction of Nitro Groups and the Baeyer Villiger
- Hydrates, Hemiacetals, and Acetals
- Synthesis of Benzene Derivatives (2) – Polarity Reversal
- Aromatic Synthesis (1) – “Order Of Operations”
- Clemmensen Reduction (MOC Membership) of Ketones/Aldehydes to Alkanes
- Wolff Kishner Reaction (MOC Membership)
- Conversion of thioacetals to alkanes using Raney Nickel
- EAS Reactions (3) – Friedel-Crafts Acylation and Friedel-Crafts Alkylation
- Aromatic Reactions and Synthesis Practice (MOC Membership)
Note 1. One way of getting around the requirement for high heat in the Wolff-Kishner is to use a strong base like t-BuOK in DMSO, which can be done around room temperature. [Ref: J. Am. Chem. Soc, 1962, 84, 1734-1735.]
Note 2. , Lewis acids such as BF3 or ZnCl2 are commonly used for this reaction [see this Org Syn reference] although in practice, protic acids such as HCl are perfectly fine if the starting material is an aldehyde or ketone [see this Org Syn reference]
In Corey and Seebach’s method for making 1,3-dithiane, the starting materials are 1,3-propanedithiol and dimethoxymethane (CH3OCH2OCH3), an equivalent of formaldehyde. BF3, a strong Lewis acid, gave better yields in this procedure than did anhydrous acid, subsequently BF3 seems to be the Lewis acid of choice in organic chemistry textbooks despite the fact that most thioacetal formation reactions seen in introductory courses are from aldehydes or ketones, not acetals.
Quiz Yourself!
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.

Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
Become a MOC member to see the clickable quiz with answers on the back.
(Advanced) References and Further Reading
- Chemischen Institut der Universität Jena: Methode zum Ersatz des Sauerstoffatoms der Ketone und Aldehyde durch Wasserstoff. [Erste Abhandlung.]
Wolff, L.
Lieb. Ann. Chem. 1912 394 (1), 86
DOI: 10.1002/jlac.19123940107
The original paper by Ludwig Wolff on the reduction of aldehydes and ketones with hydrazine. - The Wolff-Kishner Reaction of Hydrazones
Szmant, H. H.; Harmuth, C. H.
J. Am. Chem. Soc.1964, 86 (14), 2909
DOI: 10.1021/ja01068a028
A very nice Physical Organic study on the decomposition of hydrazones, involving Hammett plots (a classic tool in Physical Organic Chemistry), determining the relationship between the electron density of the carbonyl carbon and the mechanism. - Reduction of Steroid Ketones and other Carbonyl Compounds by Modified Wolff-Kishner Method
Huang-Minlon
J. Am. Chem. Soc.1949, 71 (10), 3301
DOI: 10.1021/ja01178a008
A one-pot modification for the Wolff-Kishner reaction that involves distilling off the excess water and hydrazine before heating the hydrazine. This results improved yields and significantly shorter reaction times. - REDUCTION OF KETONES BY USE OF THE TOSYLHYDRAZONE DERIVATIVES: ANDROSTAN-17 β-OL
Caglioti
Org. Synth. 1972,52, 122
DOI: 10.15227/orgsyn.052.0122
Due to the harsh conditions involved in the standard Wolff-Kishner reduction, a number of variations have been developed with milder conditions more amenable for organic synthesis. The use of tosylhydrazide instead of hydrazine allows much gentler reaction conditions – the tosylhydrazone can be reduced with NaBH4 in refluxing methanol (68 °C vs. 200 °C!). - γ-PHENYLBUTYRIC ACID
L. Martin
Org. Synth. 1935, 15, 64
DOI: 10.15227/orgsyn.015.0064
A reliable, tested procedure for a simple Clemmensen Reduction that demonstrates how this reaction can be used to reduce a ketone in the presence of a carboxylic acid. - Elrik Clemmensen: Reduktion von Ketonen und Alahyden su den entspcechenden Kohlenwaaserstoflbn unter Anwendung von amalgamiertem Zink und Salesaure.
Clemmensen, E.
Chem. Ber. 1913 46 (2), 1837-1843
DOI: 10.1002/cber.19130460292
The original paper by Clemmensen on the reduction of aldehydes and ketones with Zn metal in acid. Note that even though this was submitted to a German journal and written in German, Clemmensen was working at Parke-Davis, in the U.S.Two reviews on the Clemmensen Reduction: - THE CLEMMENSEN REDUCTION
Martin, E. L.
Org. React. 1942, 1, 155
DOI: 10.1002/0471264180.or001.07 - CLEMMENSEN REDUCTION OF KETONES IN ANHYDROUS ORGANIC SOLVENTS
Vedejs, E.
Org. React. 1975, 22, 401
DOI: 10.1002/0471264180.or022.03 - Stereoselective total synthesis of (–)-pumiliotoxin C by an aqueous intramolecular acylnitroso Diels–Alder approach
Masaichi Naruse, Sakae Aoyagi and Chihiro Kibayashi
J. Chem. Soc., Perkin Trans. 1, 1996, 1113-1124
DOI: 10.1039/P19960001113
Clemmensen reductions can be used in total synthesis due to the mild conditions – in this case 34 to 35 is a Clemmensen reduction that selectively reduces a ketone in the presence of an amide.
00 General Chemistry Review
01 Bonding, Structure, and Resonance
- How Do We Know Methane (CH4) Is Tetrahedral?
- Hybrid Orbitals and Hybridization
- How To Determine Hybridization: A Shortcut
- Orbital Hybridization And Bond Strengths
- Sigma bonds come in six varieties: Pi bonds come in one
- A Key Skill: How to Calculate Formal Charge
- The Four Intermolecular Forces and How They Affect Boiling Points
- 3 Trends That Affect Boiling Points
- How To Use Electronegativity To Determine Electron Density (and why NOT to trust formal charge)
- Introduction to Resonance
- How To Use Curved Arrows To Interchange Resonance Forms
- Evaluating Resonance Forms (1) - The Rule of Least Charges
- How To Find The Best Resonance Structure By Applying Electronegativity
- Evaluating Resonance Structures With Negative Charges
- Evaluating Resonance Structures With Positive Charge
- Exploring Resonance: Pi-Donation
- Exploring Resonance: Pi-acceptors
- In Summary: Evaluating Resonance Structures
- Drawing Resonance Structures: 3 Common Mistakes To Avoid
- How to apply electronegativity and resonance to understand reactivity
- Bond Hybridization Practice
- Structure and Bonding Practice Quizzes
- Resonance Structures Practice
02 Acid Base Reactions
- Introduction to Acid-Base Reactions
- Acid Base Reactions In Organic Chemistry
- The Stronger The Acid, The Weaker The Conjugate Base
- Walkthrough of Acid-Base Reactions (3) - Acidity Trends
- Five Key Factors That Influence Acidity
- Acid-Base Reactions: Introducing Ka and pKa
- How to Use a pKa Table
- The pKa Table Is Your Friend
- A Handy Rule of Thumb for Acid-Base Reactions
- Acid Base Reactions Are Fast
- pKa Values Span 60 Orders Of Magnitude
- How Protonation and Deprotonation Affect Reactivity
- Acid Base Practice Problems
03 Alkanes and Nomenclature
- Meet the (Most Important) Functional Groups
- Condensed Formulas: Deciphering What the Brackets Mean
- Hidden Hydrogens, Hidden Lone Pairs, Hidden Counterions
- Don't Be Futyl, Learn The Butyls
- Primary, Secondary, Tertiary, Quaternary In Organic Chemistry
- Branching, and Its Affect On Melting and Boiling Points
- The Many, Many Ways of Drawing Butane
- Wedge And Dash Convention For Tetrahedral Carbon
- Common Mistakes in Organic Chemistry: Pentavalent Carbon
- Table of Functional Group Priorities for Nomenclature
- Summary Sheet - Alkane Nomenclature
- Organic Chemistry IUPAC Nomenclature Demystified With A Simple Puzzle Piece Approach
- Boiling Point Quizzes
- Organic Chemistry Nomenclature Quizzes
04 Conformations and Cycloalkanes
- Staggered vs Eclipsed Conformations of Ethane
- Conformational Isomers of Propane
- Newman Projection of Butane (and Gauche Conformation)
- Introduction to Cycloalkanes
- Geometric Isomers In Small Rings: Cis And Trans Cycloalkanes
- Calculation of Ring Strain In Cycloalkanes
- Cycloalkanes - Ring Strain In Cyclopropane And Cyclobutane
- Cyclohexane Conformations
- Cyclohexane Chair Conformation: An Aerial Tour
- How To Draw The Cyclohexane Chair Conformation
- The Cyclohexane Chair Flip
- The Cyclohexane Chair Flip - Energy Diagram
- Substituted Cyclohexanes - Axial vs Equatorial
- Ranking The Bulkiness Of Substituents On Cyclohexanes: "A-Values"
- Cyclohexane Chair Conformation Stability: Which One Is Lower Energy?
- Fused Rings - Cis-Decalin and Trans-Decalin
- Naming Bicyclic Compounds - Fused, Bridged, and Spiro
- Bredt's Rule (And Summary of Cycloalkanes)
- Newman Projection Practice
- Cycloalkanes Practice Problems
05 A Primer On Organic Reactions
- The Most Important Question To Ask When Learning a New Reaction
- Curved Arrows (for reactions)
- Nucleophiles and Electrophiles
- The Three Classes of Nucleophiles
- Nucleophilicity vs. Basicity
- What Makes A Good Nucleophile?
- What Makes A Good Leaving Group?
- 3 Factors That Stabilize Carbocations
- Equilibrium and Energy Relationships
- 7 Factors that stabilize negative charge in organic chemistry
- 7 Factors That Stabilize Positive Charge in Organic Chemistry
- What's a Transition State?
- Hammond's Postulate
- Learning Organic Chemistry Reactions: A Checklist (PDF)
- Introduction to Oxidative Cleavage Reactions
06 Free Radical Reactions
- Free Radical Reactions
- 3 Factors That Stabilize Free Radicals
- Bond Strengths And Radical Stability
- Free Radical Initiation: Why Is "Light" Or "Heat" Required?
- Initiation, Propagation, Termination
- Monochlorination Products Of Propane, Pentane, And Other Alkanes
- Selectivity In Free Radical Reactions
- Selectivity in Free Radical Reactions: Bromination vs. Chlorination
- Halogenation At Tiffany's
- Allylic Bromination
- Bonus Topic: Allylic Rearrangements
- In Summary: Free Radicals
- Synthesis (2) - Reactions of Alkanes
- Free Radicals Practice Quizzes
07 Stereochemistry and Chirality
- Types of Isomers: Constitutional Isomers, Stereoisomers, Enantiomers, and Diastereomers
- How To Draw The Enantiomer Of A Chiral Molecule
- How To Draw A Bond Rotation
- Introduction to Assigning (R) and (S): The Cahn-Ingold-Prelog Rules
- Assigning Cahn-Ingold-Prelog (CIP) Priorities (2) - The Method of Dots
- Enantiomers vs Diastereomers vs The Same? Two Methods For Solving Problems
- Assigning R/S To Newman Projections (And Converting Newman To Line Diagrams)
- How To Determine R and S Configurations On A Fischer Projection
- The Meso Trap
- Optical Rotation, Optical Activity, and Specific Rotation
- Optical Purity and Enantiomeric Excess
- What's a Racemic Mixture?
- Chiral Allenes And Chiral Axes
- Stereochemistry Practice Problems and Quizzes
08 Substitution Reactions
- Nucleophilic Substitution Reactions - Introduction
- Two Types of Nucleophilic Substitution Reactions
- The SN2 Mechanism
- Why the SN2 Reaction Is Powerful
- The SN1 Mechanism
- The Conjugate Acid Is A Better Leaving Group
- Comparing the SN1 and SN2 Reactions
- Polar Protic? Polar Aprotic? Nonpolar? All About Solvents
- Steric Hindrance is Like a Fat Goalie
- Common Blind Spot: Intramolecular Reactions
- Substitution Practice - SN1
- Substitution Practice - SN2
09 Elimination Reactions
- Elimination Reactions (1): Introduction And The Key Pattern
- Elimination Reactions (2): The Zaitsev Rule
- Elimination Reactions Are Favored By Heat
- Two Elimination Reaction Patterns
- The E1 Reaction
- The E2 Mechanism
- E1 vs E2: Comparing the E1 and E2 Reactions
- Antiperiplanar Relationships: The E2 Reaction and Cyclohexane Rings
- Bulky Bases in Elimination Reactions
- Comparing the E1 vs SN1 Reactions
- Elimination (E1) Reactions With Rearrangements
- E1cB - Elimination (Unimolecular) Conjugate Base
- Elimination (E1) Practice Problems And Solutions
- Elimination (E2) Practice Problems and Solutions
10 Rearrangements
11 SN1/SN2/E1/E2 Decision
- Identifying Where Substitution and Elimination Reactions Happen
- Deciding SN1/SN2/E1/E2 (1) - The Substrate
- Deciding SN1/SN2/E1/E2 (2) - The Nucleophile/Base
- SN1 vs E1 and SN2 vs E2 : The Temperature
- Deciding SN1/SN2/E1/E2 - The Solvent
- Wrapup: The Key Factors For Determining SN1/SN2/E1/E2
- Alkyl Halide Reaction Map And Summary
- SN1 SN2 E1 E2 Practice Problems
12 Alkene Reactions
- E and Z Notation For Alkenes (+ Cis/Trans)
- Alkene Stability
- Alkene Addition Reactions: "Regioselectivity" and "Stereoselectivity" (Syn/Anti)
- Stereoselective and Stereospecific Reactions
- Hydrohalogenation of Alkenes and Markovnikov's Rule
- Hydration of Alkenes With Aqueous Acid
- Rearrangements in Alkene Addition Reactions
- Halogenation of Alkenes and Halohydrin Formation
- Oxymercuration Demercuration of Alkenes
- Hydroboration Oxidation of Alkenes
- m-CPBA (meta-chloroperoxybenzoic acid)
- OsO4 (Osmium Tetroxide) for Dihydroxylation of Alkenes
- Palladium on Carbon (Pd/C) for Catalytic Hydrogenation of Alkenes
- Cyclopropanation of Alkenes
- A Fourth Alkene Addition Pattern - Free Radical Addition
- Alkene Reactions: Ozonolysis
- Summary: Three Key Families Of Alkene Reaction Mechanisms
- Synthesis (4) - Alkene Reaction Map, Including Alkyl Halide Reactions
- Alkene Reactions Practice Problems
13 Alkyne Reactions
- Acetylides from Alkynes, And Substitution Reactions of Acetylides
- Partial Reduction of Alkynes With Lindlar's Catalyst
- Partial Reduction of Alkynes With Na/NH3 To Obtain Trans Alkenes
- Alkyne Hydroboration With "R2BH"
- Hydration and Oxymercuration of Alkynes
- Hydrohalogenation of Alkynes
- Alkyne Halogenation: Bromination and Chlorination of Alkynes
- Oxidation of Alkynes With O3 and KMnO4
- Alkenes To Alkynes Via Halogenation And Elimination Reactions
- Alkynes Are A Blank Canvas
- Synthesis (5) - Reactions of Alkynes
- Alkyne Reactions Practice Problems With Answers
14 Alcohols, Epoxides and Ethers
- Alcohols - Nomenclature and Properties
- Alcohols Can Act As Acids Or Bases (And Why It Matters)
- Alcohols - Acidity and Basicity
- The Williamson Ether Synthesis
- Ethers From Alkenes, Tertiary Alkyl Halides and Alkoxymercuration
- Alcohols To Ethers via Acid Catalysis
- Cleavage Of Ethers With Acid
- Epoxides - The Outlier Of The Ether Family
- Opening of Epoxides With Acid
- Epoxide Ring Opening With Base
- Making Alkyl Halides From Alcohols
- Tosylates And Mesylates
- PBr3 and SOCl2
- Elimination Reactions of Alcohols
- Elimination of Alcohols To Alkenes With POCl3
- Alcohol Oxidation: "Strong" and "Weak" Oxidants
- Demystifying The Mechanisms of Alcohol Oxidations
- Protecting Groups For Alcohols
- Thiols And Thioethers
- Calculating the oxidation state of a carbon
- Oxidation and Reduction in Organic Chemistry
- Oxidation Ladders
- SOCl2 Mechanism For Alcohols To Alkyl Halides: SN2 versus SNi
- Alcohol Reactions Roadmap (PDF)
- Alcohol Reaction Practice Problems
- Epoxide Reaction Quizzes
- Oxidation and Reduction Practice Quizzes
15 Organometallics
- What's An Organometallic?
- Formation of Grignard and Organolithium Reagents
- Organometallics Are Strong Bases
- Reactions of Grignard Reagents
- Protecting Groups In Grignard Reactions
- Synthesis Problems Involving Grignard Reagents
- Grignard Reactions And Synthesis (2)
- Organocuprates (Gilman Reagents): How They're Made
- Gilman Reagents (Organocuprates): What They're Used For
- The Heck, Suzuki, and Olefin Metathesis Reactions (And Why They Don't Belong In Most Introductory Organic Chemistry Courses)
- Reaction Map: Reactions of Organometallics
- Grignard Practice Problems
16 Spectroscopy
- Degrees of Unsaturation (or IHD, Index of Hydrogen Deficiency)
- Conjugation And Color (+ How Bleach Works)
- Introduction To UV-Vis Spectroscopy
- UV-Vis Spectroscopy: Absorbance of Carbonyls
- UV-Vis Spectroscopy: Practice Questions
- Bond Vibrations, Infrared Spectroscopy, and the "Ball and Spring" Model
- Infrared Spectroscopy: A Quick Primer On Interpreting Spectra
- IR Spectroscopy: 4 Practice Problems
- 1H NMR: How Many Signals?
- Homotopic, Enantiotopic, Diastereotopic
- Diastereotopic Protons in 1H NMR Spectroscopy: Examples
- 13-C NMR - How Many Signals
- Liquid Gold: Pheromones In Doe Urine
- Natural Product Isolation (1) - Extraction
- Natural Product Isolation (2) - Purification Techniques, An Overview
- Structure Determination Case Study: Deer Tarsal Gland Pheromone
17 Dienes and MO Theory
- What To Expect In Organic Chemistry 2
- Are these molecules conjugated?
- Conjugation And Resonance In Organic Chemistry
- Bonding And Antibonding Pi Orbitals
- Molecular Orbitals of The Allyl Cation, Allyl Radical, and Allyl Anion
- Pi Molecular Orbitals of Butadiene
- Reactions of Dienes: 1,2 and 1,4 Addition
- Thermodynamic and Kinetic Products
- More On 1,2 and 1,4 Additions To Dienes
- s-cis and s-trans
- The Diels-Alder Reaction
- Cyclic Dienes and Dienophiles in the Diels-Alder Reaction
- Stereochemistry of the Diels-Alder Reaction
- Exo vs Endo Products In The Diels Alder: How To Tell Them Apart
- HOMO and LUMO In the Diels Alder Reaction
- Why Are Endo vs Exo Products Favored in the Diels-Alder Reaction?
- Diels-Alder Reaction: Kinetic and Thermodynamic Control
- The Retro Diels-Alder Reaction
- The Intramolecular Diels Alder Reaction
- Regiochemistry In The Diels-Alder Reaction
- The Cope and Claisen Rearrangements
- Electrocyclic Reactions
- Electrocyclic Ring Opening And Closure (2) - Six (or Eight) Pi Electrons
- Diels Alder Practice Problems
- Molecular Orbital Theory Practice
18 Aromaticity
- Introduction To Aromaticity
- Rules For Aromaticity
- Huckel's Rule: What Does 4n+2 Mean?
- Aromatic, Non-Aromatic, or Antiaromatic? Some Practice Problems
- Antiaromatic Compounds and Antiaromaticity
- The Pi Molecular Orbitals of Benzene
- The Pi Molecular Orbitals of Cyclobutadiene
- Frost Circles
- Aromaticity Practice Quizzes
19 Reactions of Aromatic Molecules
- Electrophilic Aromatic Substitution: Introduction
- Activating and Deactivating Groups In Electrophilic Aromatic Substitution
- Electrophilic Aromatic Substitution - The Mechanism
- Ortho-, Para- and Meta- Directors in Electrophilic Aromatic Substitution
- Understanding Ortho, Para, and Meta Directors
- Why are halogens ortho- para- directors?
- Disubstituted Benzenes: The Strongest Electron-Donor "Wins"
- Electrophilic Aromatic Substitutions (1) - Halogenation of Benzene
- Electrophilic Aromatic Substitutions (2) - Nitration and Sulfonation
- EAS Reactions (3) - Friedel-Crafts Acylation and Friedel-Crafts Alkylation
- Intramolecular Friedel-Crafts Reactions
- Nucleophilic Aromatic Substitution (NAS)
- Nucleophilic Aromatic Substitution (2) - The Benzyne Mechanism
- Reactions on the "Benzylic" Carbon: Bromination And Oxidation
- The Wolff-Kishner, Clemmensen, And Other Carbonyl Reductions
- More Reactions on the Aromatic Sidechain: Reduction of Nitro Groups and the Baeyer Villiger
- Aromatic Synthesis (1) - "Order Of Operations"
- Synthesis of Benzene Derivatives (2) - Polarity Reversal
- Aromatic Synthesis (3) - Sulfonyl Blocking Groups
- Birch Reduction
- Synthesis (7): Reaction Map of Benzene and Related Aromatic Compounds
- Aromatic Reactions and Synthesis Practice
- Electrophilic Aromatic Substitution Practice Problems
20 Aldehydes and Ketones
- What's The Alpha Carbon In Carbonyl Compounds?
- Nucleophilic Addition To Carbonyls
- Aldehydes and Ketones: 14 Reactions With The Same Mechanism
- Sodium Borohydride (NaBH4) Reduction of Aldehydes and Ketones
- Grignard Reagents For Addition To Aldehydes and Ketones
- Wittig Reaction
- Hydrates, Hemiacetals, and Acetals
- Imines - Properties, Formation, Reactions, and Mechanisms
- All About Enamines
- Breaking Down Carbonyl Reaction Mechanisms: Reactions of Anionic Nucleophiles (Part 2)
- Aldehydes Ketones Reaction Practice
21 Carboxylic Acid Derivatives
- Nucleophilic Acyl Substitution (With Negatively Charged Nucleophiles)
- Addition-Elimination Mechanisms With Neutral Nucleophiles (Including Acid Catalysis)
- Basic Hydrolysis of Esters - Saponification
- Transesterification
- Proton Transfer
- Fischer Esterification - Carboxylic Acid to Ester Under Acidic Conditions
- Lithium Aluminum Hydride (LiAlH4) For Reduction of Carboxylic Acid Derivatives
- LiAlH[Ot-Bu]3 For The Reduction of Acid Halides To Aldehydes
- Di-isobutyl Aluminum Hydride (DIBAL) For The Partial Reduction of Esters and Nitriles
- Amide Hydrolysis
- Thionyl Chloride (SOCl2) And Conversion of Carboxylic Acids to Acid Halides
- Diazomethane (CH2N2)
- Carbonyl Chemistry: Learn Six Mechanisms For the Price Of One
- Making Music With Mechanisms (PADPED)
- Carboxylic Acid Derivatives Practice Questions
22 Enols and Enolates
- Keto-Enol Tautomerism
- Enolates - Formation, Stability, and Simple Reactions
- Kinetic Versus Thermodynamic Enolates
- Aldol Addition and Condensation Reactions
- Reactions of Enols - Acid-Catalyzed Aldol, Halogenation, and Mannich Reactions
- Claisen Condensation and Dieckmann Condensation
- Decarboxylation
- The Malonic Ester and Acetoacetic Ester Synthesis
- The Michael Addition Reaction and Conjugate Addition
- The Robinson Annulation
- Haloform Reaction
- The Hell–Volhard–Zelinsky Reaction
- Enols and Enolates Practice Quizzes
23 Amines
- The Amide Functional Group: Properties, Synthesis, and Nomenclature
- Basicity of Amines And pKaH
- 5 Key Basicity Trends of Amines
- The Mesomeric Effect And Aromatic Amines
- Nucleophilicity of Amines
- Alkylation of Amines (Sucks!)
- Reductive Amination
- The Gabriel Synthesis
- Some Reactions of Azides
- The Hofmann Elimination
- The Hofmann and Curtius Rearrangements
- The Cope Elimination
- Protecting Groups for Amines - Carbamates
- The Strecker Synthesis of Amino Acids
- Introduction to Peptide Synthesis
- Reactions of Diazonium Salts: Sandmeyer and Related Reactions
- Amine Practice Questions
24 Carbohydrates
- D and L Notation For Sugars
- Pyranoses and Furanoses: Ring-Chain Tautomerism In Sugars
- What is Mutarotation?
- Reducing Sugars
- The Big Damn Post Of Carbohydrate-Related Chemistry Definitions
- The Haworth Projection
- Converting a Fischer Projection To A Haworth (And Vice Versa)
- Reactions of Sugars: Glycosylation and Protection
- The Ruff Degradation and Kiliani-Fischer Synthesis
- Isoelectric Points of Amino Acids (and How To Calculate Them)
- Carbohydrates Practice
- Amino Acid Quizzes
25 Fun and Miscellaneous
- A Gallery of Some Interesting Molecules From Nature
- Screw Organic Chemistry, I'm Just Going To Write About Cats
- On Cats, Part 1: Conformations and Configurations
- On Cats, Part 2: Cat Line Diagrams
- On Cats, Part 4: Enantiocats
- On Cats, Part 6: Stereocenters
- Organic Chemistry Is Shit
- The Organic Chemistry Behind "The Pill"
- Maybe they should call them, "Formal Wins" ?
- Why Do Organic Chemists Use Kilocalories?
- The Principle of Least Effort
- Organic Chemistry GIFS - Resonance Forms
- Reproducibility In Organic Chemistry
- What Holds The Nucleus Together?
- How Reactions Are Like Music
- Organic Chemistry and the New MCAT
26 Organic Chemistry Tips and Tricks
- Common Mistakes: Formal Charges Can Mislead
- Partial Charges Give Clues About Electron Flow
- Draw The Ugly Version First
- Organic Chemistry Study Tips: Learn the Trends
- The 8 Types of Arrows In Organic Chemistry, Explained
- Top 10 Skills To Master Before An Organic Chemistry 2 Final
- Common Mistakes with Carbonyls: Carboxylic Acids... Are Acids!
- Planning Organic Synthesis With "Reaction Maps"
- Alkene Addition Pattern #1: The "Carbocation Pathway"
- Alkene Addition Pattern #2: The "Three-Membered Ring" Pathway
- Alkene Addition Pattern #3: The "Concerted" Pathway
- Number Your Carbons!
- The 4 Major Classes of Reactions in Org 1
- How (and why) electrons flow
- Grossman's Rule
- Three Exam Tips
- A 3-Step Method For Thinking Through Synthesis Problems
- Putting It Together
- Putting Diels-Alder Products in Perspective
- The Ups and Downs of Cyclohexanes
- The Most Annoying Exceptions in Org 1 (Part 1)
- The Most Annoying Exceptions in Org 1 (Part 2)
- The Marriage May Be Bad, But the Divorce Still Costs Money
- 9 Nomenclature Conventions To Know
- Nucleophile attacks Electrophile
27 Case Studies of Successful O-Chem Students
- Success Stories: How Corina Got The The "Hard" Professor - And Got An A+ Anyway
- How Helena Aced Organic Chemistry
- From a "Drop" To B+ in Org 2 – How A Hard Working Student Turned It Around
- How Serge Aced Organic Chemistry
- Success Stories: How Zach Aced Organic Chemistry 1
- Success Stories: How Kari Went From C– to B+
- How Esther Bounced Back From a "C" To Get A's In Organic Chemistry 1 And 2
- How Tyrell Got The Highest Grade In Her Organic Chemistry Course
- This Is Why Students Use Flashcards
- Success Stories: How Stu Aced Organic Chemistry
- How John Pulled Up His Organic Chemistry Exam Grades
- Success Stories: How Nathan Aced Organic Chemistry (Without It Taking Over His Life)
- How Chris Aced Org 1 and Org 2
- Interview: How Jay Got an A+ In Organic Chemistry
- How to Do Well in Organic Chemistry: One Student's Advice
- "America's Top TA" Shares His Secrets For Teaching O-Chem
- "Organic Chemistry Is Like..." - A Few Metaphors
- How To Do Well In Organic Chemistry: Advice From A Tutor
- Guest post: "I went from being afraid of tests to actually looking forward to them".
Thanks a lot Sir! This website is saving me in my JEE Prep. Finally found a good website that has everything in a really good way.
Sir, another way we have been taught to convert carbonyl to CH2 is by using red phosphorus(it’s a good reducing agent) along with hydroiodic acid. Can you please also include that reaction.
i came across a question where under clemmensen conditions an amide was changed into COOH in presence of ketone group but how is this possible as it is said that it selectively reduces carbonyl group and please tell its mech.
also please tell that if on beta carbon an alkyl group is present will it undergo reduction via elimination forming a double bond between alpha and beta?please tell the mechanism
Might be the aqueous acidic nature of the Clemmensen resulting in cleavage of the amide? Clemmensen not exactly known for its functional group tolerance.
Why doesnt the carbonyl group in the ester not get reduced?
In general esters and other carboxylic acid derivatives are less reactive towards nucleophiles than ketones. Furthermore the OR group of the ester can act as a leaving group, so in the end what could just happen is nucleophilic acyl substitution at the ester.
Why wolf kishner do not reduce cooh ?
Under basic conditions the carboxylic acid is deprotonated to give a carboxylate, and these do not readily undergo addition to the C=O group (unlike ketones)
Please,I would appreciate it alot if the quizzes solution were dropped aswell,I feel it will go a long way in helping out alot of us.
“Quizzes solution were dropped?” can you explain?
Hey, Could you please clarify why don’t it reduce the -COOH group?
The Wolff Kishner does not reduce the COOH because under the basic conditions, a carboxylate is formed and carboxylates COO(-) do not readily undergo nucleophilic attack at the carbonyl carbon.
https://www.masterorganicchemistry.com/2022/09/09/nucleophilic-addition/
I am not 100% sure about the fate of carboxylic acids in the Clemmensen as it is not used very often (in practice) and the last good review on it was written in like 1944.
Hey ! I am a little confused whether Wolf-Kishner Reduction would substitute a Cl attached to a benzene with an OH . Since the Cl – C bond is partially double bond (due to resonance) , is it still possible for hydrazine to replace it ?
Hi Lucy, no it would not. That would be an example of nucleophilic acyl substitution. While it is possible in some cases to replace a Cl on an aromatic ring with OH, it generally requires very strong base and very high temperatures. See here – https://www.masterorganicchemistry.com/2018/09/17/nucleophilic-aromatic-substitution-2-benzyne/
Oh thank you very much for clarifying! I Highly appreciate your quick reply!
Hey my friend! Great stuff and awesome explanation. I have a question!
Is Zinc amalgam and concentrated HCl (the reagent used in clemmensen reduction) stronger reducing agent than LiAlH4?
I think so because LiAlH4 only reduces carbonyl compounds to Alcohols but Zinc amalgam and concentrated HCl can reduce it directly to Alcohols.
I don’t think so.
The Clemmensen really isn’t all that useful, to be honest. The last really good review on it was written in 1944. It is good for ketones adjacent to benzene rings and that’s about it.
The Clemmensen wont reduce esters or amides, for example, whereas LiAlH4 does.
Why does Clemmenson reduction reduce NO2 to NH2 alongside carbonyl compound, but wolf kishner reaction does not?
Zinc amalgam can reduce many functional groups besides ketones, including nitro groups.
Reduction in the wolff-kishner is through formation of a hydrazone; nitro groups do not undergo any reaction with hydrazine in the absence of any metal catalyst.
Sir, does the HCl used in Clemmensen Reduction also add to double bond in the reactant? (If present)
It’s a possibilty. I don’t personally recommend actually using the Clemmensen in the lab if you have any concerns about functional group compatibility.
Hey, would the aromatic ring itself be reduced in catalytic hydrogenation given that high pressure is used?
Thanks.
It’s very possible to reduce the ketone without touching the aromatic ring. No, the aromatic ring is much more resistant to reduction.
Oh- is no a better leaving group then how oh- will leave in mechanism of Wolff Kishner reaction
Yes, generally HO(-) is a poor leaving group gnerally speaking, but it is a weaker base (and better leaving group) than deprotonated nitrogen.
Hey, just a little doubt, does the Wolff-Kishner reduction work for aldehydes a well?
Also does it work on non-aromatic compounds too?
Yes – it does work for aldehydes, best done under slightly modified conditions (DMSO as solvent, KOtBu as base). https://pubs.acs.org/doi/10.1021/ja00868a048
Hey man, great content. Quick question tho: during the Wolff Kischner reaction, you mention that the hydrazone proton has a pKa of 21, and that the base that deprotanates is likely to be the conjugate base of ethylene glycol. How is it then possible? Using pKa values this doesn’t add up as alcohol groups have pKa around 16 (leading to stronger acid).
A good rule of thumb is that if the pKa difference is 8 or less, then the acid-base reaction is energetically accessible enough for the reaction to proceed. Plus with the Wolff-Kishner there’s enormous heat added.